Nicotine
 Top: Concentrated nicotine liquid
Bottom left: Skeleton representation of a nicotine molecule Bottom right: Ball-and-stick model of a nicotine molecule | |||
| |||
| Clinical data | |||
|---|---|---|---|
| Trade names | Nicorette, Nicotrol | ||
| AHFS/Drugs.com | Monograph | ||
| Pregnancy category |
| ||
QP53AX13 (WHO) | |||
| Legal status | |||
| Legal status |
| ||
urine pH-dependent;[8] 10–20% (gum), 30% (inhaled); 10–30% (intranasal) | |||
| Identifiers | |||
| |||
Chiral | |||
| Density | 1.01 g/cm3 | ||
| Melting point | −79 °C (−110 °F) | ||
| Boiling point | 247 °C (477 °F) | ||
| |||
| |||
| Part of a series on |
| Tobacco |
|---|
 |
Nicotine is a
Nicotine constitutes approximately 0.6–3.0% of the dry weight of tobacco.[16] Nicotine is also present at ppb-concentrations in edible plants in the family Solanaceae, including potatoes, tomatoes, and eggplants,[17] though sources disagree on whether this has any biological significance to human consumers.[17] It functions as an antiherbivore toxin; consequently, nicotine was widely used as an insecticide in the past,[18][19] and neonicotinoids (structurally similar to nicotine), such as imidacloprid, are some of the most effective and widely used insecticides.
Nicotine is highly
Nicotine use as a tool for
Uses
Medical
The primary
Combining nicotine patch use with a faster acting nicotine replacement, like gum or spray, improves the odds of treatment success.[48]
Nicotine is being researched in clinical trials for possible benefit in treating
In contrast to recreational nicotine products, which have been designed to maximize the likelihood of addiction, nicotine replacement products (NRTs) are designed to minimize addictiveness.[41]: 112 The more quickly a dose of nicotine is delivered and absorbed, the higher the addiction risk.[33]
Pesticide
Nicotine has been used as an
Performance
Nicotine-containing products are sometimes used for the
Recreational
Nicotine is used as a
.Alcohol infused with nicotine is called nicotini.[69]
Contraindications
Nicotine use for tobacco cessation has few contraindications.[70]
It is not known whether nicotine replacement therapy is effective for smoking cessation in adolescents, as of 2014.[71] It is therefore not recommended to adolescents.[72] It is not safe to use nicotine during pregnancy or breastfeeding, although it is safer than smoking; the desirability of NRT use in pregnancy is therefore debated.[73][74][75]
Randomized trials and observational studies of nicotine replacement therapy in cardiovascular patients show no increase in adverse cardiovascular events compared to those treated with placebo.[76] Using nicotine products during cancer treatment may be contraindicated, as nicotine may promote tumour growth, but temporary use of NRTs to quit smoking may be advised for harm reduction.[77]
Adverse effects

Nicotine is classified as a poison.
| Route of administration | Dosage form | Associated side effects of nicotine | Sources |
|---|---|---|---|
| Buccal | Nicotine gum | muscle ache, burping, gum sticking to teeth, unpleasant taste, dizziness, lightheadedness, headache, and insomnia .
|
[47][78] |
| Lozenge | Nausea, dyspepsia, flatulence, headache, upper respiratory tract infections , irritation (i.e., a burning sensation), hiccups, sore throat, coughing, dry lips, and oral mucosal ulceration.
|
[47][78] | |
| Transdermal | Transdermal patch |
pruritus, burning, or erythema ), diarrhea, dyspepsia, abdominal pain, dry mouth, nausea, dizziness, nervousness or restlessness, headache, vivid dreams or other sleep disturbances, and irritability.
|
[47][78][87] |
Intranasal
|
Nasal spray | Runny nose, nasopharyngeal and ocular irritation, watery eyes, sneezing, and coughing. | [47][78][88] |
| Oral inhalation | Inhaler
|
Dyspepsia, oropharyngeal irritation (e.g., coughing, irritation of the mouth and throat), rhinitis, and headache. | [47][78][89] |
| All (nonspecific) | Peripheral vasoconstriction, tachycardia (i.e., fast heart rate), elevated blood pressure, increased alertness and cognitive performance. | [78][88] | |
Sleep
Nicotine reduces the amount of
Cardiovascular system
PMID 26788573.. You can help by adding to it . (January 2019) |
A 2018
A 2016 review of the cardiovascular toxicity of nicotine concluded, "Based on current knowledge, we believe that the cardiovascular risks of nicotine from e-cigarette use in people without cardiovascular disease are quite low. We have concerns that nicotine from e-cigarettes could pose some risk for users with cardiovascular disease."[93]
Blood pressure
In the short term, nicotine causes a transient increase in blood pressure, but in the long term, epidemiological studies generally do not show increased blood pressure or hypertension among nicotine users.[93]
Reinforcement disorders
Nicotine is highly
