Nozaki–Hiyama–Kishi reaction
| Nozaki-Hiyama-Kishi reaction | |
|---|---|
| Named after | Hitoshi Nozaki Tamejiro Hiyama Yoshito Kishi |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | nozaki-hiyama-coupling |
| RSC ontology ID | RXNO:0000191 |
The Nozaki–Hiyama–Kishi reaction is a
:Compared to
Enals give exclusively 1,2-addition. Solvents of choice are DMF and DMSO, one solvent requirement is solubility of the chromium salts. Nozaki–Hiyama–Kishi reaction is a useful method for preparing medium-size rings.[3]
In 1983 the scope was extended by the same authors to include
co-catalyst.[6]
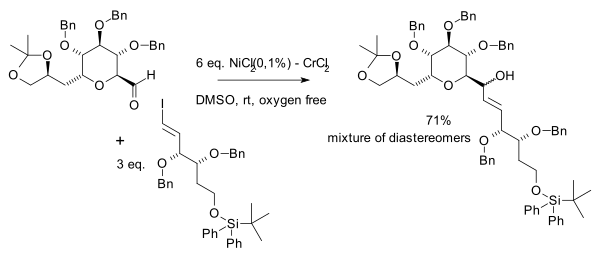
In the same year Yoshito Kishi et al. independently discovered the beneficial effects of nickel in his quest for palytoxin:[7]
Palladium acetate
was also found to be an effective cocatalyst.
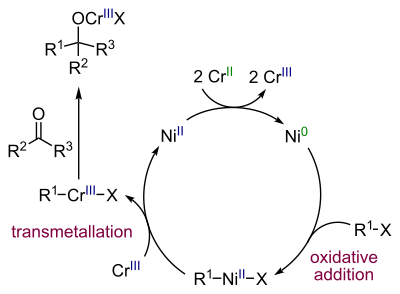
Reaction mechanism
Nickel is the actual
carbonyl group in a nucleophilic addition
.
The amount of nickel used should be low as a direct alkene coupling to a diene is a side reaction.[8]
Related reactions are the Grignard reaction (magnesium), the Barbier reaction (zinc) and addition reactions involving organolithium reagents.
References
- .
- ^ Kazuhiko Takai, Koichi Sakogawa, Yasutaka Kataoka, Koichiro Oshima, and Kiitiro Utimoto (1998). "Preparation and reactions of alkenylchromium reagents: 2-Hexyl-5-phenyl-1-penten-3-ol". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 9, p. 472.