Nuclear pore
| Nuclear Pore | |
|---|---|
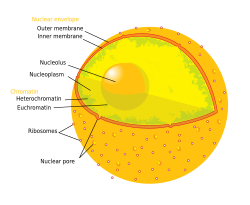 Diagram of the human cell nucleus with nuclear pores. | |
 Schematic diagram of a nuclear pore complex within the nuclear envelope (1) with the outer ring (2), spokes (3), basket (4), and filaments (5). | |
| Details | |
| Identifiers | |
| Latin | porus nuclearis |
| MeSH | D022022 |
| TH | H1.00.01.2.01005 |
| FMA | 63148 |
| Anatomical terminology | |
A nuclear pore is a channel as part of the nuclear pore complex (NPC), a large
The nuclear pore complex predominantly consists of proteins known as
The principal function of nuclear pore complexes is to facilitate selective membrane transport of various molecules across the nuclear envelope. This includes the transportation of
Evolutionary
The count of nuclear pore complexes varies across cell types and different stages of the cell's life cycle, with approximately 1,000 NPCs typically found in vertebrate cells.
Nuclear Pore Complex: Size and Composition
The nuclear pore complex (NPC) is a crucial cellular structure with a diameter of approximately 120 nanometers in vertebrates. Its channel varies from 5.2 nanometers in humans
Transport through the nuclear pore complex

Nuclear pore complex (NPC) serves highly regulated gateway for the transport of molecules between the nucleus and the cytoplasm. This intricate system enables the selective passage for molecules including proteins, RNA, and signaling molecules, ensuring proper cellular function and homeostasis. Small molecules such as proteins water and ions can diffuse through NPCs, but cargoes (>40
The largest family of
Three models have been suggested to explain the translocation mechanism:
- Affinity gradients along the central plug
- Brownian affinity gating
- Selective phase
Import of proteins
Nuclear proteins are synthesized in the cytoplasm and need to be imported through the NPCs into the nucleus. Import can be directed by various signals, of which nuclear localization signal (NLS) are best characterized.[14] Several NLS sequences are known, generally containing a conserved sequence with basic residues such as PKKKRKV. Any material with an NLS will be taken up by importins to the nucleus.
Importation begins with Importin-α binding to the NLS sequence of cargo proteins, forming a complex. Importin-β then attaches to Importin-α, facilitating transport towards the NPC.
As the complex reaches the NPC, it diffuses through the pore without the need for additional energy. Upon entry into nucleus, RanGTP binds to Importin-β and displaces it from the complex. Then the cellular apoptosis susceptibility protein (CAS), an exportin which in the nucleus is bound to RanGTP, displaces Importin-α from the cargo. The NLS-protein is thus free in the nucleoplasm. The Importinβ-RanGTP and Importinα-CAS-RanGTP complex diffuses back to the cytoplasm where GTPs are hydrolyzed to GDP leading to the release of Importinβ and Importinα which become available for a new NLS-protein import round.
While translocation through the NPC is not energy-dependent, the overall import cycle needs the hydrolysis of two GTPs molecules, making it an active transport process. The import cycle is powered by the nucleo-cytoplasmic RanGTP gradient. This gradient arises from the exclusive nuclear localization of RanGEFs, proteins that exchange GDP to GTP on Ran molecules. Thus, there is an elevated RanGTP concentration in the nucleus compared to the cytoplasm.
Export of proteins
In addition to nuclear import, certain molecules and macromolecular complexes, such as ribosome subunits and messenger RNAs, require export from the nucleus to the cytoplasm. This export process mirrors the import mechanism in complexity and importance.
In a classical export scenario, proteins with a nuclear export sequence (NES) form a
Export of RNA
Different export pathways through the NPC for various RNA classes.
In highest eukaryotes, mRNA export is believed to be spicling-dependent. Splicing recruits the TREX protein complex to spliced messages, serving as an adapter for TAP, a low-affinity RNA-binding protein However, there are alternative mRNA export pathways that do not rely on splicing for specialized messages such as histones. Recent work also suggest an interplay between splicing-dependent export and one of these alternative mRNA export pathways for secretory and mitochondrial transcripts.[15]
Assembly of the NPC
Since the NPC regulates genome access, its presence in significant quantities during cell cycle stages characterized by high transcription rates is crucial. For example, cycling mammalian and yeast cells double the amount of NPC in the nucleus between the G1 and G2 phase. Similarly, oocytes accumulate abundant NPCs in anticipation of the rapid mitotic activity during early development. Moreover, interphase cells must maintain NPC generation to sustain consistent NPC levels, as some may incur damage. Furthermore, certain cells can even increase the NPC numbers due to increased transcriptional demand.[16]
Theories of assembly
There are several theories as to how NPCs are assembled. As the immunodepletion of certain protein complexes, such as the Nup 107–160 complex, leads to the formation of poreless nuclei, it seems likely that the Nup complexes are involved in fusing the outer membrane of the nuclear envelope with the inner and not that the fusing of the membrane begins the formation of the pore. There are several ways that this could lead to the formation of the full NPC.