Normal between-cigarettes discontinuation, in unrestricted smokers, causes mild but measurable nicotine withdrawal symptoms.[35] These include mildly worse mood, stress, anxiety, cognition, and sleep, all of which briefly return to normal with the next cigarette.[35] Smokers have a worse mood than they typically would have if they were not nicotine-dependent; they experience normal moods only immediately after smoking.[35] Nicotine dependence is associated with poor sleep quality and shorter sleep duration among smokers.[102][103]
In dependent smokers, withdrawal causes impairments in memory and attention, and smoking during withdrawal returns these cognitive abilities to pre-withdrawal levels.[104] The temporarily increased cognitive levels of smokers after inhaling smoke are offset by periods of cognitive decline during nicotine withdrawal.[35] Therefore, the overall daily cognitive levels of smokers and non-smokers are roughly similar.[35]
Nicotine activates the
Cancer
This section needs more primary sources, specifically: the second paragraph of this section includes citations to primary medical sources; these need to be replaced with reviews like this one. (January 2019) |  |
Although nicotine itself does not cause cancer in humans,
Low levels of nicotine stimulate cell proliferation,
Nicotine in tobacco can form carcinogenic tobacco-specific nitrosamines through a nitrosation reaction. This occurs mostly in the curing and processing of tobacco. However, nicotine in the mouth and stomach can react to form N-nitrosonornicotine,[116] a known type 1 carcinogen,[117] suggesting that consumption of non-tobacco forms of nicotine may still play a role in carcinogenesis.[118]
Genotoxicity
Nicotine causes DNA damage in several types of human cells as judged by assays for genotoxicity such as the comet assay, cytokinesis-block micronucleus test and chromosome aberrations test. In humans, this damage can happen in primary parotid gland cells,[119] lymphocytes,[120] and respiratory tract cells.[121]
Pregnancy and breastfeeding
Nicotine has been shown to produce birth defects in some animal species, but not others;
Nicotine exposure in utero is responsible for several complications of pregnancy and birth: pregnant women who smoke are at greater risk for both miscarriage and stillbirth and infants exposed to nicotine in utero tend to have lower birth weights.[124] A McMaster University research group observed in 2010 that rats exposed to nicotine in the womb (via parenteral infusion) later in life had conditions including type 2 diabetes, obesity, hypertension, neurobehavioral defects, respiratory dysfunction, and infertility.[125]
Overdose
It is unlikely that a person would overdose on nicotine through smoking alone. The US Food and Drug Administration (FDA) stated in 2013 that there are no significant safety concerns associated with the use of more than one form of over-the-counter (OTC) nicotine replacement therapy at the same time, or using OTC NRT at the same time as another nicotine-containing product, like cigarettes.[126] The median lethal dose of nicotine in humans is unknown.[44][29] Nevertheless, nicotine has a relatively high toxicity in comparison to many other alkaloids such as caffeine, which has an LD50 of 127 mg/kg when administered to mice.[127] At sufficiently high doses, it is associated with nicotine poisoning,[41] which, while common in children (in whom poisonous and lethal levels occur at lower doses per kilogram of body weight[38]) rarely results in significant morbidity or death.[43] The estimated lower dose limit for fatal outcomes is 500–1,000 mg of ingested nicotine for an adult (6.5–13 mg/kg).[27][29]
The initial symptoms of a nicotine overdose typically include
Toxicity
Today nicotine is less commonly used in agricultural
Drug interactions
Pharmacodynamic
- Potential interaction with beta-blockers).[78]
Pharmacokinetic
Nicotine and cigarette smoke both
- Smoking cessation may decrease the metabolism of
- Possible alteration of nicotine absorption through the skin from the transdermal nicotine patch by drugs that cause vasodilation or vasoconstriction.[78]
- Possible alteration of nicotine absorption through the nasal cavity from the nicotine nasal spray by nasal vasoconstrictors (e.g., xylometazoline).[78]
- Possible alteration of nicotine absorption through oral mucosa from nicotine gum and lozenges by food and drink that modify salivary pH.[78]
Pharmacology
Pharmacodynamics
Nicotine acts as a
.Central nervous system

By binding to
Nicotine activates nicotinic receptors (particularly
Sympathetic nervous system
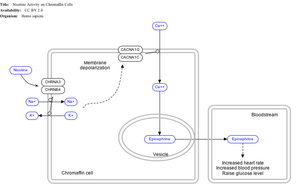
Nicotine also activates the
Adrenal medulla
By binding to
Pharmacokinetics

As nicotine enters the body, it is distributed quickly through the
The amount of nicotine absorbed by the body from smoking can depend on many factors, including the types of tobacco, whether the smoke is inhaled, and whether a filter is used. However, it has been found that the nicotine yield of individual products has only a small effect (4.4%) on the blood concentration of nicotine,[151] suggesting "the assumed health advantage of switching to lower-tar and lower-nicotine cigarettes may be largely offset by the tendency of smokers to compensate by increasing inhalation".
