Nuclear transmutation
Nuclear transmutation is the conversion of one
A transmutation can be achieved either by nuclear reactions (in which an outside particle reacts with a nucleus) or by radioactive decay, where no outside cause is needed.
Natural transmutation by stellar nucleosynthesis in the past created most of the heavier chemical elements in the known existing universe, and continues to take place to this day, creating the vast majority of the most common elements in the universe, including helium, oxygen and carbon. Most stars carry out transmutation through fusion reactions involving hydrogen and helium, while much larger stars are also capable of fusing heavier elements up to iron late in their evolution.
Elements heavier than iron, such as gold or lead, are created through elemental transmutations that can naturally occur in supernovae. One goal of alchemy, the transmutation of base substances into gold, is now known to be impossible by chemical means but possible by physical means. As stars begin to fuse heavier elements, substantially less energy is released from each fusion reaction. This continues until it reaches iron which is produced by an endothermic reaction consuming energy. No heavier element can be produced in such conditions.
One type of natural transmutation observable in the present occurs when certain
Artificial transmutation may occur in machinery that has enough energy to cause changes in the nuclear structure of the elements. Such machines include particle accelerators and tokamak reactors. Conventional fission power reactors also cause artificial transmutation, not from the power of the machine, but by exposing elements to neutrons produced by fission from an artificially produced nuclear chain reaction. For instance, when a uranium atom is bombarded with slow neutrons, fission takes place. This releases, on average, three neutrons and a large amount of energy. The released neutrons then cause fission of other uranium atoms, until all of the available uranium is exhausted. This is called a chain reaction.
Artificial nuclear transmutation has been considered as a possible mechanism for reducing the volume and hazard of radioactive waste.[2]
History
Alchemy
The term transmutation dates back to alchemy. Alchemists pursued the philosopher's stone, capable of chrysopoeia – the transformation of base metals into gold.[3] While alchemists often understood chrysopoeia as a metaphor for a mystical or religious process, some practitioners adopted a literal interpretation and tried to make gold through physical experimentation. The impossibility of the metallic transmutation had been debated amongst alchemists, philosophers and scientists since the Middle Ages. Pseudo-alchemical transmutation was outlawed[4] and publicly mocked beginning in the fourteenth century. Alchemists like Michael Maier and Heinrich Khunrath wrote tracts exposing fraudulent claims of gold making. By the 1720s, there were no longer any respectable figures pursuing the physical transmutation of substances into gold.[5] Antoine Lavoisier, in the 18th century, replaced the alchemical theory of elements with the modern theory of chemical elements, and John Dalton further developed the notion of atoms (from the alchemical theory of corpuscles) to explain various chemical processes. The disintegration of atoms is a distinct process involving much greater energies than could be achieved by alchemists.
Modern physics
It was first consciously applied to modern physics by Frederick Soddy when he, along with Ernest Rutherford in 1901, discovered that radioactive thorium was converting itself into radium. At the moment of realization, Soddy later recalled, he shouted out: "Rutherford, this is transmutation!" Rutherford snapped back, "For Christ's sake, Soddy, don't call it transmutation. They'll have our heads off as alchemists."[6]
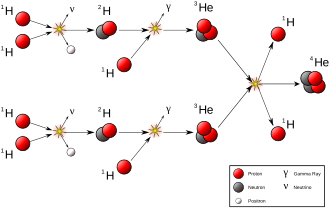
Rutherford and Soddy were observing natural transmutation as a part of
Later in the twentieth century the transmutation of elements within stars was elaborated, accounting for the relative abundance of heavier elements in the universe. Save for the first five elements, which were produced in the Big Bang and other
Transmutation of other elements into gold
The alchemical tradition sought to turn the "base metal", lead, into gold. As a nuclear transmutation, it requires far less energy to turn gold into lead; for example, this would occur via neutron capture and beta decay if gold were left in a nuclear reactor for a sufficiently long period of time.[citation needed] Glenn Seaborg succeeded in producing a minuscule amount of gold from bismuth, at a net energy loss.[11][12]
Transmutation in the universe
The Big Bang is thought to be the origin of the hydrogen (including all deuterium) and helium in the universe. Hydrogen and helium together account for 98% of the mass of ordinary matter in the universe, while the other 2% makes up everything else. The Big Bang also produced small amounts of lithium, beryllium and perhaps boron. More lithium, beryllium and boron were produced later, in a natural nuclear reaction, cosmic ray spallation.
