Ocean acidification

Ocean acidification is the ongoing decrease in the
A change in pH by 0.1 represents a 26% increase in hydrogen ion concentration in the world's oceans (the pH scale is logarithmic, so a change of one in pH units is equivalent to a tenfold change in hydrogen ion concentration). Sea-surface pH and carbonate saturation states vary depending on ocean depth and location. Colder and higher latitude waters are capable of absorbing more CO2. This can cause acidity to rise, lowering the pH and carbonate saturation levels in these areas. Other factors that influence the atmosphere-ocean CO2 exchange, and thus local ocean acidification, include: ocean currents and upwelling zones, proximity to large continental rivers, sea ice coverage, and atmospheric exchange with nitrogen and sulfur from fossil fuel burning and agriculture.[4][5][6]
Decreased ocean pH has a range of potentially harmful effects for marine organisms. These include reduced calcification, depressed metabolic rates, lowered immune responses, and reduced energy for basic functions such as reproduction.
The United Nations Sustainable Development Goal 14 ("Life below Water") has a target to "minimize and address the impacts of ocean acidification".[10] Reducing carbon dioxide emissions (i.e., climate change mitigation measures) is the only solution that addresses the root cause of ocean acidification. Mitigation measures which achieve carbon dioxide removal from the atmosphere would help to reverse ocean acidification. The more specific ocean-based mitigation methods (e.g. ocean alkalinity enhancement, enhanced weathering) could also reduce ocean acidification. These strategies are being researched, but generally have a low technology readiness level and many risks.[11][12][13]
Ocean acidification has occurred previously in Earth's history.[14] The resulting ecological collapse in the oceans had long-lasting effects on the global carbon cycle and climate.
Cause
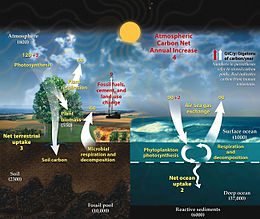
Present-day (2021) atmospheric carbon dioxide (CO2) levels of around 415 ppm are around 50% higher than preindustrial concentrations.[16] The current elevated levels and rapid growth rates are unprecedented in the past 55 million years of the geological record. The sources of this excess CO2 are clearly established as human driven: they include anthropogenic fossil fuel, industrial, and land-use/land-change emissions. The ocean acts as a carbon sink for anthropogenic CO2 and takes up roughly a quarter of total anthropogenic CO2 emissions.[17] However, the additional CO2 in the ocean results in a wholesale shift in seawater acid-base chemistry toward more acidic, lower pH conditions and lower saturation states for carbonate minerals used in many marine organism shells and skeletons.[17]
Accumulated since 1850, the ocean sink holds up to 175 ± 35 gigatons of carbon, with more than two-thirds of this amount (120 GtC) being taken up by the global ocean since 1960. Over the historical period, the ocean sink increased in pace with the exponential anthropogenic emissions increase. From 1850 until 2022, the ocean has absorbed 26 % of total anthropogenic emissions.[16] Emissions during the period 1850–2021 amounted to 670 ± 65 gigatons of carbon and were partitioned among the atmosphere (41 %), ocean (26 %), and land (31 %).[16]
The carbon cycle describes the fluxes of carbon dioxide (CO
2) between the oceans, terrestrial biosphere, lithosphere,[18] and atmosphere. The carbon cycle involves both organic compounds such as cellulose and inorganic carbon compounds such as carbon dioxide, carbonate ion, and bicarbonate ion, together referenced as dissolved inorganic carbon (DIC). These inorganic compounds are particularly significant in ocean acidification, as they include many forms of dissolved CO
2 present in the Earth's oceans.[19]
When CO
2 dissolves, it reacts with water to form a balance of ionic and non-ionic chemical species: dissolved free carbon dioxide (CO
2(aq)), carbonic acid (H
2CO
3), bicarbonate (HCO−
3) and carbonate (CO2−
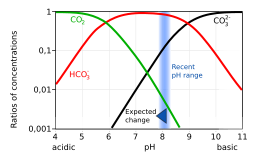
3). The ratio of these species depends on factors such as seawater temperature, pressure and salinity (as shown in a Bjerrum plot). These different forms of dissolved inorganic carbon are transferred from an ocean's surface to its interior by the ocean's solubility pump. The resistance of an area of ocean to absorbing atmospheric CO
2 is known as the Revelle factor.