Nicotine has a half-life of 1–2 hours. Cotinine is an active metabolite of nicotine that remains in the blood with a half-life of 18–20 hours, making it easier to analyze.[152]
Nicotine is
Metabolism
Nicotine decreases hunger and as a consequence food consumption, alongside increasing energy expenditure.[157][158] The majority of research shows that nicotine reduces body weight, but some researchers have found that nicotine may result in weight gain under specific types of eating habits in animal models.[158] Nicotine effect on weight appears to result from nicotine's stimulation of α3β4 nAChR receptors located in the POMC neurons in the arcuate nucleus and subsequently the melanocortin system, especially the melanocortin-4 receptors on second-order neurons in the paraventricular nucleus of the hypothalamus, thus modulating feeding inhibition.[143][158] POMC neurons are a precursor of the melanocortin system, a critical regulator of body weight and peripheral tissue such as skin and hair.[158]
Chemistry
| NFPA 704 fire diamond | |
|---|---|
The fire diamond hazard sign for nicotine[159] |
Nicotine is a
Nicotine is
2.Nicotine that is found in natural tobacco is primarily (99%) the S-enantiomer.[164] Conversely, the most common chemistry synthetic methods for generating nicotine yields a product that is approximately equal proportions of the S- and R-enantiomers.[165] This suggests that tobacco-derived and synthetic nicotine can be determined by measuring the ratio of the two different enantiomers, although means exist for adjusting the relative levels of the enantiomers or performing a synthesis that only leads to the S-enantiomer. There is limited data on the relative physiological effects of these two enantiomers, especially in people. However, the studies to date indicate that (S)-nicotine is more potent than (R)-nicotine and (S)-nicotine causes stronger sensations or irritation than (R)-nicotine. To date, studies are not adequate to determine the relative addictiveness of the two enantiomers in people.
Pod mod electronic cigarettes use nicotine in the form of a protonated nicotine, rather than free-base nicotine found in earlier generations.[166]
Preparation
The first laboratory preparation of nicotine (as its
The starting material was an N-substituted pyrrole derivative, which was heated to convert it by a [1,5] sigmatropic shift to the isomer with a carbon bond between the pyrrole and pyridine rings, followed by methylation and selective reduction of the pyrrole ring using tin and hydrochloric acid.[167][168] Many other syntheses of nicotine, in both racemic and chiral forms have since been published.[169]
Biosynthesis

The biosynthetic pathway of nicotine involves a coupling reaction between the two cyclic structures that comprise nicotine. Metabolic studies show that the pyridine ring of nicotine is derived from niacin (nicotinic acid) while the pyrrolidine is derived from N-methyl-Δ1-pyrrollidium cation.[170][171] Biosynthesis of the two component structures proceeds via two independent syntheses, the NAD pathway for niacin and the tropane pathway for N-methyl-Δ1-pyrrollidium cation.
The NAD pathway in the genus
The N-methyl-Δ1-pyrrollidium cation used in the synthesis of nicotine is an intermediate in the synthesis of tropane-derived alkaloids. Biosynthesis begins with decarboxylation of ornithine by ornithine decarboxylase (ODC) to produce putrescine. Putrescine is then converted into N-methyl putrescine via methylation by SAM catalyzed by putrescine N-methyltransferase (PMT). N-methyl putrescine then undergoes deamination into 4-methylaminobutanal by the N-methyl putrescine oxidase (MPO) enzyme, 4-methylaminobutanal then spontaneously cyclize into N-methyl-Δ1-pyrrollidium cation.[citation needed]
The final step in the synthesis of nicotine is the coupling between N-methyl-Δ1-pyrrollidium cation and niacin. Although studies conclude some form of coupling between the two component structures, the definite process and mechanism remains undetermined. The current agreed theory involves the conversion of niacin into 2,5-dihydropyridine through 3,6-dihydronicotinic acid. The 2,5-dihydropyridine intermediate would then react with N-methyl-Δ1-pyrrollidium cation to form enantiomerically pure (−)-nicotine.[172]
Detection in body fluids
Nicotine can be quantified in blood, plasma, or urine to confirm a diagnosis of poisoning or to facilitate a medicolegal death investigation. Urinary or salivary cotinine concentrations are frequently measured for the purposes of pre-employment and health insurance medical screening programs. Careful interpretation of results is important, since passive exposure to cigarette smoke can result in significant accumulation of nicotine, followed by the appearance of its metabolites in various body fluids.[173][174] Nicotine use is not regulated in competitive sports programs.[175]
Methods for analysis of enantiomers
Methods for measuring the two enantiomers are straightforward and include normal-phase liquid chromatography,[164] liquid chromatography with a chiral column.[176] However, since methods can be used to alter the two enantiomers, it may not be possible to distinguish tobacco-derived from synthetic nicotine simply by measuring the levels of the two enantiomers. A new approach uses hydrogen and deuterium nuclear magnetic resonance to distinguish tobacco-derived and synthetic nicotine based on differences the substrates used in the natural synthetic pathway performed in the tobacco plant and the substrates most used in synthesis.[177] Another approach measures the carbon-14 content which also differs between natural and laboratory-based tobacco.[178] These methods remain to be fully evaluated and validated using a wide range of samples.
