Nonylphenol
| |
| Names | |
|---|---|
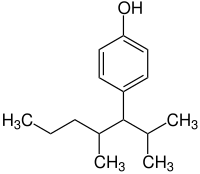
| IUPAC name
4-(2,4-dimethylheptan-3-yl)phenol
| |
| Other names
Phenol, nonyl-
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C15H24O | |
| Molar mass | 220.35 g/mol |
| Appearance | Light yellow viscous liquid with phenolic smell [1] |
| Density | 0.953 |
| Melting point | −8 to 2 °C (18 to 36 °F; 265 to 275 K) |
| Boiling point | 293 to 297 °C (559 to 567 °F; 566 to 570 K) |
| 6 mg/L (pH 7) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
low level endrocrine disruptor |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nonylphenols are a family of closely related
Structure and basic properties
Nonylphenols fall into the general chemical category of alkylphenols.[11] The structure of NPs may vary. The nonyl group can be attached to the phenol ring at various locations, usually the 4- and, to lesser extent, the 2-positions, and can be either branched or linear. A branched nonylphenol, 4-nonylphenol, is the most widely produced and marketed nonylphenol.[12] The mixture of nonylphenol isomers is a pale yellow liquid, although the pure compounds are colorless. The nonylphenols are moderately soluble in water [12] but soluble in alcohol.
Nonylphenol arises from the
Production
Nonylphenol can be produced industrially, naturally, and by the environmental degradation of alkylphenol ethoxylates. Industrially, nonylphenols are produced by the acid-catalyzed
Nonylphenols are also produced naturally in the environment. One organism, the
Another surfactant called
Applications
Nonylphenol is used in manufacturing
Prevalence in the environment
Nonylphenol persists in aquatic environments and is moderately
A major concern is that contaminated sewage sludge is frequently recycled onto agricultural land. The degradation of nonylphenol in soil depends on oxygen availability and other components in the soil. Mobility of nonylphenol in soil is low.[2]
Bioaccumulation is significant in water-dwelling organisms and birds, and nonylphenol has been found in internal organs of certain animals at concentrations of 10 to 1,000 times greater than the surrounding environment.[6] Due to this bioaccumulation and persistence of nonylphenol, it has been suggested that nonylphenol could be transported over long distances and have a global reach that stretches far from the site of contamination.[22]
Nonylphenol is not persistent in air, as it is rapidly degraded by hydroxyl radicals.[6]
Environmental hazards
Nonylphenol is considered to be an endocrine disruptor due to its ability to mimic estrogen and in turn disrupt the natural balance of hormones in affected organisms.[7][8][9][23][24] The effect is weak because nonylphenols are not very close structural mimics of estradiol, but the levels of nonylphenol can be sufficiently high to compensate.

The effects of nonylphenol in the environment are most applicable to aquatic species. Nonylphenol can cause endocrine disruption in fish by interacting with estrogen receptors and androgen receptors. Studies report that nonylphenol competitively displaces estrogen from its receptor site in rainbow trout.[25] It has much less affinity for the estrogen receptor than estrogen in trout (5 x 10−5 relative binding affinity compared to estradiol) making it 100,000 times less potent than estradiol.[25][26] Nonylphenol causes the feminization of aquatic organisms, decreases male fertility, and decreases survival in young fish.[2] Studies show that male fish exposed to nonylphenol have lower testicular weight.[25] Nonylphenol can disrupt steroidogenesis in the liver. One function of endogenous estrogen in fish is to stimulate the liver to make vitellogenin, which is a phospholipoprotein.[25] Vitellogenin is released by the maturing female and sequestered by developing oocytes to produce the egg yolk.[25] Males do not normally produce vitellogenin, but when exposed to nonylphenol they produce similar levels of vitellogenin to females.[25] The concentration needed to induce vitellogenin production in fish is 10 ug/L for NP in water.[25] Nonylphenol can also interfere with the level of FSH (follicle-stimulating hormone) being released from the pituitary gland. Concentrations of NP that inhibit reproductive development and function in fish also damages kidneys, decreases body weight, and induces stressed behavior.[27]
Human health hazards
Alkylphenols like nonylphenol and
Effects in pregnant women
Nonylphenol has also been shown to affect
Effects on metabolism
Nonylphenol has been shown to act as an obesity enhancing chemical or obesogen, though it has paradoxically been shown to have anti-obesity properties.[30] Growing embryos and newborns are particularly vulnerable when exposed to nonylphenol because low-doses can disrupt sensitive processes that occur during these important developmental periods.[31] Prenatal and perinatal exposure to nonylphenol has been linked with developmental abnormalities in adipose tissue and therefore in metabolic hormone synthesis and release (Merrill 2011). Specifically, by acting as an estrogen mimic, nonylphenol has generally been shown to interfere with hypothalamic appetite control.[30] The hypothalamus responds to the hormone leptin, which signals the feeling of fullness after eating, and nonylphenol has been shown to both increase and decrease eating behavior by interfering with leptin signaling in the midbrain.