Main effects
The ocean's chemistry is changing due to the uptake of anthropogenic carbon dioxide (CO2).[4][20]: 395 Ocean pH, carbonate ion concentrations ([CO32−]), and calcium carbonate mineral saturation states (Ω) have been declining as a result of the uptake of approximately 30% of the anthropogenic carbon dioxide emissions over the past 270 years (since around 1750). This process, commonly referred to as "ocean acidification", is making it harder for marine calcifiers to build a shell or skeletal structure, endangering coral reefs and the broader marine ecosystems.[4]
Ocean acidification has been called the "evil twin of
Reduction in pH value
Dissolving CO
2 in seawater increases the hydrogen ion (H+
) concentration in the ocean, and thus decreases ocean pH, as follows:[26]
In shallow coastal and shelf regions, a number of factors interplay to affect air-ocean CO2 exchange and resulting pH change.[27][28] These include biological processes, such as photosynthesis and respiration,[29] as well as water upwelling.[30] Also, ecosystem metabolism in freshwater sources reaching coastal waters can lead to large, but local, pH changes.[27]
Freshwater bodies also appear to be acidifying, although this is a more complex and less obvious phenomenon.[31][32]
The absorption of CO2 from the atmosphere does not affect the ocean's
Decreased calcification in marine organisms

Changes in ocean chemistry can have extensive direct and indirect effects on organisms and their habitats. One of the most important repercussions of increasing ocean acidity relates to the production of shells out of
Very little of the extra carbon dioxide that is added into the ocean remains as dissolved carbon dioxide. The majority dissociates into additional bicarbonate and free hydrogen ions. The increase in hydrogen is larger than the increase in bicarbonate,[35] creating an imbalance in the reaction:
- HCO−3 ⇌ CO2−3 + H+
To maintain chemical equilibrium, some of the carbonate ions already in the ocean combine with some of the hydrogen ions to make further bicarbonate. Thus the ocean's concentration of carbonate ions is reduced, removing an essential building block for marine organisms to build shells, or calcify:
- Ca2+ + CO2−3 ⇌ CaCO3
The increase in concentrations of dissolved carbon dioxide and bicarbonate, and reduction in carbonate, are shown in the Bjerrum plot.
Decrease in saturation state
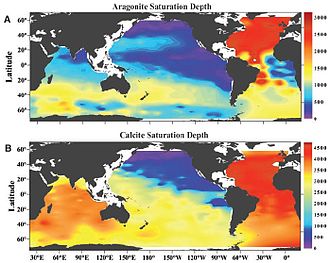
The
Here Ω is the product of the concentrations (or
3 does not readily dissolve. Most calcifying organisms live in such waters.[3] Below this depth, Ω has a value less than 1, and CaCO
3 will dissolve. The carbonate compensation depth is the ocean depth at which carbonate dissolution balances the supply of carbonate to sea floor, therefore sediment below this depth will be void of calcium carbonate.[38]
3 therefore increasing CaCO
3 dissolution.