- One possibility is that as a protein complex it binds to the chromatin. It is then inserted into the double membrane close to the chromatin. This, in turn, leads to the fusing of that membrane. Around this protein complex others eventually bind forming the NPC. This method is possible during every phase of mitosis as the double membrane is present around the chromatin before the membrane fusion proteins complex can insert. Post mitotic cells could form a membrane first with pores being inserted into after formation.
- Another model for the formation of the NPC is the production of a prepore as a start as opposed to a single protein complex. This prepore would form when several Nup complexes come together and bind to the chromatin. This would have the double membrane form around it in during mitotic reassembly. Possible prepore structures have been observed on chromatin before nuclear envelope (NE) formation using electron microscopy.[17] During the interphase of the cell cycle the formation of the prepore would happen within the nucleus, each component being transported in through existing NPCs. These Nups would bind to an importin, once formed, preventing the assembly of a prepore in the cytoplasm. Once transported into the nucleus Ran GTP would bind to the importin and cause it to release the cargo. This Nup would be free to form a prepore. The binding of importins has at least been shown to bring Nup 107 and the Nup 153 nucleoporins into the nucleus.[16] NPC assembly is a very rapid process yet defined intermediate states occur which leads to the idea that this assembly occurs in a stepwise fashion.[18]
Disassembly
During mitosis the NPC appears to disassemble in stages, except in lower eukaryotes like yeast, where NPC disassembly does not happen during mitosis.[19] Peripheral nucleoporins such as the Nup 153 Nup 98 and Nup 214 disassociate from the NPC. The rest, which can be considered a scaffold proteins remain stable, as cylindrical ring complexes within the nuclear envelope. This disassembly of the NPC peripheral groups is largely thought to be phosphate driven, as several of these nucleoporins are phosphorylated during the stages of mitosis. However, the enzyme involved in the phosphorylation is unknown in vivo. In metazoans (which undergo open mitosis) the NE degrades quickly after the loss of the peripheral Nups. The reason for this may be due to the change in the NPC's architecture. This change may make the NPC more permeable to enzymes involved in the degradation of the NE such as cytoplasmic tubulin, as well as allowing the entry of key mitotic regulator proteins. In organisms that undergo a semi-open mitosis such as the filamentous fungus Aspergillus nidulans, 14 out of the 30 nucleoporins disassemble from the core scaffold structure, driven by the activation of the NIMA and Cdk1 kinases that phosphorylate nucleoporins and open nuclear pores[20][21] thereby widening the nuclear pore and allowing the entry of mitotic regulators.[22]
Preservation of integrity
In fungi undergoing closed mitosis, where the nucleus remains intact, changes in the permeability barrier of the nuclear envelope (NE) are attributed to alterations within the NPC. These changes facilitate the entry of mitotic regulators into the nucleus. Studies in Aspergillys nidulans suggest that the NPC composition appears to be effeveted by the mitotiv kinase NIMA. NIMA potentially phosphorylates nucleoporins Nup98 and Gle2/Rae1, leading to NPC remodeling.[23] This remodeling allows the nuclear entry of the protein complex cdc2/cyclinB and various other proteins, including soluble tubulin. The NPC scaffold remains intact throughout the whole closed mitosis. This seems to preserver the integrity of NE.
References
- ^ Lin, D. H., Stuwe, T., Schilbach, S., Rundlet, E. J., Perriches, T., Mobbs, G., ... Hoelz, A. (2016). Architecture of the nuclear pore complex symmetric core. Science, 352(6283), aaf1015. http://doi.org/10.1126/science.aaf1015
- PMID 31001417.
- PMID 34995711.
- PMID 26541386.
- PMID 24066078.
- PMID 11574060.
- PMID 25691464.
- PMID 19680228.
- PMID 21326819.
- PMID 34294532.
- PMID 9023189.
- ^ "Nuclear Pore Complex - an overview | ScienceDirect Topics". www.sciencedirect.com. Retrieved February 15, 2024.
- ^ "Karyopherin - an overview | ScienceDirect Topics". www.sciencedirect.com. Retrieved February 15, 2024.
- PMID 9159081.
- PMID 21533221.
- ^ PMID 15145357.
- PMID 3339085.
- PMID 11707513. Archived from the originalon September 13, 2019. Retrieved November 4, 2008.
- PMID 30943044.
- PMID 25540430.
- S2CID 14782686.
- PMID 17660363.
- PMID 19019988.
External links
- Histology image: 20104loa – Histology Learning System at Boston University
- Nuclear+pore at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Nuclear Pore Complex animations Archived February 7, 2009, at the Wayback Machine
- Nuclear Pore Complex illustrations Archived February 7, 2009, at the Wayback Machine
- 3D electron microscopy structures of the NPC and constituent proteins from the EM Data Bank(EMDB)
- NCDIR - National Center for the Dynamic Interactome[permanent dead link]
