Nitrogen
 Liquid nitrogen (N2 at below −196 °C) | ||||||||||||||||||||||||||
| Nitrogen | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Allotropes | see § Allotropes | |||||||||||||||||||||||||
| Appearance | colorless gas, liquid or solid | |||||||||||||||||||||||||
| Standard atomic weight Ar°(N) | ||||||||||||||||||||||||||
| Nitrogen in the periodic table | ||||||||||||||||||||||||||
| ||||||||||||||||||||||||||
kJ/mol | ||||||||||||||||||||||||||
| Heat of vaporisation | (N2) 5.57 kJ/mol | |||||||||||||||||||||||||
| Molar heat capacity | (N2) 29.124 J/(mol·K) | |||||||||||||||||||||||||
Vapour pressure
| ||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||
Discovery | Daniel Rutherford (1772) | |||||||||||||||||||||||||
| Named by | Jean-Antoine Chaptal (1790) | |||||||||||||||||||||||||
| Isotopes of nitrogen | ||||||||||||||||||||||||||
| ||||||||||||||||||||||||||
Nitrogen is a
It was first discovered and isolated by Scottish physician
Elemental nitrogen is usually produced from air by
Nitrogen occurs in all organisms, primarily in
History

Nitrogen compounds have a very long history,
The discovery of nitrogen is attributed to the Scottish physician Daniel Rutherford in 1772, who called it noxious air.[8][9] Though he did not recognise it as an entirely different chemical substance, he clearly distinguished it from Joseph Black's "fixed air", or carbon dioxide.[10] The fact that there was a component of air that does not support combustion was clear to Rutherford, although he was not aware that it was an element. Nitrogen was also studied at about the same time by Carl Wilhelm Scheele,[11] Henry Cavendish,[12] and Joseph Priestley,[13] who referred to it as burnt air or phlogisticated air. French chemist Antoine Lavoisier referred to nitrogen gas as "mephitic air" or azote, from the Greek word άζωτικός (azotikos), "no life", due to it being asphyxiant.[14][15] In an atmosphere of pure nitrogen, animals died and flames were extinguished. Though Lavoisier's name was not accepted in English since it was pointed out that all gases but oxygen are either asphyxiant or outright toxic, it is used in many languages (French, Italian, Portuguese, Polish, Russian, Albanian, Turkish, etc.; the German Stickstoff similarly refers to the same characteristic, viz. ersticken "to choke or suffocate") and still remains in English in the common names of many nitrogen compounds, such as hydrazine and compounds of the azide ion. Finally, it led to the name "pnictogens" for the group headed by nitrogen, from the Greek πνίγειν "to choke".[7]
The English word nitrogen (1794) entered the language from the French nitrogène, coined in 1790 by French chemist
The earliest military, industrial, and agricultural applications of nitrogen compounds used saltpeter (
For a long time, sources of nitrogen compounds were limited. Natural sources originated either from biology or deposits of nitrates produced by atmospheric reactions.
Properties
Atomic

A nitrogen atom has seven electrons. In the ground state, they are arranged in the electron configuration 1s2
2s2
2p1
x2p1
y2p1
z. It, therefore, has five valence electrons in the 2s and 2p orbitals, three of which (the p-electrons) are unpaired. It has one of the highest electronegativities among the elements (3.04 on the Pauling scale), exceeded only by chlorine (3.16), oxygen (3.44), and fluorine (3.98). (The light noble gases, helium, neon, and argon, would presumably also be more electronegative, and in fact are on the Allen scale.)[23] Following periodic trends, its single-bond covalent radius of 71 pm is smaller than those of boron (84 pm) and carbon (76 pm), while it is larger than those of oxygen (66 pm) and fluorine (57 pm). The nitride anion, N3−, is much larger at 146 pm, similar to that of the oxide (O2−: 140 pm) and fluoride (F−: 133 pm) anions.[23] The first three ionisation energies of nitrogen are 1.402, 2.856, and 4.577 MJ·mol−1, and the sum of the fourth and fifth is 16.920 MJ·mol−1. Due to these very high figures, nitrogen has no simple cationic chemistry.[24]
The lack of radial nodes in the 2p subshell is directly responsible for many of the anomalous properties of the first row of the
Nitrogen may be usefully compared to its horizontal neighbours' carbon and oxygen as well as its vertical neighbours in the pnictogen column, phosphorus, arsenic, antimony, and bismuth. Although each period 2 element from lithium to oxygen shows some similarities to the period 3 element in the next group (from magnesium to chlorine; these are known as diagonal relationships), their degree drops off abruptly past the boron–silicon pair. The similarities of nitrogen to sulfur are mostly limited to sulfur nitride ring compounds when both elements are the only ones present.[26]
Nitrogen does not share the proclivity of carbon for catenation. Like carbon, nitrogen tends to form ionic or metallic compounds with metals. Nitrogen forms an extensive series of nitrides with carbon, including those with chain-, graphitic-, and fullerenic-like structures.[27]
It resembles oxygen with its high electronegativity and concomitant capability for hydrogen bonding and the ability to form coordination complexes by donating its lone pairs of electrons. There are some parallels between the chemistry of ammonia NH3 and water H2O. For example, the capacity of both compounds to be protonated to give NH4+ and H3O+ or deprotonated to give NH2− and OH−, with all of these able to be isolated in solid compounds.[28]
Nitrogen shares with both its horizontal neighbours a preference for forming multiple bonds, typically with carbon, oxygen, or other nitrogen atoms, through pπ–pπ interactions.