Calcium carbonate most commonly occurs in two common
3 is directly proportional to its saturation state and calcifying organisms exhibit stress in waters with lower saturation states.[40]
Natural variability and climate feedbacks
Already now large quantities of water undersaturated in
At depths of 1000s of meters in the ocean, calcium carbonate shells begin to dissolve as increasing pressure and decreasing temperature shift the chemical equilibria controlling calcium carbonate precipitation.[42] The depth at which this occurs is known as the carbonate compensation depth. Ocean acidification will increase such dissolution and shallow the carbonate compensation depth on timescales of tens to hundreds of years.[42] Zones of downwelling are being affected first.[43]
In the North Pacific and North Atlantic, saturation states are also decreasing (the depth of saturation is getting more shallow).[20]: 396 Ocean acidification is progressing in the open ocean as the CO2 travels to deeper depth as a result of ocean mixing. In the open ocean, this causes carbonate compensation depths to become more shallow, meaning that dissolution of calcium carbonate will occur below those depths. In the North Pacific these carbonate saturations depths are shallowing at a rate of 1–2 m per year.[20]: 396
It is expected that ocean acidification in the future will lead to a significant decrease in the burial of carbonate sediments for several centuries, and even the dissolution of existing carbonate sediments.[44]
Measured and estimated values
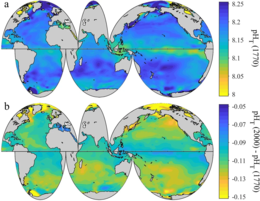
Present day and recent history
2 levels between the 1700s and the 1990s, from the Global Ocean Data Analysis Project (GLODAP) and the World Ocean Atlas
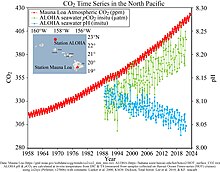
Between 1950 and 2020, the average pH value of the ocean surface is estimated to have decreased from approximately 8.15 to 8.05.[2] This represents an increase of around 26% in hydrogen ion concentration in the world's oceans (the pH scale is logarithmic, so a change of one in pH unit is equivalent to a tenfold change in hydrogen ion concentration).[47] For example, in the 15-year period 1995–2010 alone, acidity has increased 6 percent in the upper 100 meters of the Pacific Ocean from Hawaii to Alaska.[48]
The IPCC Sixth Assessment Report in 2021 stated that "present-day surface pH values are unprecedented for at least 26,000 years and current rates of pH change are unprecedented since at least that time.[49]: 76 The pH value of the ocean interior has declined over the last 20–30 years everywhere in the global ocean.[49]: 76 The report also found that "pH in open ocean surface water has declined by about 0.017 to 0.027 pH units per decade since the late 1980s".[50]: 716
The rate of decline differs by region. This is due to complex interactions between different types of forcing mechanisms:[50]: 716 "In the tropical Pacific, its central and eastern upwelling zones exhibited a faster pH decline of minus 0.022 to minus 0.026 pH unit per decade." This is thought to be "due to increased upwelling of CO2-rich sub-surface waters in addition to anthropogenic CO2 uptake."[50]: 716 Some regions exhibited a slower acidification rate: a pH decline of minus 0.010 to minus 0.013 pH unit per decade has been observed in warm pools in the western tropical Pacific.[50]: 716
The rate at which ocean acidification will occur may be influenced by the rate of surface ocean warming, because warm waters will not absorb as much CO2.[51] Therefore, greater seawater warming could limit CO2 absorption and lead to a smaller change in pH for a given increase in CO2.[51] The difference in changes in temperature between basins is one of the main reasons for the differences in acidification rates in different localities.
Current rates of ocean acidification have been likened to the greenhouse event at the Paleocene–Eocene boundary (about 56 million years ago), when surface ocean temperatures rose by 5–6 degrees Celsius. In that event, surface ecosystems experienced a variety of impacts, but bottom-dwelling organisms in the deep ocean actually experienced a major extinction.[52] Currently, the rate of carbon addition to the atmosphere-ocean system is about ten times the rate that occurred at the Paleocene–Eocene boundary.[53]
Extensive observational systems are now in place or being built for monitoring seawater CO2 chemistry and acidification for both the global open ocean and some coastal systems.[17]
| Location | Change in pH units per decade |
Period | Data source | Year of publication |
|---|---|---|---|---|
| Iceland[54] | minus 0.024 | 1984–2009 | Direct measurements | 2009 |
| Drake Passage[55] | minus 0.018 | 2002–2012 | Direct measurements | 2012 |
| Canary (ESTOC)[56] | minus 0.017 | 1995–2004 | Direct measurements | 2010 |
| Hawaii (HOT)[57] | minus 0.019 | 1989–2007 | Direct measurements | 2009 |
| Bermuda (BATS)[58] | minus 0.017 | 1984–2012 | Direct measurements | 2012 |
| Coral Sea[59] | minus 0.002 | ~1700 – ~1990 | Proxy reconstruction | 2005 |
| Eastern Mediterranean[60] | minus 0.023 | 1964–2005 | Proxy reconstruction | 2016 |
| Station, region | Study period | pH change per decade | |
|---|---|---|---|
| Equatorial Pacific | TAO | 2004–2011 | −0.026 |
| Indian Ocean | IO-STPS | 1991–2011 | −0.027 |
| Mediterranean | Dyfamed (43.42°N, 7.87°E) | 1995–2011 | −0.03 |
North Atlantic
|
Iceland Sea (68°N, 12.67°W) | 1985–2008 1985–2010 |
−0.024 −0.014 |
| North Atlantic | Irminger Sea (64.3°N, 28°W) | 1983–2004 | −0.026 |
North Pacific
|
NP-STSS | 1991–2011 | −0.01 |
| Southern Ocean | PAL-LTER, west Antarctic Peninsula | 1993–2012 | +0.02 |
Geologic past
Ocean acidification has occurred previously in Earth's history.
