Nooglutyl
| |
| Names | |
|---|---|
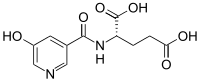
| IUPAC name
N-[(5-Hydroxy-3-pyridinyl)carbonyl]-L-glutamic acid
| |
| Systematic IUPAC name
(2S)-2-(5-Hydroxypyridine-3-carboxamido)pentanoic acid | |
| Other names
Nooglutil
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C11H12N2O6 | |
| Molar mass | 268.225 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nooglutyl is a
Russian Academy of Medical Sciences as a potential treatment for amnesia.[1]
In animal models, it has a variety of central nervous system effects.[2][3][4][5]
Nooglutyl has some structural similarities to Picamilon (N-Nicotinoyl-GABA). Nooglutyl is N-5-HydroxyNicotinoyl-L-Glutamic acid.
