Norethisterone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Many |
| Other names | NET; Norethindrone; NSC-9564; LG-202; Ethinylnortestosterone; Norpregneninolone; Anhydrohydroxy-norprogesterone; Ethinylestrenolone; 17α-Ethynyl-19-nortestosterone; 17α-Ethynylestra-4-en-17β-ol-3-one; 17α-Hydroxy-19-norpregn-4-en-20-yn-3-one |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604034 |
| License data | |
Progestin | |
| ATC code | |
| Legal status | |
| Legal status |
|
3β-HSD, and aromatase | |
| Elimination half-life | 5.2–12.8 hours (mean 8.0 hours)[1] |
| Identifiers | |
| |
JSmol) | |
| Melting point | 203 to 204 °C (397 to 399 °F) |
| |
| |
| (verify) | |
Norethisterone, also known as norethindrone and sold under many brand names, is a
Norethisterone was discovered in 1951 and was one of the first progestins to be developed.[11][12][13] It was first introduced for medical use on its own in 1957 and was introduced in combination with an estrogen for use as a birth control pill in 1963.[13][14] It is sometimes referred to as a "first-generation" progestin.[15][16] Along with desogestrel, it is one of the only progestins that is widely available as a progestogen-only "mini pill" for birth control.[17][18] Norethisterone is marketed widely throughout the world.[19] It is available as a generic medication.[20] In 2021, it was the 140th most commonly prescribed medication in the United States, with more than 4 million prescriptions.[21][22] It is on the World Health Organization's List of Essential Medicines.[23]
Medical uses
Norethisterone is used as a hormonal contraceptive in combination with an estrogen – usually ethinylestradiol (EE) – in combined oral contraceptive pills and alone in progestogen-only pills.
Another medical use of norethisterone is to alleviate
| Composition | Dose | Brand names | Use |
|---|---|---|---|
| NET only | Low (e.g., 0.35 mg) | Multiple[a] | Progestogen-only oral contraceptive
|
| NET or NETA only | High (e.g., 5 mg, 10 mg) | Multiple[b] | Gynecological disorders and other uses
|
| NETE only | Injection (e.g., 200 mg) | Multiple[c] | Progestogen-only injectable contraceptive |
| NET or NETA with ethinylestradiol | Low (e.g., 0.4 mg, 0.5 mg, 0.75 mg, 1 mg, 1.5 mg) | Multiple[d] | Combined oral contraceptive
|
| NET with mestranol | Low (e.g., 1 mg, 2 mg) | Multiple[e] | Combined oral contraceptive |
| NETA with estradiol | Low (e.g., 0.1 mg, 0.5 mg) | Multiple[f] | Combined menopausal hormone therapy
|
| NETE with estradiol valerate | Injection (e.g., 50 mg) | Multiple[g] | Combined injectable contraceptive
|
| Abbreviations: NET = Norethisterone. NETA = Norethisterone acetate. NETE = Norethisterone enanthate. Sources: [25][26] [27][28] Notes:
| |||
Contraindications
High-dose (10 mg/day) norethisterone has been associated with
Side effects
At contraceptive and hormone replacement dosages (0.35 to 1 mg/day), norethisterone has essentially progestogenic
At high doses (5 to 60 mg/day), for instance those used in the treatment of gynecological disorders, norethisterone can cause
High doses of NETA (10 mg/day) have been associated with abnormal
Androgenic
Due to its weak androgenic activity, norethisterone can produce androgenic
The sebaceous glands are highly androgen-sensitive and their size and activity are potential markers of androgenic effect.[37] A high dosage of 20 mg/day norethisterone or NETA has been found to significantly stimulate the sebaceous glands, whereas lower dosages of 5 mg/day and 2.5 mg/day norethisterone and NETA, respectively, did not significantly stimulate sebum production and were consequently regarded as devoid of significant androgenicity.[37] Conversely, dosages of norethisterone of 0.5 to 3 mg/day have been found to dose-dependently decrease SHBG levels (and hence to suppress hepatic SHBG production), which is another highly sensitive marker of androgenicity.[38]