Three of the big five mass extinction events in the geologic past were associated with a rapid increase in atmospheric carbon dioxide, probably due to volcanism and/or thermal dissociation of marine gas hydrates.[72] Elevated CO2 levels impacted biodiversity.[73] Decreased CaCO3 saturation due to seawater uptake of volcanogenic CO2 has been suggested as a possible kill mechanism during the marine mass extinction at the end of the Triassic.[74] The end-Triassic biotic crisis is still the most well-established example of a marine mass extinction due to ocean acidification, because (a) carbon isotope records suggest enhanced volcanic activity that decreased the carbonate sedimentation which reduced the carbonate compensation depth and the carbonate saturation state, and a marine extinction coincided precisely in the stratigraphic record,[70][69][75] and (b) there was pronounced selectivity of the extinction against organisms with thick aragonitic skeletons,[70][76][77] which is predicted from experimental studies.[78] Ocean acidification has also been suggested as a one cause of the end-Permian mass extinction[66][65] and the end-Cretaceous crisis.[71] Overall, multiple climatic stressors, including ocean acidification, was likely the cause of geologic extinction events.[72]
The most notable example of ocean acidification is the Paleocene-Eocene Thermal Maximum (PETM), which occurred approximately 56 million years ago when massive amounts of carbon entered the ocean and atmosphere, and led to the dissolution of carbonate sediments across many ocean basins.[73] Relatively new geochemical methods of testing for pH in the past indicate the pH dropped 0.3 units across the PETM.[79][80] One study that solves the marine carbonate system for saturation state shows that it may not change much over the PETM, suggesting the rate of carbon release at our best geological analogy was much slower than human-induced carbon emissions. However, stronger proxy methods to test for saturation state are needed to assess how much this pH change may have affected calcifying organisms.
Predicted future values
Importantly, the rate of change in ocean acidification is much higher than in the geological past. This faster change prevents organisms from gradually adapting, and prevents climate cycle feedbacks from kicking in to mitigate ocean acidification. Ocean acidification is now on a path to reach lower pH levels than at any other point in the last 300 million years.
The extent of further ocean chemistry changes, including ocean pH, will depend on climate change mitigation efforts taken by nations and their governments.[49] Different scenarios of projected socioeconomic global changes are modelled by using the Shared Socioeconomic Pathways (SSP) scenarios.
Under a very high emission scenario (SSP5-8.5), model projections estimate that surface ocean pH could decrease by as much as 0.44 units by the end of this century, compared to the end of the 19th century.[87]: 608 This would mean a pH as low as about 7.7, and represents a further increase in H+ concentrations of two to four times beyond the increase to date.
| Time period | Ocean surface pH value (approx.) |
|---|---|
| Pre-industrial (1850) | 8.17 |
| Current (2021) | 8.08 |
| Future (2100) with low emission scenario (SSP 1–2.6) | 8.0 |
| Future (2100) with very high emission scenario (SSP 5–8.5) | 7.7 |
Impacts on oceanic calcifying organisms


Complexity of research findings
The full ecological consequences of the changes in calcification due to ocean acidification are complex but it appears likely that many calcifying species will be adversely affected by ocean acidification.
