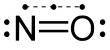Nitric oxide

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Nitrogen monoxide[1]
| |||
| Systematic IUPAC name
Oxidonitrogen(•)[2] (additive) | |||
| Other names
Nitrogen oxide
Nitrogen(II) oxide Oxonitrogen Nitrogen monoxide | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 3DMet | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.030.233 | ||
| EC Number |
| ||
| 451 | |||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1660 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| NO | |||
| Molar mass | 30.006 g·mol−1 | ||
| Appearance | Colourless gas | ||
| Density | 1.3402 g/L | ||
| Melting point | −164 °C (−263 °F; 109 K) | ||
| Boiling point | −152 °C (−242 °F; 121 K) | ||
| 0.0098 g / 100 ml (0 °C) 0.0056 g / 100 ml (20 °C) | |||
Refractive index (nD)
|
1.0002697 | ||
| Structure | |||
| linear (point group C∞v) | |||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
210.76 J/(K·mol) | ||
Std enthalpy of (ΔfH⦵298)formation |
90.29 kJ/mol | ||
| Pharmacology | |||
| R07AX01 (WHO) | |||
| License data | |||
| Inhalation | |||
| Pharmacokinetics: | |||
| good | |||
| via pulmonary capillary bed | |||
| 2–6 seconds | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
| ||
| GHS labelling: | |||
    [3][4] [3][4]
| |||
| Danger | |||
| H270, H280, H314, H330[3][4] | |||
| P220, P244, P260, P280, P303+P361+P353+P315, P304+P340+P315, P305+P351+P338+P315, P370+P376, P403, P405[3][4] | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration)
|
315 ppm (rabbit, 15 min) 854 ppm (rat, 4 h) 2500 ppm (mouse, 12 min)[5] | ||
LCLo (lowest published)
|
320 ppm (mouse)[5] | ||
| Safety data sheet (SDS) | External SDS | ||
| Related compounds | |||
Related nitrogen oxides
|
Dinitrogen pentoxide Dinitrogen tetroxide Nitroxyl (reduced form)Hydroxylamine (hydrogenated form) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Nitric oxide (nitrogen oxide or nitrogen monoxide
An important
Nitric oxide should not be confused with
Physical properties
Electronic configuration
The ground state electronic configuration of NO is, in united atom notation:[10]
The first two orbitals are actually pure atomic 1sO and 1sN from oxygen and nitrogen respectively and therefore are usually not noted in the united atom notation. Orbitals noted with an asterisk are antibonding. The ordering of 5σ and 1π according to their binding energies is subject to discussion. Removal of a 1π electron leads to 6 states whose energies span over a range starting at a lower level than a 5σ electron an extending to a higher level. This is due to the different orbital momentum couplings between a 1π and a 2π electron.
The lone electron in the 2π orbital makes NO a doublet (X ²Π) in its ground state whose degeneracy is split in the fine structure from spin-orbit coupling with a total momentum J=3⁄2 or J=1⁄2.
Dipole
The dipole of NO has been measured experimentally to 0.15740 D and is oriented from O to N (⁻NO⁺) due to the transfer of negative electronic charge from oxygen to nitrogen. [11]
Reactions
With di- and triatomic molecules
Upon condensing to a liquid, nitric oxide
Since the heat of formation of •NO is
- 2 NO → O2 + N2
When exposed to oxygen, nitric oxide converts into nitrogen dioxide:
- 2 NO + O2 → 2 NO2
This reaction is thought to occur via the intermediates ONOO• and the red compound ONOONO.[12]
In water, nitric oxide reacts with oxygen to form nitrous acid (HNO2). The reaction is thought to proceed via the following stoichiometry:
- 4 NO + O2 + 2 H2O → 4 HNO2
Nitric oxide reacts with fluorine, chlorine, and bromine to form the nitrosyl halides, such as nitrosyl chloride:
- 2 NO + Cl2 → 2 NOCl
With NO2, also a radical, NO combines to form the intensely blue dinitrogen trioxide:[6]
- NO + NO2 ⇌ ON−NO2
Organic chemistry
The addition of a nitric oxide
This reaction, which was discovered around 1898, remains of interest in nitric oxide prodrug research. Nitric oxide can also react directly with sodium methoxide, ultimately forming sodium formate and nitrous oxide by way of an N-methoxydiazeniumdiolate.[16]
Coordination complexes
Nitric oxide reacts with
Production and preparation
In commercial settings, nitric oxide is produced by the
- 4 NH3 + 5 O2 → 4 NO + 6 H2O
The uncatalyzed
- N2 + O2 → 2 NO
Laboratory methods
In the laboratory, nitric oxide is conveniently generated by reduction of dilute nitric acid with copper:
- 8 HNO3 + 3 Cu → 3 Cu(NO3)2 + 4 H2O + 2 NO
An alternative route involves the reduction of nitrous acid in the form of sodium nitrite or potassium nitrite:
- 2 NaNO2 + 2 NaI + 2 H2SO4 → I2 + 2 Na2SO4 + 2 H2O + 2 NO
- 2 NaNO2 + 2 FeSO4 + 3 H2SO4 → Fe2(SO4)3 + 2 NaHSO4 + 2 H2O + 2 NO
- 3 KNO2 + KNO3 + Cr2O3 → 2 K2CrO4 + 4 NO
The iron(II) sulfate route is simple and has been used in undergraduate laboratory experiments. So-called NONOate compounds are also used for nitric oxide generation.