The Solar System is thought to have condensed approximately 4.6 billion years before the present, from a cloud of hydrogen and helium containing heavier elements in dust grains formed previously by a large number of such stars. These grains contained the heavier elements formed by transmutation earlier in the history of the universe.
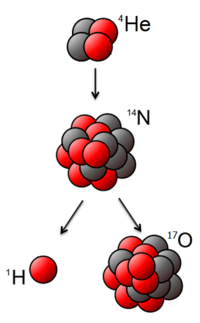
All of these natural processes of transmutation in stars are continuing today, in our own galaxy and in others. Stars fuse hydrogen and helium into heavier and heavier elements (up to iron), producing energy. For example, the observed light curves of supernova stars such as
Artificial transmutation of nuclear waste
Overview
Transmutation of
Ceramic targets containing actinides can be bombarded with neutrons to induce transmutation reactions to remove the most difficult long-lived species. These can consist of actinide-containing solid solutions such as (Am,Zr)N, (Am,Y)N, (Zr,Cm)O2, (Zr,Cm,Am)O2, (Zr,Am,Y)O2 or just actinide phases such as AmO2, NpO2, NpN, AmN mixed with some inert phases such as MgO, MgAl2O4, (Zr,Y)O2, TiN and ZrN. The role of non-radioactive inert phases is mainly to provide stable mechanical behaviour to the target under neutron irradiation.[13]
There are issues with this P&T (partitioning and transmutation) strategy however:
- it is limited by the costly and cumbersome need to separate long-lived fission product isotopes before they can undergo transmutation.
- some neutron cross-sectionand resultingly low capture rate.
The new study led by Satoshi Chiba at Tokyo Tech (called "Method to Reduce Long-lived Fission Products by Nuclear Transmutations with Fast Spectrum Reactors"[14]) shows that effective transmutation of long-lived fission products can be achieved in fast spectrum reactors without the need for isotope separation. This can be achieved by adding a yttrium deuteride moderator.[15]
Reactor types
For instance, plutonium can be reprocessed into
Fuel types
There are several fuels that can incorporate plutonium in their initial composition at their beginning of cycle and have a smaller amount of this element at the end of cycle. During the cycle, plutonium can be burnt in a power reactor, generating electricity. This process is not only interesting from a power generation standpoint, but also due to its capability of consuming the surplus
Fuels with plutonium and thorium are also an option. In these, the neutrons released in the fission of plutonium are captured by thorium-232. After this radiative capture, thorium-232 becomes thorium-233, which undergoes two beta minus decays resulting in the production of the fissile isotope uranium-233. The radiative capture cross section for thorium-232 is more than three times that of uranium-238, yielding a higher conversion to fissile fuel than that from uranium-238. Due to the absence of uranium in the fuel, there is no second generation plutonium produced, and the amount of plutonium burnt will be higher than in mixed oxide fuels. However, uranium-233, which is fissile, will be present in the used nuclear fuel. Weapons-grade and reactor-grade plutonium can be used in plutonium–thorium fuels, with weapons-grade plutonium being the one that shows a bigger reduction in the amount of plutonium-239.
Long-lived fission products
| Nuclide | t1⁄2
|
Yield | Q[a 1] | βγ
|
|---|---|---|---|---|
| ( Ma )
|
(%)[a 2] | ( keV )
|
||
| 99Tc | 0.211 | 6.1385 | 294 | β |
126Sn
|
0.230 | 0.1084 | 4050[a 3] | βγ |
| 79Se | 0.327 | 0.0447 | 151 | β |
135Cs
|
1.33 | 6.9110[a 4] | 269 | β |
93Zr
|
1.53 | 5.4575 | 91 | βγ |
107Pd
|
6.5 | 1.2499 | 33 | β |
| 129I | 15.7 | 0.8410 | 194 | βγ |
Some radioactive fission products can be converted into shorter-lived radioisotopes by transmutation. Transmutation of all fission products with half-life greater than one year is studied in Grenoble,[19] with varying results.
The next longer-lived fission product is
Sm in nuclear waste would require separation from other isotopes of samarium
Sm is less dangerous than 90
Sr and 137
Cs and can also be left to decay for ~970 years.