Natural occurrence
Nicotine is a secondary metabolite produced in a variety of plants in the family Solanaceae, most notably in tobacco Nicotiana tabacum, where it can be found at high concentrations of 0.5 to 7.5%.[179] Nicotine is also found in the leaves of other tobacco species, such as Nicotiana rustica (in amounts of 2–14%). Nicotine production is strongly induced in response to wounding as part of a jasmonate-dependent reaction.[180] Specialist insects on tobacco, such as the tobacco hornworm (Manduca sexta), have a number of adaptations to the detoxification and even adaptive re-purposing of nicotine.[181] Nicotine is also found at low concentrations in the nectar of tobacco plants, where it may promote outcrossing by affecting the behavior of hummingbird pollinators.[182]
Nicotine occurs in smaller amounts (varying from 2–7 μg/kg, or 20–70 millionths of a percent wet weight[17]) in other Solanaceaeous plants, including some crop species such as potatoes, tomatoes, eggplant, and peppers,[17][183] as well as non-crop species such as Duboisia hopwoodii.[160] The amounts of nicotine in tomatoes lowers substantially as the fruit ripens.[17] A 1999 report found "In some papers it is suggested that the contribution of dietary nicotine intake is significant when compared with exposure to ETS [environmental tobacco smoke] or by active smoking of small numbers of cigarettes. Others consider the dietary intake to be negligible unless inordinately large amounts of specific vegetables are consumed."[17] The amount of nicotine eaten per day is roughly around 1.4 and 2.25 μg/day at the 95th percentile.[17] These numbers may be low due to insufficient food intake data.[17] The concentrations of nicotine in vegetables are difficult to measure accurately, since they are very low (parts per billion range).[184]
History, society and culture
Nicotine was originally isolated from the tobacco plant in 1828 by chemists Wilhelm Heinrich Posselt and Karl Ludwig Reimann from Germany, who believed it was a poison.[185][186] Its chemical empirical formula was described by Melsens in 1843,[187] its structure was discovered by Adolf Pinner and Richard Wolffenstein in 1893,[188][189][190][clarification needed] and it was first synthesized by Amé Pictet and A. Rotschy in 1904.[167][191]
Nicotine is named after the tobacco plant Nicotiana tabacum, which in turn is named after the French ambassador in Portugal, Jean Nicot de Villemain, who sent tobacco and seeds to Paris in 1560, presented to the French King,[192] and who promoted their medicinal use. Smoking was believed to protect against illness, particularly the plague.[192]
Tobacco was introduced to Europe in 1559, and by the late 17th century, it was used not only for smoking but also as an insecticide. After World War II, over 2,500 tons of nicotine insecticide were used worldwide, but by the 1980s the use of nicotine insecticide had declined below 200 tons. This was due to the availability of other insecticides that are cheaper and less harmful to mammals.[19]
The nicotine content of popular American-brand cigarettes has increased over time, and one study found that there was an average increase of 1.78% per year between the years of 1998 and 2005.[193]
Although methods of production of synthetic nicotine have existed for decades,[194] it was believed that the cost of making nicotine by laboratory synthesis was cost prohibitive compared to extracting nicotine from tobacco.[195] However, recently synthetic nicotine started to be found in different brands of e-cigarettes and oral pouches and marketed as "tobacco-free."[196]
The US FDA is tasked with reviewing tobacco products such as e-cigarettes and determining which can be authorized for sale. In response to the likelihood that FDA would not authorize many e-cigarettes to be marketed, e-cigarette companies began marketing products that they claimed to contain nicotine that were not made or derived from tobacco, but contained synthetic nicotine instead, and thus, would be outside FDA's tobacco regulatory authority.[197] Similarly, nicotine pouches that claimed to contain non-tobacco (synthetic) nicotine were also introduced. The cost of synthetic nicotine has decreased as the market for the product increased. In March 2022, the U.S. Congress passed a law (the Consolidated Appropriations Act, 2022) that expanded FDA's tobacco regulatory authority to include tobacco products containing nicotine from any source, thereby including products made with synthetic nicotine.
Legal status
In the United States, nicotine products and nicotine replacement therapy products like Nicotrol are only available to people 18 and above; proof of age is required; not for sale in vending machine or from any source where proof of age cannot be verified. As of 2019, the minimum age to sell tobacco in the US is 21 at the federal level.[198]
In the European Union, the minimum age to purchase nicotine products is 18. However, there is no minimum age requirement to use tobacco or nicotine products.[199]
In the United Kingdom, the Tobacco and Related Products Regulations 2016 implemented the European directive 2014/40/EU, amended by Tobacco Products and Nicotine Inhaling Products (Amendment etc.) (EU Exit) Regulations 2019 and the Tobacco Products and Nicotine Inhaling Products (Amendment) (EU Exit) Regulations 2020. Additionally other regulations limit advertising, sale and display of tobacco products and other products containing nicotine for human consumption. The Sunak government has proposed banning disposable vapes, to limit the appeal, and affordability, for children.
In media
| External image | |
|---|---|
In some
Nicotine was often compared to caffeine in advertisements in the 1980s by the tobacco industry, and later in the 2010s by the electronic cigarettes industry, in an effort to reduce the stigmatization and the public perception of the risks associated with nicotine use.[201]
Research
Central nervous system
While acute/initial nicotine intake causes activation of neuronal nicotine receptors, chronic low doses of nicotine use leads to desensitization of those receptors (due to the development of tolerance) and results in an antidepressant effect, with early research showing low dose nicotine patches could be an effective treatment of major depressive disorder in non-smokers.[202]
Though tobacco smoking is associated with an increased risk of Alzheimer's disease,[203] there is evidence that nicotine itself has the potential to prevent and treat Alzheimer's disease.[204]
Smoking is associated with a decreased risk of Parkinson's disease; however, it is unknown whether this is due to people with healthier brain dopaminergic reward centers (the area of the brain affected by Parkinson's) being more likely to enjoy smoking and thus pick up the habit, nicotine directly acting as a neuroprotective agent, or other compounds in cigarette smoke acting as neuroprotective agents.[205]
Immune system
Immune cells of both the
Optopharmacology
A
Oral health
Several in vitro studies have investigated the potential effects of nicotine on a range of oral cells. A recent systematic review concluded that nicotine was unlikely to be cytotoxic to oral cells in vitro in most physiological conditions but further research is needed.[208] Understanding the potential role of nicotine in oral health has become increasingly important given the recent introduction of novel nicotine products and their potential role in helping smokers quit.[209]
See also
References
- PMID 25638335.