Overall, all marine ecosystems on Earth will be exposed to changes in acidification and several other ocean biogeochemical changes.[90] Ocean acidification may force some organisms to reallocate resources away from productive endpoints in order to maintain calcification.[91] For example, the oyster Magallana gigas is recognized to experience metabolic changes alongside altered calcification rates due to energetic tradeoffs resulting from pH imbalances.[92]
Under normal conditions, calcite and aragonite are stable in surface waters since the carbonate ions are supersaturated with respect to seawater. However, as ocean pH falls, the concentration of carbonate ions also decreases. Calcium carbonate thus becomes undersaturated, and structures made of calcium carbonate are vulnerable to calcification stress and dissolution.[93] In particular, studies show that corals,[94][95] coccolithophores,[89][27][96] coralline algae,[97] foraminifera,[98] shellfish and pteropods[99] experience reduced calcification or enhanced dissolution when exposed to elevated CO2. Even with active marine conservation practices it may be impossible to bring back many previous shellfish populations.[100]
Some studies have found different responses to ocean acidification, with coccolithophore calcification and photosynthesis both increasing under elevated atmospheric pCO2,[101] and an equal decline in primary production and calcification in response to elevated CO2,[102] or the direction of the response varying between species.[103]
Similarly, the sea star, Pisaster ochraceus, shows enhanced growth in waters with increased acidity.[104]
Reduced calcification from ocean acidification may affect the ocean's biologically driven sequestration of

Coccolithophores
A coccolithophore is a
Corals
Warm water corals are clearly in decline, with losses of 50% over the last 30–50 years due to multiple threats from ocean warming, ocean acidification, pollution and physical damage from activities such as fishing, and these pressures are expected to intensify.[107][20]: 416
The fluid in the internal compartments (the coelenteron) where corals grow their exoskeleton is also extremely important for calcification growth. When the saturation state of aragonite in the external seawater is at ambient levels, the corals will grow their aragonite crystals rapidly in their internal compartments, hence their exoskeleton grows rapidly. If the saturation state of aragonite in the external seawater is lower than the ambient level, the corals have to work harder to maintain the right balance in the internal compartment. When that happens, the process of growing the crystals slows down, and this slows down the rate of how much their exoskeleton is growing. Depending on the aragonite saturation state in the surrounding water, the corals may halt growth because pumping aragonite into the internal compartment will not be energetically favorable.[108] Under the current progression of carbon emissions, around 70% of North Atlantic cold-water corals will be living in corrosive waters by 2050–60.[109]
Acidified conditions primarily reduce the coral's capacity to build dense exoskeletons, rather than affecting the linear extension of the exoskeleton. The density of some species of corals could be reduced by over 20% by the end of this century.[110]
An in situ experiment, conducted on a 400 m2 patch of the Great Barrier Reef, to decrease seawater CO2 level (raise pH) to near the preindustrial value showed a 7% increase in net calcification.[111] A similar experiment to raise in situ seawater CO2 level (lower pH) to a level expected soon after the 2050 found that net calcification decreased 34%.[112]
However, a field study of the coral reef in Queensland and Western Australia from 2007 to 2012 found that corals are more resistant to the environmental pH changes than previously thought, due to internal homeostasis regulation; this makes thermal change (marine heatwaves), which leads to coral bleaching, rather than acidification, the main factor for coral reef vulnerability due to climate change.[113]
Studies at carbon dioxide seep sites
In some places carbon dioxide bubbles out from the sea floor, locally changing the pH and other aspects of the chemistry of the seawater. Studies of these carbon dioxide seeps have documented a variety of responses by different organisms.[114] Coral reef communities located near carbon dioxide seeps are of particular interest because of the sensitivity of some corals species to acidification. In Papua New Guinea, declining pH caused by carbon dioxide seeps is associated with declines in coral species diversity.[115] However, in Palau carbon dioxide seeps are not associated with reduced species diversity of corals, although bioerosion of coral skeletons is much higher at low pH sites.
