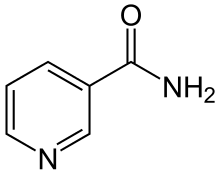
Nicotinamide
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌnaɪəˈsɪnəmaɪd/, /ˌnɪkəˈtɪnəmaɪd/ |
| Other names | NAM, 3-pyridinecarboxamide niacinamide nicotinic acid amide vitamin PP nicotinic amide vitamin B3 |
| AHFS/Drugs.com | Consumer Drug Information |
| License data |
|
topical | |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| Density | 1.40 g/cm3 g/cm3 [1] |
| Melting point | 129.5 °C (265.1 °F) |
| Boiling point | 334 °C (633 °F) |
| |
| |
Niacinamide or nicotinamide is a form of
Side effects are minimal.
Niacinamide was discovered between 1935 and 1937.
Medical uses
Niacin deficiency
Niacinamide is the preferred treatment for pellagra, caused by niacin deficiency.[3]
Acne
Niacinamide
Niacinamide increases the biosynthesis of
Skin cancer
Niacinamide at doses of 500 to 1000 mg a day decreases the risk of skin cancers, other than melanoma, in those at high risk.[21]
Side effects
Niacinamide has minimal side effects.
Chemistry
The structure of nicotinamide consists of a
from

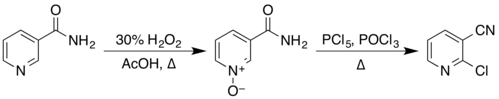
Industrial production
The hydrolysis of
Biochemistry

Nicotinamide, as a part of the cofactor
NAD+ acts as an electron carrier that mediates the interconversion of energy between nutrients and the cell's energy currency, adenosine triphosphate (ATP). In oxidation-reduction reactions, the active part of the cofactor is the nicotinamide. In NAD+, the nitrogen in the aromatic nicotinamide ring is covalently bonded to adenine dinucleotide. The formal charge on the nitrogen is stabilized by the shared electrons of the other carbon atoms in the aromatic ring. When a hydride atom is added onto NAD+ to form NADH, the molecule loses its aromaticity, and therefore a good amount of stability. This higher energy product later releases its energy with the release of a hydride, and in the case of the electron transport chain, it assists in forming adenosine triphosphate.[33]
When one mole of NADH is oxidized, 158.2 kJ of energy will be released.[33]
Biological role
Nicotinamide occurs as a component of a variety of biological systems, including within the
Nicotinamide can be methylated in the liver to biologically active 1-Methylnicotinamide when there are sufficient methyl donors.
Food sources
Niacinamide occurs in trace amounts mainly in meat, fish, nuts, and mushrooms, as well as to a lesser extent in some vegetables.[34] It is commonly added to cereals and other foods. Many multivitamins contain 20–30 mg of vitamin B3 and it is also available in higher doses.[35]
Compendial status
Research
A 2015 trial found niacinamide to reduce the rate of new nonmelanoma skin cancers and actinic keratoses in a group of people at high risk for the conditions.[38]
Niacinamide has been investigated for many additional disorders, including treatment of bullous pemphigoid nonmelanoma skin cancers.[39]
Niacinamide may be beneficial in treating psoriasis.[40]
There is tentative evidence for a potential role of niacinamide in treating acne, rosacea, autoimmune blistering disorders, ageing skin, and atopic dermatitis.[39] Niacinamide also inhibits poly(ADP-ribose) polymerases (PARP-1), enzymes involved in the rejoining of DNA strand breaks induced by radiation or chemotherapy.[41] ARCON (accelerated radiotherapy plus carbogen inhalation and nicotinamide) has been studied in cancer.[42]
Research has suggested niacinamide may play a role in the treatment of HIV.[43]
References
- ^ Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ISBN 978-1-139-43773-8. Archivedfrom the original on 30 December 2016.
- ^ ISBN 978-92-4-154765-9.
- ^ ISBN 978-0-85711-156-2.
- PMID 16029679.
- ^ (PDF) from the original on 22 September 2017. Retrieved 20 April 2018.
- ^ PMID 22646128.
- ^ a b "Niacinamide Use During Pregnancy". Drugs.com. Archived from the original on 30 December 2016. Retrieved 29 December 2016.
- ^ a b c "Niacinamide: Indications, Side Effects, Warnings". Drugs.com. 6 June 2017. Archived from the original on 5 August 2017. Retrieved 30 June 2017.
- ^ ISBN 978-3-642-12264-4. Archivedfrom the original on 10 April 2017.
- ISBN 978-1-4557-5942-2. Archivedfrom the original on 30 December 2016.
- ISBN 978-0-470-01552-0. Archivedfrom the original on 30 December 2016.
- ^ ISBN 978-3-527-30385-4.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ ISBN 978-3-13-176611-3. Archivedfrom the original on 5 November 2017.
- PMID 16871774.
- S2CID 21874670.
- S2CID 36713665.
- PMID 12133981.
- PMID 30698874.
- ; Collected Volumes, vol. 4, p. 704.
- ; Collected Volumes, vol. 4, p. 166.
- ; Collected Volumes, vol. 4, p. 706.
- ; Collected Volumes, vol. 4, p. 45.
- PMID 16347686.
- from the original on 5 November 2017.
- ISBN 978-3-13-176611-3. Archivedfrom the original on 5 November 2017.
- doi:10.1039/A809538H.
- ISBN 978-3-13-176611-3. Archivedfrom the original on 5 November 2017.
- ^ (PDF) from the original on 27 September 2007.
- PMID 15728403.
- ^ a b Casiday R, Herman C, Frey R (5 September 2008). "Energy for the Body: Oxidative Phosphorylation". www.chemistry.wustl.edu. Department of Chemistry, Washington University in St. Louis. Archived from the original on 22 November 2016. Retrieved 14 March 2017.
- S2CID 28160151.
- ^ Ranaweera A (2017). "Nicotinamide". DermNet New Zealand (www.dermnetnz.org). DermNet New Zealand Trust. Archived from the original on 25 March 2017. Retrieved 30 June 2017.
- ^ British Pharmacopoeia Commission Secretariat (2009). Index, BP 2009 (PDF). Archived from the original (PDF) on 22 July 2011. Retrieved 4 February 2010.
- ^ Japanese Pharmacopoeia (PDF) (15th ed.). 2006. Archived from the original (PDF) on 22 July 2011. Retrieved 4 February 2010.
- PMID 28681504.
- ^ S2CID 45745255.
- S2CID 39752891.
- ^ "Definition of niacinamide". NCI Drug Dictionary. National Cancer Institute. 2 February 2011. Archived from the original on 28 April 2015. Retrieved 30 June 2017.
- PMID 12473514.
- ^ Mandavilli A (7 July 2020). "Patient Is Reported Free of H.I.V., but Scientists Urge Caution". The New York Times. Archived from the original on 23 September 2020. Retrieved 22 September 2020.