Noble gas
| Noble gases | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
| ↓ Period | |||||||||||
| 1 | Helium (He) 2 | ||||||||||
| 2 | Neon (Ne) 10 | ||||||||||
| 3 | Argon (Ar) 18 | ||||||||||
| 4 | Krypton (Kr) 36 | ||||||||||
| 5 | Xenon (Xe) 54 | ||||||||||
| 6 | Radon (Rn) 86 | ||||||||||
| 7 | Oganesson (Og) 118 | ||||||||||
|
Legend
| |||||||||||
The noble gases (historically the inert gases, sometimes referred to as aerogens
The noble gases' inertness, or tendency not to react with other chemical substances, results from their electron configuration: their outer shell of valence electrons is "full", giving them little tendency to participate in chemical reactions. Only a few hundred noble gas compounds are known to exist. For the same reason[clarification needed], noble gas atoms are small, and the only intermolecular force between them is the very weak London dispersion force, so their boiling points are all cryogenic, below 165 K (−108 °C; −163 °F).[2]
The inertness of noble gases makes them useful whenever chemical reactions are unwanted. For example, argon is used as a
The seventh member of group 18 is
History
Noble gas is translated from the
Ramsay continued his search for these gases using the method of fractional distillation to separate liquid air into several components. In 1898, he discovered the elements krypton, neon, and xenon, and named them after the Greek words κρυπτός (kryptós, "hidden"), νέος (néos, "new"), and ξένος (ksénos, "stranger"), respectively. Radon was first identified in 1898 by Friedrich Ernst Dorn,[12] and was named radium emanation, but was not considered a noble gas until 1904 when its characteristics were found to be similar to those of other noble gases.[13] Rayleigh and Ramsay received the 1904 Nobel Prizes in Physics and in Chemistry, respectively, for their discovery of the noble gases;[14][15] in the words of J. E. Cederblom, then president of the Royal Swedish Academy of Sciences, "the discovery of an entirely new group of elements, of which no single representative had been known with any certainty, is something utterly unique in the history of chemistry, being intrinsically an advance in science of peculiar significance".[15]
The discovery of the noble gases aided in the development of a general understanding of
In 1962, Neil Bartlett discovered the first chemical compound of a noble gas, xenon hexafluoroplatinate.[17] Compounds of other noble gases were discovered soon after: in 1962 for radon, radon difluoride (RnF
2),[18] which was identified by radiotracer techniques and in 1963 for krypton, krypton difluoride (KrF
2).[19] The first stable compound of argon was reported in 2000 when argon fluorohydride (HArF) was formed at a temperature of 40 K (−233.2 °C; −387.7 °F).[20]
In October 2006, scientists from the Joint Institute for Nuclear Research and Lawrence Livermore National Laboratory successfully created synthetically oganesson, the seventh element in group 18,[21] by bombarding californium with calcium.[22]
Physical and atomic properties
| Property[13][23] | Helium | Neon | Argon | Krypton | Xenon | Radon | Oganesson |
|---|---|---|---|---|---|---|---|
| Density (g/dm3) | 0.1786 | 0.9002 | 1.7818 | 3.708 | 5.851 | 9.97 | 7200 (predicted)[24] |
| Boiling point (K) | 4.4 | 27.3 | 87.4 | 121.5 | 166.6 | 211.5 | 450±10 (predicted)[24] |
| Melting point (K) | –[25] | 24.7 | 83.6 | 115.8 | 161.7 | 202.2 | 325±15 (predicted)[24] |
| Enthalpy of vaporization (kJ/mol) | 0.08 | 1.74 | 6.52 | 9.05 | 12.65 | 18.1 | – |
| Solubility in water at 20 °C (cm3/kg) | 8.61 | 10.5 | 33.6 | 59.4 | 108.1 | 230 | – |
| Atomic number | 2 | 10 | 18 | 36 | 54 | 86 | 118 |
pm ) |
31 | 38 | 71 | 88 | 108 | 120 | – |
| Ionization energy (kJ/mol) | 2372 | 2080 | 1520 | 1351 | 1170 | 1037 | 839 (predicted)[26] |
Electronegativity[27] |