Table of thermal and physical properties of nitrogen (N2) at atmospheric pressure:[30][31]
| Temperature (K) | Density (kg m−3) | Specific heat (kJ kg−1 °C−1) | Dynamic viscosity (kg m−1 s−1) | Kinematic viscosity (m2 s−1) | Thermal conductivity (W m−1 °C−1) | Thermal diffusivity (m2 s−1) | Prandtl number |
| 100 | 3.4388 | 1.07 | 6.88×10−6 | 2.00×10−6 | 0.00958 | 2.60×10−6 | 0.768 |
| 150 | 2.2594 | 1.05 | 1.01×10−5 | 4.45×10−6 | 0.0139 | 5.86×10−6 | 0.759 |
| 200 | 1.7108 | 1.0429 | 1.29×10−5 | 7.57×10−6 | 0.01824 | 1.02×10−5 | 0.747 |
| 300 | 1.1421 | 1.0408 | 1.78×10−5 | 1.56×10−5 | 0.0262 | 2.20×10−5 | 0.713 |
| 400 | 0.8538 | 1.0459 | 2.20×10−5 | 2.57×10−5 | 0.03335 | 3.73×10−5 | 0.691 |
| 500 | 0.6824 | 1.0555 | 2.57×10−5 | 3.77×10−5 | 0.03984 | 5.53×10−5 | 0.684 |
| 600 | 0.5687 | 1.0756 | 2.91×10−5 | 5.12×10−5 | 0.0458 | 7.49×10−5 | 0.686 |
| 700 | 0.4934 | 1.0969 | 3.21×10−5 | 6.67×10−5 | 0.05123 | 9.47×10−5 | 0.691 |
| 800 | 0.4277 | 1.1225 | 3.48×10−5 | 8.15×10−5 | 0.05609 | 1.17×10−4 | 0.7 |
| 900 | 0.3796 | 1.1464 | 3.75×10−5 | 9.11×10−5 | 0.0607 | 1.39×10−4 | 0.711 |
| 1000 | 0.3412 | 1.1677 | 4.00×10−5 | 1.19×10−4 | 0.06475 | 1.63×10−4 | 0.724 |
| 1100 | 0.3108 | 1.1857 | 4.23×10−5 | 1.36×10−4 | 0.0685 | 1.86×10−4 | 0.736 |
| 1200 | 0.2851 | 1.2037 | 4.45×10−5 | 1.56×10−4 | 0.07184 | 2.09×10−4 | 0.748 |
| 1300 | 0.2591 | 1.219 | 4.66×10−5 | 1.80×10−4 | 0.081 | 2.56×10−4 | 0.701 |
Isotopes

Nitrogen has two stable isotopes: 14N and 15N. The first is much more common, making up 99.634% of natural nitrogen, and the second (which is slightly heavier) makes up the remaining 0.366%. This leads to an atomic weight of around 14.007 u.[23] Both of these stable isotopes are produced in the CNO cycle in stars, but 14N is more common as its proton capture is the rate-limiting step. 14N is one of the five stable odd–odd nuclides (a nuclide having an odd number of protons and neutrons); the other four are 2H, 6Li, 10B, and 180mTa.[32]
The relative abundance of 14N and 15N is practically constant in the atmosphere but can vary elsewhere, due to natural isotopic fractionation from biological redox reactions and the evaporation of natural ammonia or nitric acid.[33] Biologically mediated reactions (e.g., assimilation, nitrification, and denitrification) strongly control nitrogen dynamics in the soil. These reactions typically result in 15N enrichment of the substrate and depletion of the product.[34]
The heavy isotope 15N was first discovered by S. M. Naudé in 1929, and soon after heavy isotopes of the neighbouring elements
Of the thirteen other isotopes produced synthetically, ranging from 9N to 23N, 13N has a half-life of ten minutes and the remaining isotopes have half-lives less than eight seconds.[39][40] Given the half-life difference, 13N is the most important nitrogen radioisotope, being relatively long-lived enough to use in positron emission tomography (PET), although its half-life is still short and thus it must be produced at the venue of the PET, for example in a cyclotron via proton bombardment of 16O producing 13N and an alpha particle.[41]
The
Allotropes

Atomic nitrogen, also known as active nitrogen, is highly reactive, being a triradical with three unpaired electrons. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited N2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as CO and O or OH and H. Atomic nitrogen is prepared by passing an electric discharge through nitrogen gas at 0.1–2 mmHg, which produces atomic nitrogen along with a peach-yellow emission that fades slowly as an afterglow for several minutes even after the discharge terminates.[26]
Given the great reactivity of atomic nitrogen, elemental nitrogen usually occurs as molecular N2, dinitrogen. This molecule is a colourless, odourless, and tasteless
Other nitrogen

At
Chemistry and compounds
Dinitrogen complexes

The first example of a
Dinitrogen is able to coordinate to metals in five different ways. The more well-characterised ways are the end-on M←N≡N (
Today, dinitrogen complexes are known for almost all the transition metals, accounting for several hundred compounds. They are normally prepared by three methods:[26]
- Replacing labile ligands such as H2O, H−, or CO directly by nitrogen: these are often reversible reactions that proceed at mild conditions.
- Reducing metal complexes in the presence of a suitable co-ligand in excess under nitrogen gas. A common choice includes replacing chloride ligands with dimethylphenylphosphine (PMe2Ph) to make up for the smaller number of nitrogen ligands attached to the original chlorine ligands.
- Converting a ligand with N–N bonds, such as hydrazine or azide, directly into a dinitrogen ligand.