[30] Nonylphenol has been shown mimic the action of leptin on neuropeptide Y and anorectic POMC neurons, which has an anti-obesity effect by decreasing eating behavior. This was seen when estrogen or estrogen mimics were injected into the ventromedial hypothalamus.[32] On the other hand, nonylphenol has been shown to increase food intake and have obesity enhancing properties by lowering the expression of these anorexigenic neurons in the brain.[33] Additionally, nonylphenol affects the expression of ghrelin: an enzyme produced by the stomach that stimulates appetite.[34] Ghrelin expression is positively regulated by estrogen signaling in the stomach, and it is also important in guiding the differentiation of stem cells into adipocytes (fat cells). Thus, acting as an estrogen mimic, prenatal and perinatal exposure to nonylphenol has been shown to increase appetite and encourage the body to store fat later in life.[35] Finally, long-term exposure to nonylphenol has been shown to affect insulin signaling in the liver of adult male rats.[36]
Cancer
Nonylphenol exposure has also been associated with breast cancer.[2] It has been shown to promote the proliferation of breast cancer cells, due to its agonistic activity on ERα (estrogen receptor α) in estrogen-dependent and estrogen-independent breast cancer cells. Some argue that nonylphenol's suggested estrogenic effect coupled with its widespread human exposure could potentially influence hormone-dependent breast cancer disease.[37]
Human exposure and breakdown
Exposure
Diet seems the most significant source of exposure of nonylphenol to humans. For example, food samples were found with concentrations ranging from 0.1 to 19.4 µg/kg in a diet survey in Germany and a daily intake for an adult were calculated to be 7.5 µg/day.[38] Another study calculated a daily intake for the more exposed group of infants in the range of 0.23-0.65 µg/ kg bodyweight/ day.[39] In Taiwan, nonylphenol concentrations in food ranged from 5.8 to 235.8 µg/kg. Seafood in particular was found to have a high concentration of nonylphenol.[40]
One study conducted in Italian women showed that nonylphenol was one of the highest contaminants at a concentration of 32 ng/mL in
Drinking water does not represent a significant source of exposure in comparison to other sources such as food packing materials, cleaning products, and various skin care products. Concentrations of nonylphenol in treated drinking water varied from 85 ng/L in Spain to 15 ng/L in Germany.[2]
Microgram amounts of nonylphenol have also been found in the saliva of patients with dental sealants.[37]
Breakdown
When humans orally ingest nonylphenol, it is rapidly absorbed in the
Analytics
There are standard GC-MS and HPLC protocols for the detection of nonylphenols within environmental sample matrices such as foodstuffs, drinking water and biological tissue.[41][42] Industrially produced nonylphenol (the source most likely to be found in the environment) contains a mixture of structural isomers,[43] and while these protocols are able to detect this mixture, they are typically unable to resolve the individual nonylphenol isomers within it. However, a methodological study has indicated that better isomeric resolution can be achieved in bulk nonylphenol samples using a GC-MS/MS (tandem mass-analyzer) system,[44] suggesting that this technique could also improve the resolution of nonylphenol isomers in environmental sample analyses; further improvements in the resolution of nonylphenol isomers have been achieved through the use of two-dimensional GC at the separation stage, as part of a GC x GC-TOF-MS system.[45]
In contrast to environmental sample analyses, synthetic studies of nonylphenols have more control over sample state, concentration and preparation, simplifying the use of powerful structural identification techniques like NMR - capable of identifying the individual nonylphenol isomers.[46] In a preliminary investigation of the relationship between nonylphenol sidechain branching patterns and estrogenic potential, the authors identified 211 possible structural isomers of p-nonylphenol alone, which expanded to 550 possible p-nonylphenol compounds when taking chiral C-atoms into consideration.[47] Because stereochemical factors are thought to contribute to the biological activity of nonylphenols, analytical techniques sensitive to chirality, such as enantioselective HPLC and certain NMR protocols, are desirable in order to further study these relationships.[48][49][50]
Regulation
The production and use of nonylphenol and nonylphenol ethoxylates is prohibited for certain situations in the European Union due to its effects on health and the environment.[2][51] In Europe, due to environmental concerns, they also have been replaced by more expensive alcohol ethoxylates, which are less problematic for the environment due to their ability to degrade more quickly than nonylphenols. The European Union has also included NP on the list of priority hazardous substances for surface water in the Water Framework Directive. They are now implementing a drastic reduction policy of NP's in surface waterways. The Environmental quality standard for NP was proposed to be 0.3 ug/L.[2] In 2013 nonylphenols were registered on the REACH candidate list.