Pteropods and brittle stars
Pteropods and brittle stars both form the base of the Arctic food webs and are both seriously damaged from acidification. Pteropods shells dissolve with increasing acidification and the brittle stars lose muscle mass when re-growing appendages.[116] For pteropods to create shells they require aragonite which is produced through carbonate ions and dissolved calcium and strontium. Pteropods are severely affected because increasing acidification levels have steadily decreased the amount of water supersaturated with carbonate.[117] The degradation of organic matter in Arctic waters has amplified ocean acidification; some Arctic waters are already undersaturated with respect to aragonite.[118][119][120]
The brittle star's eggs die within a few days when exposed to expected conditions resulting from Arctic acidification.
Other impacts on ecosystems

Other biological impacts
Aside from the slowing and/or reversal of calcification, organisms may suffer other adverse effects, either indirectly through negative impacts on food resources, or directly as reproductive or physiological effects.[3] For example, the elevated oceanic levels of CO2 may produce CO
2-induced acidification of body fluids, known as hypercapnia.[123]
Increasing acidity has been observed to reduce metabolic rates in
Acoustic properties
Another potential route to ecosystem impacts is through
Algae and seagrasses
Another possible effect would be an increase in
Fish larvae
Ocean acidification can also have effects on marine
In an experimental aquarium facility, clownfish were sustained in non-manipulated seawater with pH 8.15 ± 0.07, which is similar to our current ocean's pH.[113] To test for effects of different pH levels, the seawater was modified to two other pH levels, which corresponded with climate change models that predict future atmospheric CO2 levels.[113] In the year 2100 the model projects possible CO2 levels of 1,000 ppm, which correlates with the pH of 7.8 ± 0.05.
This experiment showed that when larvae are exposed to a pH of 7.8 ± 0.05 their reaction to environmental cues differs drastically from their reaction to cues at pH equal to current ocean levels.[113] At pH 7.6 ± 0.05 larvae had no reaction to any type of cue. However, a meta-analysis published in 2022 found that the effect sizes of published studies testing for ocean acidification effects on fish behavior have declined by an order of magnitude over the past decade, and have been negligible for the past five years.[132]
Eel embryos, a "critically endangered" species[133] yet profound[clarification needed] in aquaculture, are also being affected by ocean acidification, specifically the European eel. Although they spend most of their lives in fresh water, usually in rivers, streams, or estuaries, they go to spawn and die in the Sargasso Sea. Here is where European eels are experiencing the effects of acidification in one of their key life stages.
Fish embryos and larvae are usually more sensitive to pH changes than adults, as organs for pH regulation are not full developed.[134] Because of this, European eel embryos are more vulnerable to changes in pH in the Sargasso Sea. A study of the European Eel in the Sargasso Sea was conducted in 2021 to analyze the specific effects of ocean acidification on embryos. The study found that exposure to predicted end-of-century ocean pCO2 conditions may affect normal development of this species in nature during sensitive early life history stages with limited physiological response capacities, while extreme acidification would negatively influence embryonic survival and development under hatchery conditions.[135]
Compounded effects of acidification, warming and deoxygenation
There is a substantial body of research showing that a combination of ocean acidification and elevated ocean temperature have a compounded effect on marine life and the ocean environment. This effect far exceeds the individual harmful impact of either.[138] In addition, ocean warming, along with increased productivity of phytoplankton from higher CO2 levels exacerbates ocean deoxygenation. Deoxygenation of ocean waters is an additional stressor on marine organisms that increases ocean stratification therefore limiting nutrients over time and reducing biological gradients.[139][140]
Meta analyses have quantified the direction and magnitude of the harmful effects of combined ocean acidification, warming and deoxygenation on the ocean.[141][142] These meta-analyses have been further tested by mesocosm studies that simulated the interaction of these stressors and found a catastrophic effect on the marine food web: thermal stress more than negates any primary producer to herbivore increase in productivity from elevated CO2.[143][144]
Impacts on the economy and societies
The increase of ocean acidity decelerates the rate of calcification in salt water, leading to smaller and slower growing coral reefs which supports approximately 25% of marine life.[145][146] Impacts are far-reaching from fisheries and coastal environments down to the deepest depths of the ocean.[17] The increase in ocean acidity in not only killing the coral, but also the wildly diverse population of marine inhabitants which coral reefs support.[147]
Fishing and tourism industry
The threat of acidification includes a decline in
Some 1 billion people are wholly or partially dependent on the fishing, tourism, and coastal management services provided by coral reefs. Ongoing acidification of the oceans may therefore threaten future
Arctic
In the Arctic, commercial fisheries are threatened because acidification harms calcifying organisms which form the base of the Arctic food webs (pteropods and brittle stars, see above). Acidification threatens Arctic food webs from the base up. Arctic food webs are considered simple, meaning there are few steps in the food chain from small organisms to larger predators. For example, pteropods are "a key prey item of a number of higher predators – larger plankton, fish, seabirds, whales".[148] Both pteropods and sea stars serve as a substantial food source and their removal from the simple food web would pose a serious threat to the whole ecosystem. The effects on the calcifying organisms at the base of the food webs could potentially destroy fisheries.