4.16 | 4.79 | 3.24 | 2.97 | 2.58 | 2.60 | 2.59[28] |
The noble gases have weak
The noble gas
The
The noble gases are nearly
Chemical properties
The noble gases are colorless, odorless, tasteless, and nonflammable under standard conditions.[34] They were once labeled group 0 in the periodic table because it was believed they had a valence of zero, meaning their atoms cannot combine with those of other elements to form compounds. However, it was later discovered some do indeed form compounds, causing this label to fall into disuse.[13]
Electron configuration
Like other groups, the members of this family show patterns in its electron configuration, especially the outermost shells resulting in trends in chemical behavior:
| Z | Element | No. of electrons/shell |
|---|---|---|
| 2 | helium | 2 |
| 10 | neon | 2, 8 |
| 18 | argon | 2, 8, 8 |
| 36 | krypton | 2, 8, 18, 8 |
| 54 | xenon | 2, 8, 18, 18, 8 |
| 86 | radon | 2, 8, 18, 32, 18, 8 |
| 118 | oganesson | 2, 8, 18, 32, 32, 18, 8 (predicted) |
The noble gases have full valence
As a result of a full shell, the noble gases can be used in conjunction with the electron configuration notation to form the noble gas notation. To do this, the nearest noble gas that precedes the element in question is written first, and then the electron configuration is continued from that point forward. For example, the electron notation of phosphorus is 1s2 2s2 2p6 3s2 3p3, while the noble gas notation is [Ne] 3s2 3p3. This more compact notation makes it easier to identify elements, and is shorter than writing out the full notation of atomic orbitals.[36]
The noble gases cross the boundary between blocks—helium is an s-element whereas the rest of members are p-elements—which is unusual among the IUPAC groups. All other IUPAC groups contain elements from one block each. This causes some inconsistencies in trends across the table, and on those grounds some chemists have proposed that helium should be moved to group 2 to be with other s2 elements,[37][38][39] but this change has not generally been adopted.
Compounds
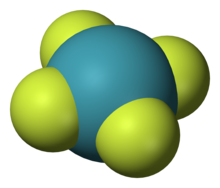
4), one of the first noble gas compounds to be discovered
The noble gases show extremely low chemical reactivity; consequently, only a few hundred noble gas compounds have been formed. Neutral compounds in which helium and neon are involved in chemical bonds have not been formed (although some helium-containing ions exist and there is some theoretical evidence for a few neutral helium-containing ones), while xenon, krypton, and argon have shown only minor reactivity.[40] The reactivity follows the order Ne < He < Ar < Kr < Xe < Rn ≪ Og.
In 1933,
2), xenon tetrafluoride (XeF
4), xenon hexafluoride (XeF
6), xenon tetroxide (XeO
4), and sodium perxenate
4XeO
6). Xenon reacts with fluorine to form numerous xenon fluorides according to the following equations:
- Xe + F2 → XeF2
- Xe + 2F2 → XeF4
- Xe + 3F2 → XeF6
Some of these compounds have found use in
Radon is more reactive than xenon, and forms chemical bonds more easily than xenon does. However, due to the high radioactivity and short half-life of radon isotopes, only a few fluorides and oxides of radon have been formed in practice.[47] Radon goes further towards metallic behavior than xenon; the difluoride RnF2 is highly ionic, and cationic Rn2+ is formed in halogen fluoride solutions. For this reason, kinetic hindrance makes it difficult to oxidize radon beyond the +2 state. Only tracer experiments appear to have succeeded in doing so, probably forming RnF4, RnF6, and RnO3.[48][49][50]
Krypton is less reactive than xenon, but several compounds have been reported with krypton in the oxidation state of +2.[40] Krypton difluoride is the most notable and easily characterized. Under extreme conditions, krypton reacts with fluorine to form KrF2 according to the following equation:
