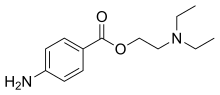
Procaine
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | Parenteral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Metabolism | Hydrolysis by plasma esterases |
| Elimination half-life | 40–84 seconds |
| Excretion | Renal |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Procaine is a local anesthetic drug of the amino ester group. It is most commonly used in dental procedures to numb the area around a tooth[1] and is also used to reduce the pain of intramuscular injection of penicillin. Owing to the ubiquity of the trade name Novocain or Novocaine, in some regions, procaine is referred to generically as novocaine. It acts mainly as a sodium channel blocker.[2] Today, it is used therapeutically in some countries due to its sympatholytic, anti-inflammatory, perfusion-enhancing, and mood-enhancing effects.[3]
Procaine was first synthesized in 1905,[4] shortly after amylocaine.[5] It was created by the chemist Alfred Einhorn who gave the chemical the trade name Novocaine, from the Latin nov- (meaning "new") and -caine, a common ending for alkaloids used as anesthetics. It was introduced into medical use by surgeon Heinrich Braun.
Prior to the discovery of amylocaine and procaine, cocaine was a commonly used local anesthetic.[6] Einhorn wished his new discovery to be used for amputations, but for this surgeons preferred general anesthesia. Dentists, however, found it very useful.[7]
Pharmacology

The primary use for procaine is as an anaesthetic.
Aside from its use as a dental anesthetic, procaine is used less frequently today, since more effective (and
Procaine, an
.A 1% procaine injection has been recommended for the treatment of extravasation complications associated with venipuncture, steroids, and antibiotics. It has likewise been recommended for treatment of inadvertent intra-arterial injections (10 ml of 1% procaine), as it helps relieve pain and vascular spasm.
Procaine is an occasional additive in illicit street drugs, such as cocaine. MDMA manufacturers also use procaine as an additive at ratios ranging from 1:1 up to 10% MDMA with 90% procaine, which can be life-threatening.[9]
Adverse effects
Application of procaine leads to the depression of neuronal activity. The depression causes the nervous system to become hypersensitive, producing restlessness and shaking, leading to minor to severe convulsions.[
Procaine can also cause allergic reactions causing individuals to have problems with breathing, rashes, and swelling. Allergic reactions to procaine are usually not in response to procaine itself, but to its metabolite PABA. Allergic reactions are in fact quite rare, estimated to have an incidence of 1 per 500,000 injections. About one in 3000 white North Americans is homozygous (i.e. has two copies of the abnormal gene) for the most common atypical form of the enzyme pseudocholinesterase,[12][13] and do not hydrolyze ester anesthetics such as procaine. This results in a prolonged period of high levels of the anesthetic in the blood and increased toxicity.
However, certain populations in the world such as the
Synthesis
Procaine can be synthesized in two ways.

- The first consists of the direct reaction of the 4-aminobenzoic acid ethyl ester with 2-diethylaminoethanolin the presence of sodium ethoxide.
- The second is by oxidizing 4-nitrotoluene to 4-nitrobenzoic acid, which is further reacted with thionyl chloride, the resulting acid chloride is then esterified with 2-diethylaminoethanol to give Nitrocaine. Finally, the nitro group is reduced by hydrogenation over Raney nickelcatalyst.
See also
References
- ^ a b "How long does numbness last after the dentist?". Medical News Today. May 22, 2018. Retrieved July 14, 2020.
- ^ "Procaine (DB00721)". DrugBank. 2009-06-23.
- .
- ISBN 0-08-040296-8.
- ^ Minard R (18 October 2006). "The Preparation of the Local Anesthetic, Benzocaine, by an Esterification Reaction" (PDF). Archived from the original (PDF) on 20 July 2011. Retrieved 10 March 2011.
Adapted from Introduction to Organic Laboratory Techniques: A Microscale Approach, Pavia, Lampman, Kriz & Engel, 1989.
- PMID 11895133.
- PMID 10272260.
- PMID 8250339.
- ^ "Procaine". ecstasydata.org.
- PMID 2622091.
- ^ "Novocain Official FDA information". drugs.com. August 2007.
- ISBN 978-0-7020-3145-8.
- ^ a b "Butyrylcholinesterase". OMIM. Retrieved 4 September 2015.
- .
- ^ Einhorn A Höchst Ag U.S. patent 812,554; DE 179627, "Verfahren zur Darstellung von p-Aminobenzoësäureaalkaminestern [Method for the prepareation of p-aminobenzoic acid alkamine esters]", published 1906-12-11, assigned to Höchst A.M.; DE 194748, "Verfahren zur Darstellung von p-Aminobenzoësäureaalkaminestern [Method for the prepareation of p-aminobenzoic acid alkamine esters]", published 1908-01-28, assigned to Höchst A.M., addendendum to DE 179627.
Further reading
- Hahn-Godeffroy JD (2007). "Wirkungen und Nebenwirkungen von Procain: Was ist gesichert?". Komplement Integr. Med. 2: 32–4.