Finally, there are seven long-lived fission products. They have much longer half-lives in the range 211,000 years to 15.7 million years. Two of them, technetium-99 and iodine-129, are mobile enough in the environment to be potential dangers, are free (Technetium has no known stable isotopes) or mostly free of mixture with stable isotopes of the same element, and have neutron cross sections that are small but adequate to support transmutation.
Additionally, 99
Tc can substitute for uranium-238 in supplying Doppler broadening for negative feedback for reactor stability.[20]
Most studies of proposed transmutation schemes have assumed 99
Tc, 129
I, and transuranium elements as the targets for transmutation, with other fission products,
Tc (the result of 99
Tc capturing a neutron) decaying with a relatively short half life to a stable isotope of ruthenium, a precious metal
Of the remaining five long-lived fission products,
Zr could be reused for new cladding material has not been subject of much study thus far.
See also
- Neutron activation
- Nuclear power
- List of nuclear waste treatment technologies
- Synthesis of precious metals
- Fertile material
References
- .
- ^ http://www.oecd-nea.org/trw/ "Transmutation of Radioactive Waste." Nuclear Energy Agency. Feb 3rd 2012.
- ^ "Alchemy", Dictionary.com
- ^ John Hines, II, R. F. Yeager. John Gower, Trilingual Poet: Language, Translation, and Tradition. Boydell & Brewer. 2010. p.170
- ^ Lawrence Principe. New Narratives in Eighteenth-Century Chemistry. Springer. 2007. p.8
- ^ Muriel Howorth, Pioneer Research on the Atom: The Life Story of Frederick Soddy, New World, London 1958, pp 83-84; Lawrence Badash, Radium, Radioactivity and the Popularity of Scientific Discovery, Proceedings of the American Philosophical Society 122,1978: 145-54; Thaddeus J. Trenn, The Self-Splitting Atom: The History of the Rutherford-Soddy Collaboration, Taylor & Francis, London, 1977, pp 42, 58-60, 111-17.
- ^ "Rutherford's Nuclear World: The Story of the Discovery of the Nucleus | Sections | American Institute of Physics".
- ^ Cockcroft and Walton split lithium with high energy protons April 1932. Archived 2012-09-02 at the Wayback Machine
- . Retrieved 20 June 2022.
- ^ William Alfred Fowler, Margaret Burbidge, Geoffrey Burbidge, and Fred Hoyle, 'Synthesis of the Elements in Stars', Reviews of Modern Physics, vol. 29, Issue 4, pp. 547–650
- ^ Aleklett, K.; Morrissey, D.; Loveland, W.; McGaughey, P.; Seaborg, G. (1981). "Energy dependence of 209Bi fragmentation in relativistic nuclear collisions". .
- ^ Matthews, Robert (December 2, 2001). "The Philosopher's Stone". The Daily Telegraph. Archived from the original on July 23, 2013. Retrieved July 23, 2013.
- ^ "Crystalline Materials for Actinide Immobilisation". London: Imperial College Press. 2010. p. 198. Archived from the original on 9 March 2012.
- PMID 29066843.
- ^ A fast reactor system to shorten the lifetime of long-lived fission products
- ^ Rita Plukiene, Evolution Of Transuranium Isotopic Composition In Power Reactors And Innovative Nuclear Systems For Transmutation Archived 2007-09-27 at the Wayback Machine, PhD Thesis, Vytautas Magnus University, 2003, retrieved January 2008
- ^ Takibayev A., Saito M., Artisyuk V., and Sagara H., 'Fusion-driven transmutation of selected long-lived fission products', Progress in nuclear energy, Vol. 47, 2005, retrieved January 2008.
- ^ Transmutation of Transuranic Elements and Long Lived Fission Products in Fusion Devices, Y. Gohar, Argonne National Laboratory
- ^ Method for net decrease of hazardous radioactive nuclear waste materials - US Patent 4721596 Description
- ^ Transmutation of Selected Fission Products in a Fast Reactor
- ^ The Nuclear Alchemy Gamble – Institute for Energy and Environmental Research
External links
- "Radioactive change", Rutherford & Soddy article (1903), online and analyzed on Bibnum [click 'à télécharger' for English version].