Occasionally the N≡N bond may be formed directly within a metal complex, for example by directly reacting coordinated ammonia (NH3) with nitrous acid (HNO2), but this is not generally applicable. Most dinitrogen complexes have colours within the range white-yellow-orange-red-brown; a few exceptions are known, such as the blue [{Ti(η5-C5H5)2}2-(N2)].[26]
Nitrides, azides, and nitrido complexes
Nitrogen bonds to almost all the elements in the periodic table except the first two
- 3 Ca + N2 → Ca3N2
- 3 Mg + 2 NH3 → Mg3N2 + 3 H2 (at 900 °C)
- 3 Zn(NH2)2 → Zn3N2 + 4 NH3
Many variants on these processes are possible. The most ionic of these nitrides are those of the alkali metals and alkaline earth metals, Li3N (Na, K, Rb, and Cs do not form stable nitrides for steric reasons) and M3N2 (M = Be, Mg, Ca, Sr, Ba). These can formally be thought of as salts of the N3− anion, although charge separation is not actually complete even for these highly electropositive elements. However, the alkali metal azides NaN3 and KN3, featuring the linear N−
3 anion, are well-known, as are Sr(N3)2 and Ba(N3)2. Azides of the B-subgroup metals (those in groups 11 through 16) are much less ionic, have more complicated structures, and detonate readily when shocked.[54]
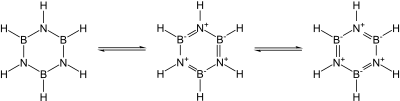
Many covalent binary nitrides are known. Examples include cyanogen ((CN)2), triphosphorus pentanitride (P3N5), disulfur dinitride (S2N2), and tetrasulfur tetranitride (S4N4). The essentially covalent silicon nitride (Si3N4) and germanium nitride (Ge3N4) are also known: silicon nitride, in particular, would make a promising ceramic if not for the difficulty of working with and sintering it. In particular, the group 13 nitrides, most of which are promising semiconductors, are isoelectronic with graphite, diamond, and silicon carbide and have similar structures: their bonding changes from covalent to partially ionic to metallic as the group is descended. In particular, since the B–N unit is isoelectronic to C–C, and carbon is essentially intermediate in size between boron and nitrogen, much of organic chemistry finds an echo in boron–nitrogen chemistry, such as in borazine ("inorganic benzene"). Nevertheless, the analogy is not exact due to the ease of nucleophilic attack at boron due to its deficiency in electrons, which is not possible in a wholly carbon-containing ring.[54]
The largest category of nitrides are the interstitial nitrides of formulae MN, M2N, and M4N (although variable composition is perfectly possible), where the small nitrogen atoms are positioned in the gaps in a metallic cubic or
The nitride anion (N3−) is the strongest π donor known among ligands (the second-strongest is O2−). Nitrido complexes are generally made by the thermal decomposition of azides or by deprotonating ammonia, and they usually involve a terminal {≡N}3− group. The linear azide anion (N−
3), being isoelectronic with nitrous oxide, carbon dioxide, and cyanate, forms many coordination complexes. Further catenation is rare, although N4−
4 (isoelectronic with carbonate and nitrate) is known.[54]
Hydrides

Industrially,
4. It can also act as an extremely weak acid, losing a proton to produce the amide anion, NH−
2. It thus undergoes self-dissociation, similar to water, to produce ammonium and amide. Ammonia burns in air or oxygen, though not readily, to produce nitrogen gas; it burns in fluorine with a greenish-yellow flame to give nitrogen trifluoride. Reactions with the other nonmetals are very complex and tend to lead to a mixture of products. Ammonia reacts on heating with metals to give nitrides.[56]
Many other binary nitrogen hydrides are known, but the most important are
Hydrazine is generally made by reaction of ammonia with alkaline sodium hypochlorite in the presence of gelatin or glue:[57]
- NH3 + OCl− → NH2Cl + OH−
- NH2Cl + NH3 → N
2H+
5 + Cl− (slow) - N
2H+
5 + OH− → N2H4 + H2O (fast)
(The attacks by hydroxide and ammonia may be reversed, thus passing through the intermediate NHCl− instead.) The reason for adding gelatin is that it removes metal ions such as Cu2+ that catalyses the destruction of hydrazine by reaction with monochloramine (NH2Cl) to produce ammonium chloride and nitrogen.[57]
Halides and oxohalides

All four simple nitrogen trihalides are known. A few mixed halides and hydrohalides are known, but are mostly unstable; examples include NClF2, NCl2F, NBrF2, NF2H, NFH2, NCl2H, and NClH2.[59]
Nitrogen trifluoride (NF3, first prepared in 1928) is a colourless and odourless gas that is thermodynamically stable, and most readily produced by the electrolysis of molten ammonium fluoride dissolved in anhydrous hydrogen fluoride. Like carbon tetrafluoride, it is not at all reactive and is stable in water or dilute aqueous acids or alkalis. Only when heated does it act as a fluorinating agent, and it reacts with copper, arsenic, antimony, and bismuth on contact at high temperatures to give tetrafluorohydrazine (N2F4). The cations NF+
4 and N
2F+
3 are also known (the latter from reacting tetrafluorohydrazine with strong fluoride-acceptors such as arsenic pentafluoride), as is ONF3, which has aroused interest due to the short N–O distance implying partial double bonding and the highly polar and long N–F bond. Tetrafluorohydrazine, unlike hydrazine itself, can dissociate at room temperature and above to give the radical NF2•. Fluorine azide (FN3) is very explosive and thermally unstable. Dinitrogen difluoride (N2F2) exists as thermally interconvertible cis and trans isomers, and was first found as a product of the thermal decomposition of FN3.[59]
Nitrogen trichloride (NCl3) is a dense, volatile, and explosive liquid whose physical properties are similar to those of carbon tetrachloride, although one difference is that NCl3 is easily hydrolysed by water while CCl4 is not. It was first synthesised in 1811 by Pierre Louis Dulong, who lost three fingers and an eye to its explosive tendencies. As a dilute gas it is less dangerous and is thus used industrially to bleach and sterilise flour. Nitrogen tribromide (NBr3), first prepared in 1975, is a deep red, temperature-sensitive, volatile solid that is explosive even at −100 °C. Nitrogen triiodide (NI3) is still more unstable and was only prepared in 1990. Its adduct with ammonia, which was known earlier, is very shock-sensitive: it can be set off by the touch of a feather, shifting air currents, or even alpha particles.[59][60] For this reason, small amounts of nitrogen triiodide are sometimes synthesised as a demonstration to high school chemistry students or as an act of "chemical magic".[61] Chlorine azide (ClN3) and bromine azide (BrN3) are extremely sensitive and explosive.[62][63]
Two series of nitrogen oxohalides are known: the nitrosyl halides (XNO) and the nitryl halides (XNO2). The first is very reactive gases that can be made by directly halogenating nitrous oxide. Nitrosyl fluoride (NOF) is colourless and a vigorous fluorinating agent. Nitrosyl chloride (NOCl) behaves in much the same way and has often been used as an ionising solvent. Nitrosyl bromide (NOBr) is red. The reactions of the nitryl halides are mostly similar: nitryl fluoride (FNO2) and nitryl chloride (ClNO2) are likewise reactive gases and vigorous halogenating agents.[59]
Oxides

2 converts to colourless dinitrogen tetroxide (N
2O
4) at low temperatures, and reverts to NO
2 at higher temperatures.