Detection and assay
Nitric oxide concentration can be determined using a chemiluminescent reaction involving ozone.[17] A sample containing nitric oxide is mixed with a large quantity of ozone. The nitric oxide reacts with the ozone to produce oxygen and nitrogen dioxide, accompanied with emission of light (chemiluminescence):
- NO + O3 → NO2 + O2 + hν
which can be measured with a photodetector. The amount of light produced is proportional to the amount of nitric oxide in the sample.
Other methods of testing include electroanalysis (amperometric approach), where ·NO reacts with an electrode to induce a current or voltage change. The detection of NO radicals in biological tissues is particularly difficult due to the short lifetime and concentration of these radicals in tissues. One of the few practical methods is spin trapping of nitric oxide with iron-dithiocarbamate complexes and subsequent detection of the mono-nitrosyl-iron complex with electron paramagnetic resonance (EPR).[18][19]
A group of
Environmental effects
Acid rain deposition
Nitric oxide reacts with the hydroperoxyl radical (HO•
2) to form nitrogen dioxide (NO2), which then can react with a hydroxyl radical (•OH) to produce nitric acid (HNO3):
- •NO + HO•
2 → •NO2 + •OH - •NO2 + •OH → HNO3
Nitric acid, along with sulfuric acid, contributes to acid rain deposition.
Ozone depletion
•NO participates in
- •NO + O3 → NO2 + O2
This reaction is also utilized to measure concentrations of •NO in control volumes.
Precursor to NO2
As seen in the
Biological functions
NO is a
Nitric oxide, an
The endothelium (inner lining) of blood vessels uses nitric oxide to signal the surrounding smooth muscle to relax, resulting in vasodilation and increasing blood flow.[26] Sildenafil (Viagra) is a drug that uses the nitric oxide pathway. Sildenafil does not produce nitric oxide, but enhances the signals that are downstream of the nitric oxide pathway by protecting cyclic guanosine monophosphate (cGMP) from degradation by cGMP-specific phosphodiesterase type 5 (PDE5) in the corpus cavernosum, allowing for the signal to be enhanced, and thus vasodilation.[25] Another endogenous gaseous transmitter, hydrogen sulfide (H2S) works with NO to induce vasodilation and angiogenesis in a cooperative manner.[28][29]
Nasal breathing produces nitric oxide within the body, while oral breathing does not.[30][31]
Occupational safety and health
In the U.S., the
Explosion hazard
Liquid nitrogen oxide is very sensitive to detonation even in the absence of fuel, and can be initiated as readily as nitroglycerin. Detonation of the endothermic liquid oxide close to its b.p. (-152°C) generated a 100 kbar pulse and fragmented the test equipment. It is the simplest molecule that is capable of detonation in all three phases. The liquid oxide is sensitive and may explode during distillation, and this has been the cause of industrial accidents.[33] Gaseous nitric oxide detonates at about 2300 m/s, but as a solid it can reach a detonation velocity of 6100 m/s.[34]
References
Notes
- ^ a b Nomenclature of Inorganic Chemistry, IUPAC Recommendations (PDF). International Union of Pure and Applied Chemistry. 2005. p. 69.
- ^ "Nitric Oxide (CHEBI:16480)". Chemical Entities of Biological Interest (ChEBI). UK: European Bioinformatics Institute.
- ^ a b c "Nitrogen monoxide - Registration Dossier - ECHA". Retrieved 2020-11-02.
- ^ a b c d "Safety Data Sheet - Nitric Oxide, compressed - Registration Dossier" (PDF). Retrieved 2020-11-02.
- ^ a b "Nitric oxide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ ISBN 978-0-08-037941-8.
- PMID 10390607.
- PMID 1361684.
- ^ "The Nobel Prize in Physiology or Medicine 1998". NobelPrize.org. Retrieved 2022-06-17.
- .
- doi:10.1139/p75-254.
- PMID 19437472.
- Traube purine synthesis.
Further reading
- Butler A. and Nicholson R.; "Life, death and NO." Cambridge 2003. ISBN 978-0-85404-686-7.
- van Faassen, E. E.; Vanin, A. F. (eds); "Radicals for life: The various forms of Nitric Oxide." Elsevier, Amsterdam 2007. ISBN 978-0-444-52236-8.
- Ignarro, L. J. (ed.); "Nitric oxide:biology and pathobiology." Academic Press, San Diego 2000. ISBN 0-12-370420-0.
External links
- International Chemical Safety Card 1311
- "Nitric oxide and its role in health and diabetes". 21 October 2015.
- Microscale Gas Chemistry: Experiments with Nitrogen Oxides
- Your Brain Boots Up Like a Computer – new insights about the biological role of nitric oxide.
- Assessing The Potential of Nitric Oxide in the Diabetic Foot
- New Discoveries About Nitric Oxide Can Provide Drugs For Schizophrenia
- Nitric Oxide at the Chemical Database
- "Immediately Dangerous to Life or Health Concentrations (IDLH): Nitric oxide". National Institute for Occupational Safety and Health. 2 November 2018.