- ^ PMID 22003417.
Withdrawal symptoms upon cessation of nicotine intake: Chronic nicotine use induces neuroadaptations in the brain's reward system that result in the development of nicotine dependence. Thus, nicotine-dependent smokers must continue nicotine intake to avoid distressing somatic and affective withdrawal symptoms. Newly abstinent smokers experience symptoms such as depressed mood, anxiety, irritability, difficulty concentrating, craving, bradycardia, insomnia, gastrointestinal discomfort, and weight gain (Shiffman and Jarvik, 1976; Hughes et al., 1991). Experimental animals, such as rats and mice, exhibit a nicotine withdrawal syndrome that, like the human syndrome, includes both somatic signs and a negative affective state (Watkins et al., 2000; Malin et al., 2006). The somatic signs of nicotine withdrawal include rearing, jumping, shakes, abdominal constrictions, chewing, scratching, and facial tremors. The negative affective state of nicotine withdrawal is characterized by decreased responsiveness to previously rewarding stimuli, a state called anhedonia.
- PMID 22114542.
- ISBN 978-1-4200-4742-4.
- FDA. Retrieved 22 October 2023.
- ^ "The Medicines (Products Other Than Veterinary Drugs) (General Sale List) Amendment Order 2001". legislation.gov.uk. Retrieved 2 August 2022.
- ^ a b Nicotine. PubChem Compound Database. United States National Library of Medicine – National Center for Biotechnology Information. 16 February 2019. Retrieved 18 April 2024.
- ^ a b c d Landoni JH. "Nicotine (PIM)". INCHEM. International Programme on Chemical Safety. Retrieved 29 January 2019.
- .
- PMID 26661236.
- ^ "Nicotine: Clinical data". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 18 April 2024.
Used as an aid to smoking cessation and for the relief of nicotine withdrawal symptoms.
- ISBN 978-1-118-68285-2.
- ^ a b c d "Nicotinic acetylcholine receptors: Introduction". IUPHAR Database. International Union of Basic and Clinical Pharmacology. Archived from the original on 29 June 2017. Retrieved 1 September 2014.
- ^ ISBN 9780071481274.
Nicotine ... is a natural alkaloid of the tobacco plant. Lobeline is a natural alkaloid of Indian tobacco. Both drugs are agonists are nicotinic cholinergic receptors ...
- PMID 24882143.
- ^ "Smoking and Tobacco Control Monograph No. 9" (PDF). Archived (PDF) from the original on 9 October 2022. Retrieved 19 December 2012.
- ^ PMID 10552617.
- LCCN 2008018913.[page needed]
- ^ a b c Ujváry I (1999). "Nicotine and Other Insecticidal Alkaloids". In Yamamoto I, Casida J (eds.). Nicotinoid Insecticides and the Nicotinic Acetylcholine Receptor. Tokyo: Springer-Verlag. pp. 29–69.
- PMID 23494236.
- ^ PMID 24821826.
- ^ PMID 27994114.
- PMID 17640334.
- S2CID 11855403.
- ^ a b "Evidence Review of E-Cigarettes and Heated Tobacco Products" (PDF). Public Health England. 2018.
- ^ "Tobacco more addictive than Nicotine". Archived from the original on 20 April 2023.
- ^ a b c d e Royal College of Physicians (28 April 2016). "Nicotine without smoke: Tobacco harm reduction". Retrieved 16 September 2020.
- ^ PMID 26955970.
- ^ PMID 24091634.
- S2CID 207223131.
- PMID 18452252.
- PMID 23631239.
- ^ S2CID 7408200.
- S2CID 11356233.
- ^ S2CID 26903942.
- S2CID 43763340.
- ^ "E-Cigarette Use Among Youth and Young Adults. 2016 Surgeon General's report.lts" (PDF). surgeongeneral.gov. Archived (PDF) from the original on 9 October 2022.
- ^ PMID 25830075.
- ^ IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Personal Habits and Indoor Combustions. Lyon (FR): International Agency for Research on Cancer; 2012. (IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 100E.) TOBACCO SMOKING. Available from: https://www.ncbi.nlm.nih.gov/books/NBK304395/
- ^ a b "Does nicotine cause cancer?". European Code Against Cancer. World Health Organization – International Agency for Research on Cancer. Retrieved 23 January 2019.
- ^ PMID 24455788.
- S2CID 29298949.
- ^ a b c d e f g h i j "Nicotine". United States National Library of Medicine – Toxicology Data Network. Hazardous Substances Data Bank. 20 August 2009.
- ^ a b "Nicotine". European Chemicals Agency: Committee for Risk Assessment. September 2015. Retrieved 23 January 2019.
- PMID 36154615.
- PMID 2050970.
- ^ PMID 29852054.