Nitrogen forms nine molecular oxides, some of which were the first gases to be identified: N2O (
Nitrous oxide (N2O), better known as laughing gas, is made by thermal decomposition of molten
Blue dinitrogen trioxide (N2O3) is only available as a solid because it rapidly dissociates above its melting point to give nitric oxide, nitrogen dioxide (NO2), and dinitrogen tetroxide (N2O4). The latter two compounds are somewhat difficult to study individually because of the equilibrium between them, although sometimes dinitrogen tetroxide can react by heterolytic fission to
The thermally unstable and very reactive dinitrogen pentoxide (N2O5) is the anhydride of
- N2O5 ⇌ NO2 + NO3 → NO2 + O2 + NO
- N2O5 + NO ⇌ 3 NO2
Oxoacids, oxoanions, and oxoacid salts
Many nitrogen
2O2−
2 anion) are stable to reducing agents and more commonly act as reducing agents themselves. They are an intermediate step in the oxidation of ammonia to nitrite, which occurs in the nitrogen cycle. Hyponitrite can act as a bridging or chelating bidentate ligand.[68]
Nitrous acid (HNO2) is not known as a pure compound, but is a common component in gaseous equilibria and is an important aqueous reagent: its aqueous solutions may be made from acidifying cool aqueous nitrite (NO−
2, bent) solutions, although already at room temperature disproportionation to nitrate and nitric oxide is significant. It is a weak acid with pKa 3.35 at 18 °C. They may be titrimetrically analysed by their oxidation to nitrate by permanganate. They are readily reduced to nitrous oxide and nitric oxide by sulfur dioxide, to hyponitrous acid with tin(II), and to ammonia with hydrogen sulfide. Salts of hydrazinium N
2H+
5 react with nitrous acid to produce azides which further react to give nitrous oxide and nitrogen. Sodium nitrite is mildly toxic in concentrations above 100 mg/kg, but small amounts are often used to cure meat and as a preservative to avoid bacterial spoilage. It is also used to synthesise hydroxylamine and to diazotise primary aromatic amines as follows:[68]
- ArNH2 + HNO2 → [ArNN]Cl + 2 H2O
Nitrite is also a common ligand that can coordinate in five ways. The most common are nitro (bonded from the nitrogen) and nitrito (bonded from an oxygen). Nitro-nitrito isomerism is common, where the nitrito form is usually less stable.[68]

Nitric acid (HNO3) is by far the most important and the most stable of the nitrogen oxoacids. It is one of the three most used acids (the other two being sulfuric acid and hydrochloric acid) and was first discovered by alchemists in the 13th century. It is made by the catalytic oxidation of ammonia to nitric oxide, which is oxidised to nitrogen dioxide, and then dissolved in water to give concentrated nitric acid. In the United States of America, over seven million tonnes of nitric acid are produced every year, most of which is used for nitrate production for fertilisers and explosives, among other uses. Anhydrous nitric acid may be made by distilling concentrated nitric acid with phosphorus pentoxide at low pressure in glass apparatus in the dark. It can only be made in the solid state, because upon melting it spontaneously decomposes to nitrogen dioxide, and liquid nitric acid undergoes self-ionisation to a larger extent than any other covalent liquid as follows:[68]
- 2 HNO3 ⇌ H
2NO+
3 + NO−
3 ⇌ H2O + [NO2]+ + [NO3]−
Two hydrates, HNO3·H2O and HNO3·3H2O, are known that can be crystallised. It is a strong acid and concentrated solutions are strong oxidising agents, though
- HNO3 + 2 H2SO4 ⇌ NO+
2 + H3O+ + 2 HSO−
4
The thermal stabilities of nitrates (involving the trigonal planar NO−
3 anion) depends on the basicity of the metal, and so do the products of decomposition (thermolysis), which can vary between the nitrite (for example, sodium), the oxide (potassium and lead), or even the metal itself (silver) depending on their relative stabilities. Nitrate is also a common ligand with many modes of coordination.[68]
Finally, although orthonitric acid (H3NO4), which would be analogous to
4 is known in its sodium and potassium salts:[68]
These white crystalline salts are very sensitive to water vapour and carbon dioxide in the air:[68]
- Na3NO4 + H2O + CO2 → NaNO3 + NaOH + NaHCO3