A large clinical study of high to very high oral dosages of norethisterone (10 to 40 mg/day) administered for prolonged periods of time (4 to 35 weeks) to prevent
In a letter to the editor on the topic of virilization caused by high dosages of NETA in women, a physician expressed that they had not observed the "slightest evidence of virilization" and that there had "certainly been no hirsutism nor any voice changes" in 55 women with advanced breast cancer that they had treated with 30 to 60 mg/day norethisterone for up to six months.[39]
High-dosage norethisterone has been used to suppress
Estrogenic
Norethisterone is weakly
Overdose
There have been no reports of serious side effects with overdose of norethisterone, even in small children.[48] As such, overdose usually does not require treatment.[48] High dosages of as much as 60 mg/day norethisterone have been studied for extended treatment durations without serious adverse effects described.[39]
Interactions
Pharmacology
Pharmacodynamics
Norethisterone is a potent
| Compound | Typea | PR | AR | ER | GR | MR | SHBG | CBG
|
|---|---|---|---|---|---|---|---|---|
| Norethisterone | – | 67–75 | 15 | 0 | 0–1 | 0–3 | 16 | 0 |
| 5α-Dihydronorethisterone | Metabolite | 25 | 27 | 0 | 0 | ? | ? | ? |
| 3α,5α-Tetrahydronorethisterone | Metabolite | 1 | 0 | 0–1 | 0 | ? | ? | ? |
| 3α,5β-Tetrahydronorethisterone | Metabolite | ? | 0 | 0 | ? | ? | ? | ? |
| 3β,5α-Tetrahydronorethisterone | Metabolite | 1 | 0 | 0–8 | 0 | ? | ? | ? |
| Ethinylestradiol | Metabolite | 15–25 | 1–3 | 112 | 1–3 | 0 | 0.18 | 0 |
| Norethisterone acetate | Prodrug | 20 | 5 | 1 | 0 | 0 | ? | ? |
| Norethisterone enanthate | Prodrug | ? | ? | ? | ? | ? | ? | ? |
| Noretynodrel | Prodrug | 6 | 0 | 2 | 0 | 0 | 0 | 0 |
| Etynodiol | Prodrug | 1 | 0 | 11–18 | 0 | ? | ? | ? |
| Etynodiol diacetate | Prodrug | 1 | 0 | 0 | 0 | 0 | ? | ? |
| Lynestrenol | Prodrug | 1 | 1 | 3 | 0 | 0 | ? | ? |
| Notes: Values are percentages (%). Reference , or neither of norethisterone. Sources: See template. | ||||||||
Progestogenic activity
Norethisterone is a potent
Androgenic activity
Norethisterone has approximately 15% of the affinity of the
Norethisterone (0.5 to 3 mg/day) has been found to dose-dependently decrease circulating SHBG levels, which is a common property of androgens and is due to AR-mediated suppression of hepatic SHBG production.[38] The drug also has estrogenic activity, and estrogens are known to increase SHBG hepatic production and circulating levels, so it would appear that the androgenic activity of norethisterone overpowers its estrogenic activity in this regard.[38]
Norethisterone is bound to a considerable extent (36%) to SHBG in circulation.[3] Although it has lower affinity for SHBG than endogenous androgens and estrogens,[55] Norethisterone may displace testosterone from SHBG and thereby increase free testosterone levels, and this action may contribute to its weak androgenic effects.[56]
Estrogenic activity

Norethisterone binds to the ERs, the
Neurosteroid activity
Like
Steroidogenesis inhibition
Norethisterone is a substrate for and is known to be an
Norethisterone and its major active metabolite 5α-DHNET have been found to act as
Other activities
Norethisterone is a very weak inhibitor of CYP2C9 and CYP3A4 (IC50 = 46 μM and 51 μM, respectively), but these actions require very high concentrations of norethisterone that are far above therapeutic circulating levels (which are in the nanomolar range) and hence are probably not clinically relevant.[3]
Norethisterone and some of its 5α-reduced metabolites have been found to produce
Norethisterone stimulates the