In the US, the
In other Asian and South American countries nonylphenol is still widely available in commercial detergents, and there is little regulation.[52]
References
- ^ Record of Nonylphenol, mixed isomers in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 6 April 2011.
- ^ PMID 18282600.
- ^ "Epoxy Nonyl Phenol Alert is it in your epoxy?". www.epoxyproducts.com. Retrieved 2021-03-18.
- ^ "para nonyl phenol" (PDF). squarespace.
- ^ "No, no, nonyl(phenol)". Healthy Building Network. Retrieved 2021-03-18.
- ^ a b c d e Mergel, Maria. "Nonylphenol and Nonylphenol Ethoxylates." Toxipedia.org. N.p., 1 Nov. 2011. Web. 27 Apr. 2014.
- ^ S2CID 39833642.
- ^ PMID 18800507.
- ^ PMID 24345275.
- PMID 24530924.
- ^ S2CID 16158451.
- ^ a b c d EPA. 2010. Nonylphenol (NP) and Nonylphenal Ethoxylates (NPEs) Action Plan. February, 2014.
- PMID 15260341.
- PMID 16083769.
- ISSN 0021-9665.
- )
- .
- ^ Nonylphenol and nonylphenol ethoxylates action plan. U.S. Environmental Protection Agency (EPA). August 2010.
- ^ Samaras et al. 2013. Fate of selected pharmaceuticals and synthetic endocrine disruptive compounds during wastewater treatment and sludge anaerobic digestion. Journal of Hazardous Materials, vol. 244-245, January 2013, p. 259-267.Samaras et al., 2013
- ^ Stasinakis et al., 2013. Contribution of primary and secondary treatment on the removal of benzothiazoles, benzotriazoles, endocrine disruptors, pharmaceuticals and perfluorinated compounds in a sewage treatment plant. Sci. Total Environ. Vol. 463-464, October 2013, p. 1067-1075. Stasinakis et al., 2013
- PMID 11851490.
- ^ Pesticide Action Network North America. PANNA. Nonylphenol Etoxylates.. Accessed 9/30/2011. UK Environment Agency
- PMID 18282600.
- ^ Minnesota Pollution Control Agency Statewide Endocrine Disrupting Compound Monitoring Study, 2007 - 2008
- ^ a b c d e f g WHO (World Health Organization). 2002. Integrated Risk Assessment: Nonylphenol Case Study. WHO/IPCS/IRA/12/04, World Health Organization, Geneva, Switzerland.
- ^ Canada, Health (2004-07-26). "ARCHIVED - Priority Substances List Assessment Report for Nonylphenol and its Ethoxylates". aem. Retrieved 2019-10-03.
- PMID 16818251.
- PMID 21888958.
- ^ PMID 20194071.
- ^ PMID 19433244.
- ^ "Endocrine Disruption Fact Sheet." Endocrinedisruption.org. TEDX the Endocrine Disruption Exchange, 7 Nov. 2011. Web. 25 Apr. 2014.
- S2CID 20370652.
- S2CID 9419249.
- PMID 17003276.
- PMID 15178745.
- S2CID 38257683.
- ^ S2CID 24384354.
- PMID 11993862.
- PMID 21185059.
- ^ PMID 18410965.
- PMID 12458934.
- PMID 14664841.
- .
- PMID 16271268.
- PMID 16427065.
- PMID 20452641.
- S2CID 39833642.
- PMID 17027900.
- PMID 18553459.
- S2CID 11005914.
- ^ Official Journal of the European Union: DIRECTIVE 2003/53/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 18 June 2003 amending for the 26th time Council Directive 76/769/EEC relating to restrictions on the marketing and use of certain dangerous substances and preparations (nonylphenol, nonylphenol ethoxylate and cement), July 17, 2003
- ^ PMID 19476957.