There is high-quality evidence that all of the licensed forms of NRT (gum, transdermal patch, nasal spray, inhalator and sublingual tablets/lozenges) can help people who make a quit attempt to increase their chances of successfully stopping smoking. NRTs increase the rate of quitting by 50% to 60%, regardless of setting, and further research is very unlikely to change our confidence in the estimate of the effect. The relative effectiveness of NRT appears to be largely independent of the intensity of additional support provided to the individual.
A meta-analysis of adverse events associated with NRT included 92 RCTs and 28 observational studies, and addressed a possible excess of chest pains and heart palpitations among users of NRT compared with placebo groups (Mills 2010). The authors report an OR of 2.06 (95% CI 1.51 to 2.82) across 12 studies. We replicated this data collection exercise and analysis where data were available (included and excluded) in this review, and detected a similar but slightly lower estimate, OR 1.88 (95% CI 1.37 to 2.57; 15 studies; 11,074 participants; OR rather than RR calculated for comparison; Analysis 6.1). Chest pains and heart palpitations were an extremely rare event, occurring at a rate of 2.5% in the NRT groups compared with 1.4% in the control groups in the 15 trials in which they were reported at all. A recent network meta-analysis of cardiovascular events associated with smoking cessation pharmacotherapies (Mills 2014), including 21 RCTs comparing NRT with placebo, found statistically significant evidence that the rate of cardiovascular events with NRT was higher (RR 2.29 95% CI 1.39 to 3.82). However, when only serious adverse cardiac events (myocardial infarction, stroke and cardiovascular death) were considered, the finding was not statistically significant (RR 1.95 95% CI 0.26 to 4.30). - PMID 37335995.
- ^ The MIND Study. "Why Nicotine?". MIND. Retrieved 6 December 2020.
- ^ PMID 15822177.
- LCCN 2008018913.[page needed]
- ^ "Tobacco and its evil cousin nicotine are good as a pesticide – American Chemical Society". American Chemical Society. Retrieved 29 October 2018.
- ^ USEPA (3 June 2009). "Nicotine; Product Cancellation Order". Federal Register: 26695–26696. Retrieved 8 April 2012.
- ^ US Code of Federal Regulations. 7 CFR 205.602 – Nonsynthetic substances prohibited for use in organic crop production
- ^ Tharp C (5 September 2014). "Safety for Homemade Remedies for Pest Control" (PDF). Montana Pesticide Bulletin. Montana State University. Archived from the original (PDF) on 5 September 2014. Retrieved 21 September 2020.
- ^ S2CID 22662492.
- .
- PMID 27908457.
- PMID 29110618.
- PMID 20414766.
- PMID 28607947.
- ^ "Nicotine: Biological activity". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 7 February 2016.
Kis as follows; α2β4=9900nM [5], α3β2=14nM [1], α3β4=187nM [1], α4β2=1nM [4,6]. Due to the heterogeneity of nACh channels we have not tagged a primary drug target for nicotine, although the α4β2 is reported to be the predominant high affinity subtype in the brain which mediates nicotine addiction
- S2CID 3758298.
- PMID 29606560.
- ^ S2CID 8731555.
- ^ Wilder N, Daley C, Sugarman J, Partridge J (April 2016). "Nicotine without smoke: Tobacco harm reduction". UK: Royal College of Physicians. pp. 58, 125.
- ^ S2CID 21302112.
- PMID 25745382.
- ^ "2003: The 3rd Annual Year In Ideas; Nicotini, The". www.nytimes.com. The New York Times Magazine. Archived from the original on 27 May 2015. Retrieved 28 March 2024.
- S2CID 12064318.
- PMID 23488726.
- ^ PMID 22248234.
- ^ "Electronic Cigarettes – What are the health effects of using e-cigarettes?" (PDF). Centers for Disease Control and Prevention. 22 February 2018. Archived (PDF) from the original on 9 October 2022.
Nicotine is a health danger for pregnant women and their developing babies.
- PMID 20363831.
there is no safe dose of nicotine during pregnancy... The general consensus among clinicians is that more information is needed about the risks of NRT use during pregnancy before well-informed definitive recommendations can be made to pregnant women... Overall, the evidence provided in this review overwhelmingly indicates that nicotine should no longer be considered the safe component of cigarette smoke. In fact, many of the adverse postnatal health outcomes associated with maternal smoking during pregnancy may be attributable, at least in part, to nicotine alone.
- S2CID 27085986.
- PMID 30527452.
- ^ PMID 26380225.
- ^ a b c d e f g h i j k l m n o "Nicotine". Drugs.com. American Society of Health-System Pharmacists. Retrieved 24 January 2019.
- ^ Detailed reference list is located on a separate image page.
- ^ "NICOTINE : Systemic Agent". 8 July 2021.
- ^ Royal College of Physicians. "Nicotine Without Smoke -- Tobacco Harm Reduction". p. 125. Retrieved 30 September 2020.
Use of nicotine alone, in the doses used by smokers, represents little if any hazard to the user.
- S2CID 47016482.
It is the smoke from combustible tobacco products—not nicotine—that injures and kills millions of smokers.
- PMID 27705269.
Beyond its addictive properties, short-term or long-term exposure to nicotine in adults has not been established as dangerous
- ^ PMID 29852054.
- PMID 25794473.