Antigonadotropic effects
Due to its progestogenic activity, norethisterone suppresses the
The ovulation-inhibiting dosage of both oral norethisterone and oral NETA is about 0.5 mg/day in women.[3][66][67] However, some conflicting data exist, suggesting that higher doses might be necessary for full inhibition of ovulation.[68] An intramuscular injection of 200 mg NETE has been found to prevent ovulation and suppress levels of estradiol, progesterone, luteinizing hormone (LH), and follicle-stimulating hormone (FSH) in women.[69][70][71][72]
Early studies of oral norethisterone in men employing doses of 20 to 50 mg/day observed suppression of
A single 200 mg intramuscular injection of NETE alone or in combination with 2 mg estradiol valerate has been found to produce a rapid, strong, and sustained decrease in gonadotropin and testosterone levels for up to one month in men.[65][81][82] Intramuscular injections of 200 mg NETE once every 3 weeks have also been found to suppress spermatogenesis in men.[73][83] Similarly, a single intramuscular injection of 50 mg NETE in combination with 5 mg estradiol valerate has been found to strongly suppress testosterone levels in men.[80] Levels of testosterone decreased from ~503 ng/dL at baseline to ~30 ng/dL at the lowest point (–94%) which occurred at day 7 post-injection.[80]
Pharmacokinetics
The pharmacokinetics of norethisterone have been reviewed.[3][84]
Absorption
The
Distribution
The plasma protein binding of norethisterone is 97%.[3] It is bound 61% bound to albumin and 36% bound to SHBG.[3]
Metabolism
Norethisterone has an
Norethisterone is used in birth control pills, opposed to progesterone itself, because it is not metabolized as rapidly as progesterone when consumed orally. When progesterone is consumed orally it is rapidly metabolized in the gastrointestinal tract and the liver, and broken down into many different metabolites. Whereas, norethisterone is not as rapidly metabolized allowing norethisterone to be present in higher quantities allowing it to more effectively compete for progesterone receptor binding sites.[3]
Elimination
Norethisterone is eliminated 33 to 81% in urine and 35 to 43% in feces.[100]
Chemistry
Norethisterone, also known as 17α-ethynyl-19-nortestosterone or as 17α-ethynylestra-4-en-17β-ol-3-one, is a
Derivatives
Norethisterone (NET) is the
|
|
Several of these act as prodrugs of norethisterone, including NETA, NETE, etynodiol diacetate, lynestrenol, and quingestanol acetate.[105][106][107] Noretynodrel may also be a prodrug of norethisterone.[3][1] NETA is taken by mouth similarly to norethisterone, while NETE is given by injection into muscle.[10]
Non-17α-ethynylated
19-Nortestosterone (19-NT) progestins which are technically not derivatives of norethisterone (as they do not have a C17α ethynyl group) but are still closely related (with other substitutions at the C17α and/or C16β positions) include the following marketed medications:[101][27]
- The C17α vinyl (ethenyl) derivatives norgesterone (17α-vinyl-δ5(10)-19-NT) and norvinisterone (17α-vinyl-19-NT)
- The C17α allyl derivatives allylestrenol (3-deketo-17α-allyl-19-NT) and altrenogest (17α-allyl-δ9,11-19-NT)
- The C17α alkyl derivative normethandrone (17α-methyl-19-NT)
- The C17α cyanomethyl derivative dienogest (17α-cyanomethyl-δ9-19-NT)
- The C16β ethyl derivative oxendolone (16β-ethyl-19-NT)
Many anabolic steroids of the 19-nortestosterone family, like norethandrolone and ethylestrenol, are also potent progestogens, but were never marketed as such.
Synthesis
Chemical syntheses of norethisterone have been published.[101][84]
Synthesis 1

Estradiol 3-methyl ether (1, EME) is partially reduced to the 1,5-diene (2) as also occurs for the first step in the synthesis of nandrolone.