Despite its limited chemistry, the orthonitrate anion is interesting from a structural point of view due to its regular tetrahedral shape and the short N–O bond lengths, implying significant polar character to the bonding.[68]
Organic nitrogen compounds
Nitrogen is one of the most important elements in
Occurrence
Nitrogen is the most common pure element in the earth, making up 78.1% of the volume of the
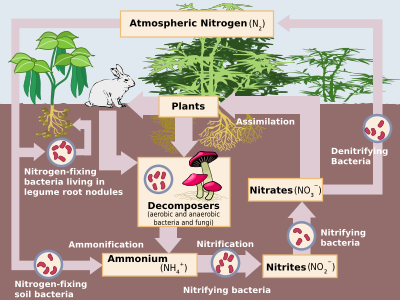
Nitrogen compounds constantly interchange between the atmosphere and living organisms. Nitrogen must first be processed, or "fixed", into a plant-usable form, usually ammonia. Some nitrogen fixation is done by lightning strikes producing the nitrogen oxides, but most is done by diazotrophic bacteria through enzymes known as nitrogenases (although today industrial nitrogen fixation to ammonia is also significant). When the ammonia is taken up by plants, it is used to synthesise proteins. These plants are then digested by animals who use the nitrogen compounds to synthesise their proteins and excrete nitrogen-bearing waste. Finally, these organisms die and decompose, undergoing bacterial and environmental oxidation and denitrification, returning free dinitrogen to the atmosphere. Industrial nitrogen fixation by the Haber process is mostly used as fertiliser, although excess nitrogen–bearing waste, when leached, leads to eutrophication of freshwater and the creation of marine dead zones, as nitrogen-driven bacterial growth depletes water oxygen to the point that all higher organisms die. Furthermore, nitrous oxide, which is produced during denitrification, attacks the atmospheric ozone layer.[72]
Many saltwater fish manufacture large amounts of
Nitric oxide's rapid reaction with water in animals results in the production of its metabolite
Production
Nitrogen gas is an industrial gas produced by the fractional distillation of liquid air, or by mechanical means using gaseous air (pressurised reverse osmosis membrane or pressure swing adsorption). Nitrogen gas generators using membranes or pressure swing adsorption (PSA) are typically more cost and energy efficient than bulk-delivered nitrogen.[76] Commercial nitrogen is often a byproduct of air-processing for industrial concentration of oxygen for steelmaking and other purposes. When supplied compressed in cylinders it is often called OFN (oxygen-free nitrogen).[77] Commercial-grade nitrogen already contains at most 20 ppm oxygen, and specially purified grades containing at most 2 ppm oxygen and 10 ppm argon are also available.[78]
In a chemical laboratory, it is prepared by treating an aqueous solution of ammonium chloride with sodium nitrite.[79]
- NH4Cl + NaNO2 → N2 + NaCl + 2 H2O
Small amounts of the impurities NO and HNO3 are also formed in this reaction. The impurities can be removed by passing the gas through aqueous sulfuric acid containing potassium dichromate.[79]
It can also be obtained by the thermal decomposition of ammonium dichromate. [80]
- 3(NH4)2Cr2O7 → 2N2 + 9H2O + 3Cr2O3 + 2NH3 + 32O2
Very pure nitrogen can be prepared by the thermal decomposition of barium azide or sodium azide.[81]
- 2 NaN3 → 2 Na + 3 N2
Applications
The applications of nitrogen compounds are naturally extremely widely varied due to the huge size of this class: hence, only applications of pure nitrogen itself will be considered here. Two-thirds (2/3) of nitrogen produced by industry is sold as gas and the remaining one-third (1/3) as a liquid.
Gas
The gas is mostly used as a low reactivity safe atmosphere wherever the oxygen in the air would pose a fire, explosion, or oxidising hazard. Some examples include:[78]
- As a modified atmosphere, pure or mixed with carbon dioxide, to nitrogenate and preserve the freshness of packaged or bulk foods (by delaying rancidity and other forms of oxidative damage). Pure nitrogen as food additive is labeled in the European Union with the E number E941.[82]
- In incandescent light bulbs as an inexpensive alternative to argon.[83]
- In fire suppression systems for Information technology (IT) equipment.[78]
- In the manufacture of stainless steel.[84]
- In the case-hardening of steel by nitriding.[85]
- In some aircraft fuel systems to reduce fire hazard (see inerting system).