- ^ "Nicotine Transdermal Patch" (PDF). United States Food and Drug Administration. Retrieved 24 January 2019.
- ^ a b "Nicotrol NS" (PDF). United States Food and Drug Administration. Retrieved 24 January 2019.
- ^ "Nicotrol" (PDF). Pfizer. Retrieved 24 January 2019.
- ^ S2CID 22703103.
- PMID 15532213.
- PMID 19345124.
- ^ PMID 27079891.
- PMID 11572966.
Although the ΔFosB signal is relatively long-lived, it is not permanent. ΔFosB degrades gradually and can no longer be detected in brain after 1–2 months of drug withdrawal ... Indeed, ΔFosB is the longest-lived adaptation known to occur in adult brain, not only in response to drugs of abuse, but to any other perturbation (that doesn't involve lesions) as well.
- PMID 23430970.
The 35–37 kD ΔFosB isoforms accumulate with chronic drug exposure due to their extraordinarily long half-lives. ... As a result of its stability, the ΔFosB protein persists in neurons for at least several weeks after cessation of drug exposure. ... ΔFosB overexpression in nucleus accumbens induces NFκB
- S2CID 10076758.
- ^ PMID 15496937.
- ^ "Acetaldehyde | RIVM".
- PMID 28756728.
- PMID 20414766.
The significant effects of nicotine on motor abilities, attention, and memory likely represent true performance enhancement because they are not confounded by withdrawal relief. The beneficial cognitive effects of nicotine have implications for initiation of smoking and maintenance of tobacco dependence.
- S2CID 1267977.
- PMID 27816041.
- S2CID 1066283.
- PMID 22405889.
Discontinuation of smoking leads to negative affective symptoms such as depressed mood, increased anxiety, and impaired memory and attention...Smoking cessation leads to a relatively mild somatic withdrawal syndrome and a severe affective withdrawal syndrome that is characterized by a decrease in positive affect, an increase in negative affect, craving for tobacco, irritability, anxiety, difficulty concentrating, hyperphagia, restlessness, and a disruption of sleep. Smoking during the acute withdrawal phase reduces craving for cigarettes and returns cognitive abilities to pre-smoking cessation level
- ^ PMID 24459410.
- ^ S2CID 19157711.
The knowledge of ΔFosB induction in chronic drug exposure provides a novel method for the evaluation of substance addiction profiles (i.e. how addictive they are). Xiong et al. used this premise to evaluate the potential addictive profile of propofol (119). Propofol is a general anaesthetic, however its abuse for recreational purpose has been documented (120). Using control drugs implicated in both ΔFosB induction and addiction (ethanol and nicotine), ...
Conclusions
ΔFosB is an essential transcription factor implicated in the molecular and behavioral pathways of addiction following repeated drug exposure. The formation of ΔFosB in multiple brain regions, and the molecular pathway leading to the formation of AP-1 complexes is well understood. The establishment of a functional purpose for ΔFosB has allowed further determination as to some of the key aspects of its molecular cascades, involving effectors such as GluR2 (87,88), Cdk5 (93) and NFkB (100). Moreover, many of these molecular changes identified are now directly linked to the structural, physiological and behavioral changes observed following chronic drug exposure (60,95,97,102). New frontiers of research investigating the molecular roles of ΔFosB have been opened by epigenetic studies, and recent advances have illustrated the role of ΔFosB acting on DNA and histones, truly as a molecular switch (34). As a consequence of our improved understanding of ΔFosB in addiction, it is possible to evaluate the addictive potential of current medications (119), as well as use it as a biomarker for assessing the efficacy of therapeutic interventions (121,122,124). - S2CID 8551216.
- S2CID 11372110.
- ISBN 9780309468343.
- PMID 18844224.
- PMID 17369603.
- PMID 14651253.
- S2CID 9774853.
- license.
- PMID 25343018.
- PMID 22923602.
- ^ "List of Classifications – IARC Monographs on the Identification of Carcinogenic Hazards to Humans". monographs.iarc.fr. Retrieved 22 July 2020.
- PMID 26380225.
- PMID 24698733.
- PMID 23200805.
- PMID 22001448.
- PMID 23439891.
- ^ "State Health Officer's Report on E-Cigarettes: A Community Health Threat" (PDF). California Department of Public Health. January 2015. Archived (PDF) from the original on 9 October 2022.
- PMID 27297020.
- PMID 20363831.
- ^ "Consumer Updates: Nicotine Replacement Therapy Labels May Change". FDA. 1 April 2013.
- ^ Toxicology and Applied Pharmacology. Vol. 44, Pg. 1, 1978.
- ^ S2CID 28312730.
- S2CID 27707333.
- PMID 20652661.
- PMID 4479133.
- ^ "CDC – NIOSH Pocket Guide to Chemical Hazards – Nicotine". www.cdc.gov. Retrieved 20 November 2015.
- S2CID 23847303.
- S2CID 12080532.
- ^ Katzung BG (2006). Basic and Clinical Pharmacology. New York: McGraw-Hill Medical. pp. 99–105.
- PMID 19252481.
- ^ Nesbitt P (1969). Smoking, physiological arousal, and emotional response. Unpublished doctoral dissertation, Columbia University.
- PMID 9624709.
- PMID 27610066.
- S2CID 9393548.
- ^ ISBN 9780071481274.