In practice, these and all other combined oral contraceptives are mixtures of 1 to 2% EE or
Synthesis 2
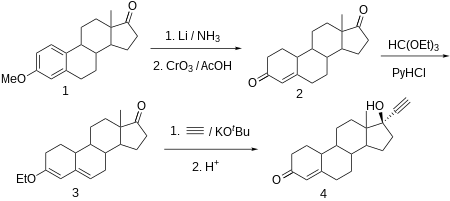
Norethisterone is made from
History
Norethisterone was synthesized for the first time by chemists
Society and culture
Generic names
Norethisterone is the
Brand names
Norethisterone is available in Bangladesh as Menogia (ACI), Normens (Renata) etc. Norethisterone (NET), including as NETA and NETE, has been marketed under many brand names throughout the world.[27][19]
Availability
United States
Norethisterone was previously available alone in 5 mg tablets under the brand names Norlutin in the United States, but this formulation has since been discontinued in this country.[26] However, NETA remains available alone in 5 mg tablets under the brand name Aygestin in the United States.[26] It is one of the only non-contraceptive progestogen-only drug formulations that remains available in the United States.[26] The others include progesterone, medroxyprogesterone acetate, megestrol acetate, and hydroxyprogesterone caproate, as well as the atypical agent danazol.[26]
Both norethisterone and NETA are also available in the United States as contraceptives.[26] Norethisterone is available both alone (brand names Camila, Errin, Heather, Micronor, Nor-QD, others) and in combination with EE (Norinyl, Ortho-Novum, others) or mestranol (Norinyl, Ortho-Novum, others), while NETA is available only in combination with EE (Norlestrin, others).[26] NETE is not available in the United States in any form.[26][114][115]
Research
Norethisterone, as NETA and NETE, has been studied for use as a potential male hormonal contraceptive in combination with testosterone in men.[116][117]
Long-acting norethisterone
References
- ^ S2CID 27018468.
- ^ PMID 8842581.
- ^ (PDF) from the original on 22 August 2016. Retrieved 6 September 2018.
- ^ S2CID 10809537.
- ^ PMID 8520623.
- ISBN 978-0-323-29368-6. Archivedfrom the original on 9 March 2023. Retrieved 8 January 2018.
- ^ PMID 2170822.
- ^ https://www.accessdata.fda.gov/drugsatfda_docs/label/2007/018405s023lbl.pdf Archived 10 February 2017 at the Wayback Machine [bare URL PDF]
- ^ PMID 14450719.
- ^ ISBN 978-92-832-1291-1. Archivedfrom the original on 10 January 2023. Retrieved 12 October 2016.
Norethisterone and its acetate and enanthate esters are progestogens that have weak estrogenic and androgenic properties.
- ^ PMID 16389045.
- ^ ISBN 978-1-59745-150-5.
- ^ ISBN 978-0-300-16791-7.
- ^ ISBN 978-0-8155-1856-3.
- ISBN 978-1-59708-001-9. Archivedfrom the original on 10 January 2023. Retrieved 8 January 2018.
- ISBN 978-93-5025-240-6. Archivedfrom the original on 9 March 2023. Retrieved 8 January 2018.
- PMID 24226383.
- PMID 14759612.
- ^ a b "Norethisterone". Archived from the original on 15 September 2018. Retrieved 8 January 2018.
- ^ "Generic Aygestin Availability". Archived from the original on 24 August 2018. Retrieved 8 January 2018.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Norethindrone - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- PMID 23303565.
- ^ "Norethisterone". Drugs.com.
- ^ a b c d e f g h "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Archived from the original on 16 November 2016. Retrieved 27 November 2016.
- ^ ISBN 978-3-88763-075-1.
- ISBN 978-92-832-1272-0.
- ^ ISBN 978-0-08-093292-7.
- PMID 9845522.
- ^ S2CID 26886374.
- ^ S2CID 83941717.
- ^ PMID 11836281.
- ^ PMID 20159314.
- ^ PMID 22786490.
- S2CID 9947402.
- ^ PMID 5843402.
- ^ PMID 9494772.
- ^ PMC 1958463.
- ^ S2CID 204094551. Archived from the originalon 20 December 2016. Retrieved 6 December 2016.
- ^ PMID 4746398.
- PMID 14272499.
- PMID 22154396.
- ^ PMID 13942007.
- ^ PMID 17341557.
- ISBN 978-1-108-16595-2.
- ^ S2CID 205931204.
- ^ a b https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/016954s106lbl.pdf Archived 10 February 2017 at the Wayback Machine [bare URL PDF]
- ^ a b c d e Kuhl H (2011). "Pharmacology of Progestogens" (PDF). J Reproduktionsmed Endokrinol. 8 (1): 157–177. Archived (PDF) from the original on 11 October 2016. Retrieved 10 October 2016.
- PMID 2934946.
- ISBN 978-0-323-29348-8.
- PMID 13332050.
- S2CID 41356223.