- Kr + F2 → KrF2
Compounds in which krypton forms a single bond to nitrogen and oxygen have also been characterized,[51] but are only stable below −60 °C (−76 °F) and −90 °C (−130 °F) respectively.[40]
Krypton atoms chemically bound to other nonmetals (hydrogen, chlorine, carbon) as well as some late transition metals (copper, silver, gold) have also been observed, but only either at low temperatures in noble gas matrices, or in supersonic noble gas jets.[40] Similar conditions were used to obtain the first few compounds of argon in 2000, such as argon fluorohydride (HArF), and some bound to the late transition metals copper, silver, and gold.[40] As of 2007, no stable neutral molecules involving covalently bound helium or neon are known.[40]
Extrapolation from periodic trends predict that oganesson should be the most reactive of the noble gases; more sophisticated theoretical treatments indicate greater reactivity than such extrapolations suggest, to the point where the applicability of the descriptor "noble gas" has been questioned.[52] Oganesson is expected to be rather like silicon or tin in group 14:[53] a reactive element with a common +4 and a less common +2 state,[54][55] which at room temperature and pressure is not a gas but rather a solid semiconductor. Empirical / experimental testing will be required to validate these predictions.[24][56] (On the other hand, flerovium, despite being in group 14, is predicted to be unusually volatile, which suggests noble gas-like properties.)[57][58]
The noble gases—including helium—can form stable
In addition to the compounds where a noble gas atom is involved in a
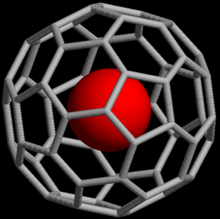
Noble gases can form
60 can be formed (the @ notation indicates He is contained inside C
60 but not covalently bound to it).[63] As of 2008, endohedral complexes with helium, neon, argon, krypton, and xenon have been created.[64] These compounds have found use in the study of the structure and reactivity of fullerenes by means of the nuclear magnetic resonance of the noble gas atom.[65]

2 according to the 3-center-4-electron bond model
Noble gas compounds such as
The chemistry of the heavier noble gases, krypton and xenon, are well established. The chemistry of the lighter ones, argon and helium, is still at an early stage, while a neon compound is yet to be identified.
Occurrence and production
The abundances of the noble gases in the universe decrease as their
| Abundance | Helium | Neon | Argon | Krypton | Xenon | Radon |
|---|---|---|---|---|---|---|
| Solar System (for each atom of silicon)[76] | 2343 | 2.148 | 0.1025 | 5.515 × 10−5 | 5.391 × 10−6 | – |
| Earth's atmosphere (volume fraction in ppm)[77] |
5.20 | 18.20 | 9340.00 | 1.10 | 0.09 | (0.06–18) × 10−19[78] |
| Igneous rock (mass fraction in ppm)[23] | 3 × 10−3 | 7 × 10−5 | 4 × 10−2 | – | – | 1.7 × 10−10 |
| Gas | 2004 price ( USD/m3)[79]
|
|---|---|
| Helium (industrial grade) | 4.20–4.90 |
| Helium (laboratory grade) | 22.30–44.90 |
| Argon | 2.70–8.50 |
| Neon | 60–120 |
| Krypton | 400–500 |
| Xenon | 4000–5000 |
For large-scale use, helium is extracted by fractional distillation from natural gas, which can contain up to 7% helium.[80]
Neon, argon, krypton, and xenon are obtained from air using the methods of liquefaction of gases, to convert elements to a liquid state, and fractional distillation, to separate mixtures into component parts. Helium is typically produced by separating it from natural gas, and radon is isolated from the radioactive decay of radium compounds.[13] The prices of the noble gases are influenced by their natural abundance, with argon being the cheapest and xenon the most expensive. As an example, the adjacent table lists the 2004 prices in the United States for laboratory quantities of each gas.