- To inflate race car and aircraft tires,[86] reducing the problems of inconsistent expansion and contraction caused by moisture and oxygen in natural air.[78]
Nitrogen is commonly used during sample preparation in
Nitrogen can be used as a replacement, or in combination with,
Equipment
Some construction equipment uses pressurized nitrogen gas to help hydraulic system to provide extra power to devices such as hydraulic hammer. Nitrogen gas, formed from the decomposition of sodium azide, is used for the inflation of airbags.[92]
Execution
As nitrogen is an asphyxiant gas in itself, some jurisdictions have considered asphyxiation by inhalation of pure nitrogen as a means of capital punishment (as a substitute for lethal injection).[93][94][95] In January 2024, Kenneth Eugene Smith became the first person executed by nitrogen asphyxiation.[96]
Liquid
Liquid nitrogen is a

Like
Safety
Gas
Although nitrogen is non-toxic, when released into an enclosed space it can displace oxygen, and therefore presents an
When inhaled at high partial pressures (more than about 4 bar, encountered at depths below about 30 m in scuba diving), nitrogen is an anesthetic agent, causing nitrogen narcosis, a temporary state of mental impairment similar to nitrous oxide intoxication.[101][102]
Nitrogen dissolves in the blood and body fats. Rapid decompression (as when divers ascend too quickly or astronauts decompress too quickly from cabin pressure to spacesuit pressure) can lead to a potentially fatal condition called decompression sickness (formerly known as caisson sickness or the bends), when nitrogen bubbles form in the bloodstream, nerves, joints, and other sensitive or vital areas.[103][104] Bubbles from other "inert" gases (gases other than carbon dioxide and oxygen) cause the same effects, so replacement of nitrogen in breathing gases may prevent nitrogen narcosis, but does not prevent decompression sickness.[105]
Liquid
As a
Because the liquid-to-gas expansion ratio of nitrogen is 1:694 at 20 °C, a tremendous amount of force can be generated if liquid nitrogen is rapidly vaporised in an enclosed space. In an incident on January 12, 2006, at Texas A&M University, the pressure-relief devices of a tank of liquid nitrogen were malfunctioning and later sealed. As a result of the subsequent pressure buildup, the tank failed catastrophically. The force of the explosion was sufficient to propel the tank through the ceiling immediately above it, shatter a reinforced concrete beam immediately below it, and blow the walls of the laboratory 0.1–0.2 m off their foundations.[108]
Liquid nitrogen readily evaporates to form gaseous nitrogen, and hence the precautions associated with gaseous nitrogen also apply to liquid nitrogen.[109][110][111] For example, oxygen sensors are sometimes used as a safety precaution when working with liquid nitrogen to alert workers of gas spills into a confined space.[112]
Vessels containing liquid nitrogen can condense oxygen from air. The liquid in such a vessel becomes increasingly enriched in oxygen (boiling point −183 °C, higher than that of nitrogen) as the nitrogen evaporates, and can cause violent oxidation of organic material.[113]
Oxygen deficiency monitors
Oxygen deficiency monitors are used to measure levels of oxygen in confined spaces and any place where nitrogen gas or liquid are stored or used. In the event of a nitrogen leak, and a decrease in oxygen to a pre-set alarm level, an oxygen deficiency monitor can be programmed to set off audible and visual alarms, thereby providing notification of the possible impending danger. Most commonly the oxygen range to alert personnel is when oxygen levels get below 19.5%. OSHA specifies that a hazardous atmosphere may include one where the oxygen concentration is below 19.5% or above 23.5%.[114] Oxygen deficiency monitors can either be fixed, mounted to the wall and hard-wired into the building's power supply or simply plugged into a power outlet, or a portable hand-held or wearable monitor.
See also
References
- ^ "Standard Atomic Weights: Nitrogen". CIAAW. 2009.
- ISSN 1365-3075.
- ^ a b c Lide, David R. (1990–1991). CRC Handbook of Physics and Chemistry (71st ed.). Boca Raton, Ann Arbor, Boston: CRC Press, inc. pp. 4-22 (one page).
- ^ "Gases - Density". The Engineering Toolbox. Retrieved 27 January 2019.
- ^ Tetrazoles contain a pair of double-bonded nitrogen atoms with oxidation state 0 in the ring. A Synthesis of the parent 1H-tetrazole, CH2N4 (two atoms N(0)) is given in Ronald A. Henry and William G. Finnegan, "An Improved Procedure for the Deamination of 5-Aminotetrazole", _J. Am. Chem. Soc._ (1954), 76, 1, 290–291, https://doi.org/10.1021/ja01630a086.
- ^ Common Bond Energies (D) and Bond Lengths (r) Archived 2010-05-15 at the Wayback Machine. wiredchemist.com
- ^ a b c Greenwood and Earnshaw, pp. 406–07
- .
- .
- ^ Ihde, Aaron J. (1964). The Development of Modern Chemistry. New York: Harper & Row.
- ^ Carl Wilhelm Scheele, Chemische Abhandlung von der Luft und dem Feuer [Chemical treatise on air and fire] (Upsala, Sweden: Magnus Swederus, 1777; and Leipzig, (Germany): Siegfried Lebrecht Crusius, 1777). In the section titled "Die Luft muß aus elastischen Flüßigkeiten von zweyerley Art, zusammengesetzet seyn." (The air must be composed of elastic fluids of two sorts), pp. 6–14, Scheele presents the results of eight experiments in which air was reacted with various substances. He concluded (p. 13): "So viel sehe ich aus angeführten Versuchen, daß die Luft aus 2 von einander unterschiedenen Flußigkeiten bestehe, von welchen die eine die Eigenschaft das Phlogiston anzuziehen gar nicht äussere, die andere aber zur solchen Attraction eigentlich aufgeleget ist und welche zwischen dem 3:ten und 4:ten Theil von der ganzen Luftmasse aus machet." (So I see [this] much from the experiments [that were] conducted: that the air consists of two fluids [that] differ from one another, of which the one doesn't express at all the property of attracting phlogiston; the other, however, is capable of such attraction and which makes up between 1/3 and 1/4 part of the entire mass of the air.)