Many synthetic steroids with high myotrophic activity exhibit myotrophic–androgenic dissociation, since, due to changes introduced in the structure of ring A, they will probably not be substrates for the 5α-reductases [85]. 5α-Reduction does not always amplify the androgenic potency in spite of high RBA of androgens to the AR. This is the case for norethisterone (Fig. 1, 34), a synthetic 19-nor-17α-ethynyl testosterone derivative, which also undergoes enzyme-mediated 5α-reduction and exerts potent androgenic effects in target organs. 5α-Reduced norethisterone displays a higher AR binding but shows a significantly lower androgenic potency than unchanged norethisterone [102,103].
- ^ S2CID 33771349.
- ISBN 978-1-4831-6366-6.
Norethisterone binds to SHBG with less affinity than endogenous androgens and oestrogens [...]
- ISBN 978-1-59745-179-6.
- PMID 9048584.
- S2CID 27088428.
- ^ PMID 27339142.
- ^ PMID 8205267.
- PMID 12954372.
- ^ S2CID 29808177.
- PMID 31512725.
- (PDF) from the original on 28 August 2021. Retrieved 11 December 2019.
- ^ PMID 14723979.
- PMID 4878826.
- S2CID 44601264.
- PMID 22078182.
- ISSN 0178-0328.
- PMID 1122756.
- PMID 606499.
- PMID 572279.
- ^ S2CID 24859886.
- S2CID 32637425.
- PMID 14400846. Archived from the originalon 18 December 2018. Retrieved 11 December 2019.
- PMID 14261416.
- ISSN 0011-3891. Archived from the original(PDF) on 11 December 2019. Retrieved 11 December 2019.
- ISSN 0010-7824.
- S2CID 41502711.
- ^ a b c d Alvarez BD (11 May 2011). Efecto de una Dosis de 50 mg de Enantato de Noretisterona y 5 mg de Valerato de Estradiol en los Niveles de Testosterona Total en Hombres Mexicanos Sanos [Effect of a Dose of 50 mg of Norethisterone Enanthate and 5 mg of Estradiol Valerate on Total Testosterone Levels in Healthy Mexican Men] (MSc). National Polytechnic Institute of Mexico. Archived from the original on 16 September 2022. Retrieved 12 September 2022.
- ISBN 978-1-61737-314-5.
- S2CID 70515136.
- )
- ^ ISBN 978-3-642-99941-3. Archivedfrom the original on 10 January 2023. Retrieved 19 September 2018.
- PMID 2620531.
- S2CID 3813229.
- OCLC 278011135.
- ISSN 0389-2328.
- ISBN 978-1-349-05954-6.
- ISBN 978-1-4684-5036-1.
- S2CID 43298472.
- PMID 4206183.
- S2CID 20759583.
- PMID 2143719.
- ^ S2CID 19797254.
[...] several mono- and disulphated as well as mono- and diglucuronidated metabolites of NET have been detected in urine from NET treated women [16,17]. In unconjugated form these NET (or MeNET) metabolites are represented by 5α- and 5β-reduced NET (5α-NET or 5β-NET) and by 3α- and 3β-hydrogenated 5α-NET and 5β-NET, leading to 3α,5α-NET, 3β,5α-NET, 3α,5β-NET and 3β,5β-NET or their corresponding MeNET metabolites (Figs. 1 and 2). These steroid conversions of NET or MeNET may take place in the liver, but also in the pituitary, endometrium, prostate, vagina and breast. The enzymes involved in these metabolic processes are 5α- and 5β-reductase as well as 3α- and 3β-hydroxysteroid dehydrogenase (HSD).
- PMID 3871879.
- PMID 3090814.
- PMID 19008332.
- PMID 27562873.
- ISBN 978-0-7216-7843-6.
- ^ ISBN 978-1-4757-2085-3. Archivedfrom the original on 10 January 2023. Retrieved 5 October 2016.
- S2CID 2882899.
- ISBN 978-1-4612-2730-4.
- ISBN 978-0-323-00201-1.
- PMID 2256526.
- PMID 20851224.
- PMID 3543501.
Similar androgenic potential is inherent to norethisterone and its prodrugs (norethisterone acetate, ethynodiol diacetate, lynestrenol, norethynodrel, quingestanol [acetate]).
- ^ a b Frank B. Colton, U.S. patent 2,655,518 (1952 to Searle & Co).
- .