Applications

Noble gases have very low boiling and melting points, which makes them useful as
Helium is used as a component of

Since the Hindenburg disaster in 1937,[88] helium has replaced hydrogen as a lifting gas in blimps and balloons: despite an 8.6%[89] decrease in buoyancy compared to hydrogen, helium is not combustible.[13]
In many applications, the noble gases are used to provide an inert atmosphere. Argon is used in the synthesis of
Noble gases are commonly used in
The noble gases are used in
Some noble gases have direct application in medicine. Helium is sometimes used to improve the ease of breathing of people with
Noble gases, particularly xenon, are predominantly used in
Oganesson is too unstable to work with and has no known application other than research.
Discharge color

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
| Helium | Neon | Argon | Krypton | Xenon |
The color of gas discharge emission depends on several factors, including the following:[94]
- discharge parameters (local value of current density and electric field, temperature, etc. – note the color variation along the discharge in the top row);
- gas purity (even small fraction of certain gases can affect color);
- material of the discharge tube envelope – note suppression of the UV and blue components in the bottom-row tubes made of thick household glass.
See also
- Noble gas (data page), for extended tables of physical properties.
- Noble metal, for metals that are resistant to corrosion or oxidation.
- Inert gas, for any gas that is not reactive under normal circumstances.
- Industrial gas
- Octet rule
Notes
- PMID 25950423.
- ^ "Xenon | Definition, Properties, Atomic Mass, Compounds, & Facts". Britannica. 28 November 2023. Retrieved 12 January 2024.
- ^ from the original on 16 December 2023. Retrieved 23 October 2023.
- (PDF) from the original on 18 December 2023.
- S2CID 34534533.
- ^ Ozima 2002, p. 30
- ^ Ozima 2002, p. 4
- ^ "argon". Encyclopædia Britannica. 2008.
- ^ Oxford English Dictionary (1989), s.v. "helium". Retrieved 16 December 2006, from Oxford English Dictionary Online. Also, from quotation there: Thomson, W. (1872). Rep. Brit. Assoc. xcix: "Frankland and Lockyer find the yellow prominences to give a very decided bright line not far from D, but hitherto not identified with any terrestrial flame. It seems to indicate a new substance, which they propose to call Helium."
- ^ a b Ozima 2002, p. 1
- ^ Mendeleev 1903, p. 497
- S2CID 4251991.
- ^ a b c d e f g h i j "Noble Gas". Encyclopædia Britannica. 2008.
- ^ Cederblom, J. E. (1904). "The Nobel Prize in Physics 1904 Presentation Speech".
- ^ a b Cederblom, J. E. (1904). "The Nobel Prize in Chemistry 1904 Presentation Speech".
- PMID 17109437.
- ^ .
- .
- PMID 17812982.
- S2CID 4382128.
- . Retrieved 30 May 2014.
- .
- ^ a b c d Greenwood 1997, p. 891
- ^ PMID 32959952.
- ^ Liquid helium will only solidify if exposed to pressures well above atmospheric pressure, an effect explainable with quantum mechanics
- ^ Winter, Mark (2020). "Organesson: Properties of Free Atoms". WebElements: THE periodic table on the WWW. Retrieved 30 December 2020.
- .
- PMID 33828104.
- ^ ISBN 978-0-19-851245-5.
- ^ "John Beamish's Research on Solid Helium". Department of Physics, University of Alberta. 2008. Archived from the original on 31 May 2008.
- S2CID 40164915.
- .
- .
- ^ Wiley-VCH (2003). Ullmann's Encyclopedia of Industrial Chemistry – Volume 23. John Wiley & Sons. p. 217.
- ^ Ozima 2002, p. 35
- ^ CliffsNotes 2007, p. 15
- .
- ^ Bent Weberg, Libby (18 January 2019). ""The" periodic table". Chemical & Engineering News. 97 (3). Archived from the original on 1 February 2020. Retrieved 27 March 2020.
- PMID 23609097.
- ^
- .
- ^ Holloway 1968
- .
- . Retrieved 16 October 2007.
- PMID 11672087.
- ^ Harding 2002, pp. 90–99
- S2CID 250906059.
- S2CID 100225806.
- .
- PMID 25418862.
- .
- .