- S2CID 186210131. ; see: "VII. Of air infected with the fumes of burning charcoal." pp. 225–28. Archived 2016-09-03 at the Wayback Machine
- ^ Lavoisier, Antoine with Robert Kerr, trans., Elements of Chemistry, 4th ed. (Edinburgh, Scotland: William Creech, 1799), pp. 85–86. [p. 85:] Archived 2020-08-06 at the Wayback Machine "In reflecting upon the circumstances of this experiment, we readily perceive; that the mercury, during its calcination [i.e., roasting in air], absorbs the salubrious and respirable part of the air, or, to speak more strictly, the base of this respirable part; that the remaining air is a species of mephitis [i.e., a poisonous gas emitted from the earth], incapable of supporting combustion or respiration; ... " [p. 86:] Archived 2020-08-06 at the Wayback Machine "I shall afterwards shew, that at least in our climate, the atmospheric air is composed of respirable and mephitic airs, in the proportion of 27 and 73; ... "
- ^ Lavoisier, Antoine with Robert Kerr, trans., Elements of Chemistry, 4th ed. (Edinburgh, Scotland: William Creech, 1799), p. 101: "The chemical properties of the noxious portion of the atmospheric air being hitherto but little known, we have been satisfied to derive the name of its base from its known quality of killing such animals as are forced to breathe it, giving it the name of azot, from the Greek privitive particle α and ξωη, vita; hence the name of the noxious part of atmospheric air is azotic gas."
- ^ Chaptal, J. A. and Nicholson, William trans. (1800) Elements of Chemistry, 3rd ed. London, England: C.C. and J. Robinson, vol. 1. pp. xxxv–xxxvi: "In order to correct the Nomenclature on this head [i.e., in this regard], nothing more is necessary than to substitute to [i.e., for] this word a denomination which is derived from the general system made use of; and I have presumed to propose that of Nitrogene Gas. In the first place, it is deduced from the characteristic and exclusive property of this gas, which forms the radical of the nitric acid. By this means we shall preserve to the combinations [i.e., compounds] of this substance the received [i.e., prevailing] denominations, such as those of the Nitric Acid, Nitrates, Nitrites, &c."
- ^ nitrogen Archived 2017-07-02 at the Wayback Machine. Etymonline.com. Retrieved 2011-10-26.
- ^ Strutt, R. J. (1911) "Bakerian Lecture. A chemically active modification of nitrogen, produced by the electric discharge", Archived 2016-12-20 at the Wayback Machine Proceedings of the Royal Society A, 85 (577): 219–29.
- ^ Lord Rayleigh's Active Nitrogen Archived 2012-11-01 at the Wayback Machine. Lateralscience.co.uk. Retrieved 2011-10-26.
- S2CID 94880859.
- ^ GB 190200698, Ostwald, Wilhelm, "Improvements in the Manufacture of Nitric Acid and Nitrogen Oxides", published 1902-03-20
- ^ GB 190208300, Ostwald, Wilhelm, "Improvements in and relating to the Manufacture of Nitric Acid and Oxides of Nitrogen", published 1903-02-26
- ^ a b c d e Greenwood and Earnshaw, pp. 411–12
- ^ Greenwood and Earnshaw, p. 550
- S2CID 12677737.
- ^ a b c d e f g h i j Greenwood and Earnshaw, pp. 412–16
- PMID 28594419.
- ISBN 978-0-12-804697-5.
- .
- OCLC 46959719.
- )
- PMID 17835673.
- ^ CIAAW (2003). "Atomic Weight of Nitrogen". ciaaw.org. CIAAW. Archived from the original on 14 October 2016. Retrieved 13 October 2016.
- ISBN 978-0-08-052528-0. Archivedfrom the original on 5 February 2016. Retrieved 20 December 2015.
- ^ Greenwood and Earnshaw, p. 408
- ^ "Evaluated Nuclear Data File (ENDF) Retrieval & Plotting". National Nuclear Data Center. Archived from the original on 2020-08-09. Retrieved 2016-11-23.
- ISBN 978-0-12-164491-8.
- ISBN 978-0-471-79372-4.
- science.org. Retrieved 27 September 2023.
- ^
- ISBN 978-0-205-23939-9.
- ^ ISBN 978-3-11-013242-7. Archivedfrom the original on 2016-02-05. Retrieved 2015-12-20.
- ^ "Universal Industrial Gases, Inc...Nitrogen N2 Properties, Uses, Applications - Gas and Liquid".
- ISBN 978-1-4020-6972-7.
- ^ "Polymeric nitrogen synthesized". physorg.com. 5 August 2004. Archived from the original on 2012-01-24. Retrieved 2009-06-22.
- ISBN 978-1-57912-814-2.
- .
- PMID 16427786.
- ^ "Flowing nitrogen ice glaciers seen on surface of Pluto after New Horizons flyby". ABC News. 25 July 2015. Archived from the original on 29 September 2015. Retrieved 6 October 2015.
- ISBN 978-0-12-416034-7. Archivedfrom the original on 2016-09-03. Retrieved 2016-04-30.
- ^ "Neptune: Moons: Triton". NASA. Archived from the original on October 15, 2011. Retrieved September 21, 2007.
- .
- PMID 16359167.
- ^ a b c d e Greenwood and Earnshaw, pp. 417–20
- ^ Greenwood and Earnshaw, pp. 434–38
- ^ Greenwood and Earnshaw, pp. 420–26
- ^ a b c d Greenwood and Earnshaw, pp. 426–33
- PMID 12123065.
- ^ a b c d Greenwood and Earnshaw, pp. 438–42
- S2CID 137728239.
- ISBN 978-0-486-67628-9.
- .
- (PDF) from the original on 25 August 2021. Retrieved 25 August 2021.
- ^ a b c d e f Greenwood and Earnshaw, pp. 443–58
- S2CID 32952729.
- PMID 10390607.
- PMID 15979786.
- ^ a b c d e f g h i Greenwood and Earnshaw, pp. 459–72
- ^ OCLC 642506595
- ISBN 978-1-4020-6753-2.
- ^ Depending on the average thickness which is somewhere between 10 and 30 km, the mass of the earth's crust is between 15×1018 and 45×1018 tonnes.
- ^ a b Greenwood and Earnshaw, pp. 407–09
- PMID 15186102.