- ^ S2CID 206815322. Archived from the original(PDF) on 4 August 2020. Retrieved 23 September 2019.
This reward link comprises a dopamine projection from the ventral tegmental area (VTA) to the nucleus accumbens together with a cholinergic input, arising primarily from the laterodorsal tegmental area.
- ^ PMID 23632083.
Rat studies have shown that nicotine administration can decrease food intake and body weight, with greater effects in female animals (Grunberg et al., 1987). A similar nicotine regimen also decreases body weight and fat mass in mice as a result of β4* nAChR-mediated activation of POMC neurons and subsequent activation of MC4 receptors on second order neurons in the paraventricular nucleus of the hypothalamus (Mineur et al., 2011).
- PMID 22049069.
- PMID 22049068.
- S2CID 37754794.
- ISBN 978-0-8053-5909-1.[page needed]
- ISBN 978-3-540-69248-5.
- PMID 12971663.
- PMID 7077531.
- PMID 7417765.
- ^ Bhalala O (Spring 2003). "Detection of Cotinine in Blood Plasma by HPLC MS/MS". MIT Undergraduate Research Journal. 8: 45–50. Archived from the original on 24 December 2013.
- S2CID 14374018.
- S2CID 206939025.
- ^ "The danger of third-hand smoke: Plain language summary – Petrick et al., "Thirdhand smoke: heterogeneous oxidation of nicotine and secondary aerosol formation in the indoor environment" in Environmental Science & Technology". The Column. Vol. 7, no. 3. Chromatography Online. 22 February 2011.
- S2CID 16044557.
- ^ "Nicotine' actions on energy balance: Friend or foe?". Pharmacology & Therapeutics. 219. March 2021.
- ^ S2CID 51727199.
Nicotine's weight effects appear to result especially from the drug's stimulation of α3β4 nicotine acetylcholine receptors (nAChRs), which are located on pro-opiomelanocortin (POMC) neurons in the arcuate nucleus (ARC), leading to activation of the melanocortin circuit, which is associated with body weight. Further, α7- and α4β2-containing nAChRs have been implicated in weight control by nicotine.
- ^ "NFPA Hazard Rating Information for Common Chemicals". Archived from the original on 17 February 2015. Retrieved 15 March 2015.
- ^ a b c Metcalf RL (2007), "Insect Control", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, p. 9
- ^ "L-Nicotine Material Safety Data Sheet". Sciencelab.com, Inc.
- ^ a b c Henry TA (1949). The Plant Alkaloids (PDF) (4th ed.). Philadelphia, Toronto: The Blakiston Company. pp. 36–43.
- ^ Gause GF (1941). "Chapter V: Analysis of various biological processes by the study of the differential action of optical isomers". In Luyet BJ (ed.). Optical Activity and Living Matter. A series of monographs on general physiology. Vol. 2. Normandy, Missouri: Biodynamica.
- ^ PMID 29722457.
- PMID 27943582.
- license.
- ^ .
- .
- S2CID 247687372.
- PMID 13651178.
- .
- ISBN 978-0-470-74703-2.[page needed]
- PMID 19184645.
- ISBN 978-0-9626523-9-4.
- S2CID 41954065.
- PMID 29426492.
- S2CID 197593505.
- PMID 35421170.
- ^ "Tobacco (leaf tobacco)". Transportation Information Service.
- PMID 11743088.
- ^ N.d. Natural history-driven, plant-mediated RNAi-based study reveals CYP6B46's role in a nicotine-mediated antipredator herbivore defense | PNAS.
- PMID 22448647.
- PMID 8326992.
- .
- S2CID 38290573.
- ^ Posselt W, Reimann L (1828). "Chemische Untersuchung des Tabaks und Darstellung eines eigenthümlich wirksamen Prinzips dieser Pflanze" [Chemical investigation of tobacco and preparation of a characteristically active constituent of this plant]. Magazin für Pharmacie (in German). 6 (24): 138–161.
- ^ Melsens LH (1843). "Note sur la nicotine" [Note on nicotine]. Annales de Chimie et de Physique. third series (in French). 9: 465–479, see especially page 470. [Note: The empirical formula that Melsens provides is incorrect because at that time, chemists used the wrong atomic mass for carbon (6 instead of 12).]
- .
- .
- S2CID 83703998.
- ISSN 1059-1028. Retrieved 11 October 2022.
- ^ ISBN 978-0-8089-2354-1.
- PMID 17897974.
- ^ "Industry Documents Library". www.industrydocuments.ucsf.edu. Retrieved 11 October 2022.
- ISSN 0362-4331. Retrieved 11 October 2022.
- ISSN 0362-4331. Retrieved 11 October 2022.
- PMID 34493630.
- ^ Center for Tobacco Products (26 September 2022). "Tobacco 21". FDA.
- ^ "21, 18, or 14: A look at the legal age for smoking around the world". Straits Times. 3 October 2017. Retrieved 1 March 2019.
- ^ S2CID 71246970.
- ^ Becker R (26 April 2019). "Why Big Tobacco and Big Vape love comparing nicotine to caffeine". The Verge.
- PMID 20965579.
- PMID 19105840.
- PMID 19184661.
- PMID 18683238.
- ^ PMID 28932225.
- PMID 29578537.
- S2CID 169035502.
- PMID 31171977.