- ^ Kulsha, A. V. "Есть ли граница у таблицы Менделеева?" [Is there a boundary to the Mendeleev table?] (PDF). www.primefan.ru (in Russian). Retrieved 8 September 2018.
- ISBN 978-3-540-07109-9. Retrieved 4 October 2013.
- ^ Russian periodic table poster by A. V. Kulsha and T. A. Kolevich
- PMID 31343819.
- S2CID 97915854.
- ^ "Indication for a volatile element 114" (PDF). doc.rero.ch.
- .
- S2CID 4134617.
- ^ Greenwood 1997, p. 893
- .
- S2CID 41794612.
- .
- PMID 17924634.
- ^ Greenwood 1997, p. 897
- ^ Weinhold 2005, pp. 275–306
- .
- ^ Weiss, Achim. "Elements of the past: Big Bang Nucleosynthesis and observation". Max Planck Institute for Gravitational Physics. Archived from the original on 8 February 2007. Retrieved 23 June 2008.
- S2CID 16276658.
- ^ S2CID 85015694.
- Technische Universität Bergakademie Freiberg. Archived from the originalon 14 October 2007. Retrieved 26 June 2008.
- S2CID 31226092.
- ^ Tyler Irving (May 2011). "Xenon Dioxide May Solve One of Earth's Mysteries". L'Actualité chimique canadienne (Canadian Chemical News). Archived from the original on 9 February 2013. Retrieved 18 May 2012.
- ^ "A Citizen's Guide to Radon". U.S. Environmental Protection Agency. 26 November 2007. Retrieved 26 June 2008.
- S2CID 42498829. Archived from the original(PDF) on 7 November 2015. Retrieved 1 September 2015.
- ^ "The Atmosphere". National Weather Service. Retrieved 1 June 2008.
- ^ ISBN 3-527-30673-0.
- ^ .
- ^ Winter, Mark (2008). "Helium: the essentials". University of Sheffield. Retrieved 14 July 2008.
- ^ "Neon". Encarta. 2008.
- .
- PMID 4082343. Archived from the originalon 25 December 2010. Retrieved 8 April 2008.
- ^ Bennett 1998, p. 176
- ^ Vann, R. D., ed. (1989). "The Physiological Basis of Decompression". 38th Undersea and Hyperbaric Medical Society Workshop. 75(Phys)6-1-89: 437. Archived from the original on 7 October 2008. Retrieved 31 May 2008.
- ^ Maiken, Eric (1 August 2004). "Why Argon?". Decompression. Retrieved 26 June 2008.
- .
- ^ "Disaster Ascribed to Gas by Experts". The New York Times. 7 May 1937. p. 1.
- ^ Freudenrich, Craig (2008). "How Blimps Work". HowStuffWorks. Retrieved 3 July 2008.
- .
- ISBN 3-540-20056-8.
- PMID 15728132.
- PMID 11543065.
- ISBN 0-240-51323-1.
References
- Bennett, Peter B.; Elliott, David H. (1998). The Physiology and Medicine of Diving. ISBN 0-7020-2410-4.
- Bobrow Test Preparation Services (5 December 2007). CliffsAP Chemistry. ISBN 978-0-470-13500-6.
- Greenwood, N. N.; Earnshaw, A. (1997). Chemistry of the Elements (2nd ed.). Oxford:Butterworth-Heinemann. ISBN 0-7506-3365-4.
- Harding, Charlie J.; Janes, Rob (2002). Elements of the P Block. ISBN 0-85404-690-9.
- Holloway, John H. (1968). Noble-Gas Chemistry. ISBN 0-412-21100-9.
- Mendeleev, D. (1902–1903). Osnovy Khimii (The Principles of Chemistry) (in Russian) (7th ed.). New York, Collier.
- Ozima, Minoru; Podosek, Frank A. (2002). Noble Gas Geochemistry. ISBN 0-521-80366-7.
- Weinhold, F.; Landis, C. (2005). Valency and bonding. ISBN 0-521-83128-8.