- ISBN 978-0-8493-3394-1. Archivedfrom the original on 2021-10-01. Retrieved 2020-08-24.
- ISBN 978-0-8135-3387-2. Archivedfrom the original on 2021-10-01. Retrieved 2020-08-24.
- ^ Froehlich, Peter (May 2013). "A Sustainable Approach to the Supply of Nitrogen". www.parker.com. Parker Hannifin Corporation. Archived from the original on 16 March 2016. Retrieved 24 November 2016.
- .
- ^ a b c d e Greenwood and Earnshaw, pp. 409–11
- ^ .
- ISSN 0022-1902.
- PMID 15268087.
- ISBN 978-92-893-0829-8. Archivedfrom the original on 2016-02-05. Retrieved 2015-12-20.
- ISBN 978-0-85404-690-4. Archivedfrom the original on 2021-10-01. Retrieved 2020-08-24.
- ISBN 978-3-540-66411-6. Archivedfrom the original on 2021-10-01. Retrieved 2020-08-24.
- .
- ^ "Why don't they use normal air in race car tires?". Howstuffworks. 2001-03-16. Archived from the original on 2011-07-12. Retrieved 2006-07-22.
- PMID 11833649.
- ISBN 978-0-85404-588-4. Archivedfrom the original on 2020-03-21. Retrieved 2015-06-20.
- ^ "How does the widget in a beer can work?". Howstuffworks. 2000-08-16. Archived from the original on 2007-11-02. Retrieved 2008-07-30.
- ISBN 978-0-8018-9569-2. Archivedfrom the original on 5 February 2016. Retrieved 20 December 2015.
- hdl:1721.1/45820.
- S2CID 96404307.
- ^ Sanburn, Josh (2015-04-10). "The Dawn of a New Form of Capital Punishment". Time. Archived from the original on 2015-04-11. Retrieved 2015-04-11.
- ^ Sexton, Mike (18 December 2012). "Euthanasia campaigner under scrutiny". ABC. Archived from the original on 7 July 2013. Retrieved 6 May 2013.
- ^ Berman, Mark (April 17, 2015). "Oklahoma says it will now use nitrogen gas as its backup method of execution". The Washington Post. Archived from the original on June 23, 2019. Retrieved June 22, 2019.
- ^ "Kenneth Eugene Smith executed by nitrogen hypoxia in Alabama, marking a first for the death penalty". CBS News. 26 January 2024.
- S2CID 96762552.
- S2CID 221325640.
- ^ "Biology Safety – Cryogenic materials. The risks posed by them". University of Bath. Archived from the original on February 6, 2007. Retrieved 2007-01-03.
- ^ "Space Shuttle Columbia Fast Facts". CNN. September 30, 2013. Archived from the original on February 2, 2016. Retrieved January 20, 2016.
- PMID 4082343. Archived from the original on 2010-12-25. Retrieved 2008-09-21.)
{{cite journal}}: CS1 maint: unfit URL (link - PMID 2741255. Archived from the original on 2009-09-01. Retrieved 2008-09-21.)
{{cite journal}}: CS1 maint: unfit URL (link - OCLC 16986801. Archived from the original on 2011-09-05. Retrieved 2008-09-21.)
{{cite journal}}: CS1 maint: unfit URL (link - PMID 1226586. Archived from the original on 2011-07-27. Retrieved 2008-09-21.)
{{cite journal}}: CS1 maint: unfit URL (link - ^ US Navy Diving Manual, 6th revision. United States: US Naval Sea Systems Command. 2006. Archived from the original on 2008-05-02. Retrieved 2008-04-24.
- ^ Walker, Jearl. "Boiling and the Leidenfrost Effect" (PDF). Fundamentals of Physics: 1–4. Archived (PDF) from the original on 13 December 2019. Retrieved 11 October 2014.
- ^ "Liquid nitrogen cocktail leaves teen in hospital". BBC News. 2012-10-08. Archived from the original on 2017-04-12.
- ^ Mattox, Brent S. "Investigative Report on Chemistry 301A Cylinder Explosion" (PDF). Texas A&M University. Archived from the original (reprint) on 2014-04-30.
- ISSN 0260-4809. Archived from the originalon 2007-07-18..
- ^ "Confined Space Entry – Worker and Would-be Rescuer Asphyxiated" (PDF). Valero Refinery Asphyxiation Incident Case Study. Archived from the original (PDF) on 2015-09-22.
- ^ "Inquiry after man dies in chemical leak". BBC News. 1999-10-25. Archived from the original on 2017-01-07.
- ^ Liquid Nitrogen – Code of practice for handling. United Kingdom: Birkbeck, University of London. 2007. Archived from the original on 2018-06-12. Retrieved 2012-02-08.
- ^ Levey, Christopher G. "Liquid Nitrogen Safety". Thayer School of Engineering at Dartmouth. Archived from the original on 2016-03-05. Retrieved 2016-11-23.
- ^ National Institutes of Health. Protocol for Use and Maintenance of Oxygen Monitoring Devices. February 2014, at 1:35 UTC. Available at: https://www.ors.od.nih.gov/sr/dohs/documents/protocoloxygenmonitoring.pdf Archived 2020-12-05 at the Wayback Machine. Accessed June 23, 2020
Bibliography
- ISBN 978-0-08-037941-8.
External links
- Etymology of Nitrogen
- Nitrogen at The Periodic Table of Videos(University of Nottingham)
- Nitrogen podcast from the Royal Society of Chemistry's Chemistry World

![{\displaystyle {\ce {NaNO3{}+Na2O->[{\ce {Ag~crucible}}][{\ce {300^{\circ }C~for~7days}}]Na3NO4}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/ec729bc88f520e08fdce8a013dec8ae601d28509)