Factor IX
| F9 | |||
|---|---|---|---|
Gene ontology | |||
| Molecular function | |||
| Cellular component | |||
| Biological process | |||
| Sources:Amigo / QuickGO | |||
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr X: 139.53 – 139.56 Mb | Chr X: 59.04 – 59.08 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
Factor IX (or Christmas factor) (
Coagulation factor IX is on the World Health Organization's List of Essential Medicines.[6]
Physiology
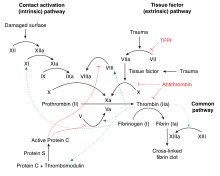
Factor IX is produced as a
Factor IX is inhibited by antithrombin.[7]
Factor IX expression increases with age in humans and mice. In mouse models, mutations within the promoter region of factor IX have an age-dependent phenotype.[9]
Domain architecture

The N-terminal EGF domain has been shown to at least in part be responsible for binding tissue factor.[10] Wilkinson et al. conclude that residues 88 to 109 of the second EGF domain mediate binding to platelets and assembly of the factor X activating complex.[11]
The structures of all four domains have been solved. A structure of the two EGF domains and the trypsin-like domain was determined for the pig protein.
Several structures of 'super active' mutants have been solved,[14] which reveal the nature of factor IX activation by other proteins in the clotting cascade.
Genetics

Because the
Polly, a transgenic cloned Poll Dorset sheep carrying the gene for factor IX, was produced by Dr Ian Wilmut at the Roslin Institute in 1997.[17]
Role in disease
| Clinical data | |
|---|---|
| Trade names | Benefix |
| License data | |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Clinical data | |
|---|---|
| Trade names | Idelvion |
| License data | |
| ATC code |
|
| Legal status | |
| Legal status | |
| Clinical data | |
|---|---|
| Trade names | Alprolix |
| License data | |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Clinical data | |
|---|---|
| Trade names | Refixia |
| ATC code |
|
| Legal status | |
| Legal status |
|
Deficiency of factor IX causes Christmas disease (
- nonacog alfa (brand name Benefix)[21]
- albutrepenonacog alfa (brand name Idelvion)[22]
- eftrenonacog alfa (brand name Alprolix)[23]
- nonacog beta pegol (brand name Refixia)[24]
- coagulation factor IX [recombinant] (Benefix)[25]
- coagulation factor IX [recombinant] (Idelvion)[26]
- coagulation factor IX (recombinant), Fc fusion protein (Alprolix)[27]
- coagulation factor IX [recombinant] (Ixinity)[28][29]
- coagulation factor IX [recombinant] (Rebinyn)[30]
- coagulation factor IX [recombinant] (Rixubis)[31]
- coagulation factor IX (human) (Alphanine SD)[32]
Some rare mutations of factor IX result in elevated clotting activity, and can result in clotting diseases, such as deep vein thrombosis. This gain of function mutation renders the protein hyperfunctional and is associated with familial early-onset thrombophilia.[33]
Factor IX deficiency is treated by injection of purified factor IX produced through cloning in various animal or animal cell vectors. Tranexamic acid may be of value in patients undergoing surgery who have inherited factor IX deficiency in order to reduce the perioperative risk of bleeding.[34]
A list of all the mutations in Factor IX is compiled and maintained by EAHAD.[35]
Coagulation factor IX is on the World Health Organization's List of Essential Medicines.[6]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000101981 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000031138 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ PMID 12997790.
- ^ hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ PMID 659613.
- PMID 9331959.
- PMID 7662969.
- ^ PMID 11723140.
- PMID 11714704.
- PMID 7568220.
- PMID 7547952.
- PMID 20004170.
- PMID 31819097.
- PMID 6959130.
- ^ Nicholl D. (2002). An Introduction to Genetic Engineering Second Edition. Cambridge University Press. p. 257.
- ^ "Health Canada New Drug Authorizations: 2016 Highlights". Health Canada. 14 March 2017. Retrieved 7 April 2024.
- PMID 25851415.
- S2CID 25251813.
- ^ "BeneFIX EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 17 June 2020. Retrieved 17 June 2020.
- ^ "Idelvion EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 17 June 2020. Retrieved 17 June 2020.
- ^ "Alprolix EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 11 August 2020. Retrieved 17 June 2020.
- ^ "Refixia EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 18 June 2020. Retrieved 17 June 2020.
- ^ "Benefix (coagulation factor ix- recombinant kit". DailyMed. 1 March 2023. Archived from the original on 29 January 2023. Retrieved 23 March 2024.
- ^ "Idelvion- coagulation factor ix recombinant human kit". DailyMed. 30 June 2023. Archived from the original on 27 January 2023. Retrieved 23 March 2024.
- ^ "Alprolix (coagulation factor ix- recombinant, fc fusion protein kit". DailyMed. 25 May 2023. Archived from the original on 7 February 2023. Retrieved 23 March 2024.
- ^ "Ixinity (coagulation factor ix- recombinant kit". DailyMed. 23 February 2021. Archived from the original on 28 September 2023. Retrieved 23 March 2024.
- ^ "Ixinity (coagulation factor ix- recombinant kit". DailyMed. 9 January 2024. Archived from the original on 3 December 2022. Retrieved 23 March 2024.
- ^ "Rebinyn ((coagulation factor ix- recombinant, glycopegylated kit". DailyMed. 11 August 2022. Archived from the original on 29 November 2022. Retrieved 23 March 2024.
- ^ "Rixubis (coagulation factor ix- recombinant kit". DailyMed. 22 March 2023. Archived from the original on 2 July 2022. Retrieved 23 March 2024.
- ^ "Alphanine SD (coagulation factor ix- human kit". DailyMed. 18 January 2024. Archived from the original on 18 February 2024. Retrieved 23 March 2024.
- PMID 19846852.
- PMID 21712351.
- ^ "Home: EAHAD Factor 9 Gene Variant Database". Archived from the original on 2020-10-28. Retrieved 2020-10-23.
Further reading
- Davie EW, Fujikawa K (1975). "Basic mechanisms in blood coagulation". Annual Review of Biochemistry. 44: 799–829. PMID 237463.
- Sommer SS (Jul 1992). "Assessing the underlying pattern of human germline mutations: lessons from the factor IX gene". FASEB Journal. 6 (10): 2767–74. S2CID 15211597.
- Lenting PJ, van Mourik JA, Mertens K (Dec 1998). "The life cycle of coagulation factor VIII in view of its structure and function". Blood. 92 (11): 3983–96. PMID 9834200.
- Lowe GD (Dec 2001). "Factor IX and thrombosis" (PDF). British Journal of Haematology. 115 (3): 507–13. (PDF) from the original on 2021-06-19. Retrieved 2019-12-11.
- O'Connell NM (Jun 2003). "Factor XI deficiency--from molecular genetics to clinical management". Blood Coagulation & Fibrinolysis. 14 (Suppl 1): S59-64. PMID 14567539.
- Du X (May 2007). "Signaling and regulation of the platelet glycoprotein Ib-IX-V complex". Current Opinion in Hematology. 14 (3): 262–9. S2CID 39904506.
External links
- Overview of all the structural information available in the PDB for UniProt: P00740 (Coagulation factor IX) at the PDBe-KB.
- GeneReviews/NCBI/NIH/UW entry on Hemophilia B
- The MEROPS online database for peptidases and their inhibitors: S01.214 Archived 2005-05-05 at the Wayback Machine