- .
- PMID 4914401.
- ^ U.S. patent 2,744,122
- ^ U.S. patent 2,774,777
- ISBN 978-1-57607-181-6. Archivedfrom the original on 10 January 2023. Retrieved 27 November 2016.
- ISBN 978-1-58901-807-5.
- (PDF) from the original on 5 December 2020. Retrieved 28 December 2018.
- ISBN 9781139003353.
- S2CID 45898359.
Further reading
- Brogden RN, Speight TM, Avery GS (1973). "Progestagen-only oral contraceptives: a preliminary report of the action and clinical use of norgestrel and norethisterone". Drugs. 6 (3): 169–81. S2CID 42295736.
- "Norethisterone and norethisterone acetate". IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. 21: 441–60. December 1979. PMID 120838.
- Stanczyk FZ, Roy S (July 1990). "Metabolism of levonorgestrel, norethindrone, and structurally related contraceptive steroids". Contraception. 42 (1): 67–96. PMID 2143719.
- Wiseman LR, McTavish D (March 1994). "Transdermal estradiol/norethisterone. A review of its pharmacological properties and clinical use in postmenopausal women". Drugs & Aging. 4 (3): 238–56. S2CID 68007924.
- Taitel HF, Kafrissen ME (1995). "Norethindrone--a review of therapeutic applications". International Journal of Fertility and Menopausal Studies. 40 (4): 207–23. PMID 8520623.
- Maier WE, Herman JR (August 2001). "Pharmacology and toxicology of ethinyl estradiol and norethindrone acetate in experimental animals". Regulatory Toxicology and Pharmacology. 34 (1): 53–61. PMID 11502156.
- Riis BJ, Lehmann HJ, Christiansen C (October 2002). "Norethisterone acetate in combination with estrogen: effects on the skeleton and other organs. A review". American Journal of Obstetrics and Gynecology. 187 (4): 1101–16. PMID 12389012.
- Draper BH, Morroni C, Hoffman M, Smit J, Beksinska M, Hapgood J, Van der Merwe L (July 2006). "Depot medroxyprogesterone versus norethisterone oenanthate for long-acting progestogenic contraception". The Cochrane Database of Systematic Reviews (3): CD005214. PMID 16856087.
- Kuhl H, Wiegratz I (August 2007). "Can 19-nortestosterone derivatives be aromatized in the liver of adult humans? Are there clinical implications?". Climacteric. 10 (4): 344–53. S2CID 20759583.
- Casey CL, Murray CA (2008). "HT update: spotlight on estradiol/norethindrone acetate combination therapy". Clinical Interventions in Aging. 3 (1): 9–16. PMID 18488874.
- Paulen ME, Curtis KM (October 2009). "When can a woman have repeat progestogen-only injectables--depot medroxyprogesterone acetate or norethisterone enantate?". Contraception. 80 (4): 391–408. PMID 19751863.
- Chwalisz K, Surrey E, Stanczyk FZ (June 2012). "The hormonal profile of norethindrone acetate: rationale for add-back therapy with gonadotropin-releasing hormone agonists in women with endometriosis". Reproductive Sciences. 19 (6): 563–71. S2CID 2882899.

![Norethisterone and ethinylestradiol levels over 24 hours after a single oral dose of 10 mg NETA in postmenopausal women.[38]](http://upload.wikimedia.org/wikipedia/commons/thumb/e/e5/Norethisterone_and_ethinylestradiol_levels_after_a_single_oral_dose_of_10_mg_norethisterone_acetate_in_postmenopausal_women.png/450px-Norethisterone_and_ethinylestradiol_levels_after_a_single_oral_dose_of_10_mg_norethisterone_acetate_in_postmenopausal_women.png)
![Norethisterone and ethinylestradiol levels over 8 weeks after a single intramuscular injection of 200 mg NETE in premenopausal women.[86]](http://upload.wikimedia.org/wikipedia/commons/thumb/d/df/Norethisterone_and_ethinylestradiol_levels_after_a_single_intramuscular_injection_of_200_mg_norethisterone_enanthate_in_premenopausal_women.png/450px-Norethisterone_and_ethinylestradiol_levels_after_a_single_intramuscular_injection_of_200_mg_norethisterone_enanthate_in_premenopausal_women.png)