US commercial fisheries

The value of fish caught from US commercial fisheries in 2007 was valued at $3.8 billion and of that 73% was derived from calcifiers and their direct predators.[149] Other organisms are directly harmed as a result of acidification. For example, decrease in the growth of marine calcifiers such as the American lobster, ocean quahog, and scallops means there is less shellfish meat available for sale and consumption.[150] Red king crab fisheries are also at a serious threat because crabs are also calcifiers. Baby red king crab when exposed to increased acidification levels experienced 100% mortality after 95 days.[151] In 2006, red king crab accounted for 23% of the total guideline harvest levels and a serious decline in red crab population would threaten the crab harvesting industry.[152]
Possible responses
Climate change mitigation
Reducing carbon dioxide emissions (i.e. climate change mitigation measures) is the only solution that addresses the root cause of ocean acidification. For example, some mitigation measures focus on carbon dioxide removal (CDR) from the atmosphere (e.g. direct air capture (DAC), bioenergy with carbon capture and storage (BECCS)). These would also slow the rate of acidification.
Approaches that remove carbon dioxide from the ocean include
In total, "ocean-based methods have a combined potential to remove 1–100 gigatons of CO2 per year".[154]: TS-94 Their costs are in the order of USD40–500 per ton of CO2. For example, enhanced weathering could remove 2–4 gigatons of CO2 per year. This technology comes with a cost of 50–200 USD per ton of CO2.[154]: TS-94
Carbon removal technologies which add alkalinity
Some
Ocean alkalinity enhancement
Ocean alkalinity enhancement (OAE) is a proposed "carbon dioxide removal (CDR) method that involves deposition of alkaline minerals or their dissociation products at the ocean surface".
Enhanced weathering is one type of ocean alkalinity enhancement. Enhanced weathering increases alkalinity by scattering fine rock particles. This can happen on land and in the ocean (even though the outcome eventually affects the ocean).
In addition to sequestering CO2, alkalinity addition buffers the pH of the ocean therefore reducing ocean acidification. However, little is known about how organisms respond to added alkalinity, even from natural sources.[85] For example, weathering of some silicate rocks could release a large amount of trace metals at the weathering site.
Cost and energy consumed by ocean alkalinity enhancement (mining, pulverizing, transport) is high compared to other CDR techniques.[85] The cost is estimated to be 20–50 USD per ton of CO2 (for "direct addition of alkaline minerals to the ocean").[153]: 12–50
Carbon sequestered as bicarbonate in the ocean amounts to about 30% of carbon emissions since the Industrial Revolution.
Experimental materials include limestone, brucite, olivine and alkaline solutions. Another approach is to use electricity to raise alkalinity during desalination to capture waterborne CO2.[155]
Electrochemical methods
Electrochemical methods, or electrolysis, can strip carbon dioxide directly from seawater.[85] Electrochemical process are a type of ocean alkalinity enhancement, too. Some methods focus on direct CO2 removal (in the form of carbonate and CO2 gas) while others increase the alkalinity of seawater by precipitating metal hydroxide residues, which absorbs CO2 in a matter described in the ocean alkalinity enhancement section. The hydrogen produced during direct carbon capture can then be upcycled to form hydrogen for energy consumption, or other manufactured laboratory reagents such as hydrochloric acid.
However, implementation of electrolysis for carbon capture is expensive and the energy consumed for the process is high compared to other CDR techniques.[85] In addition, research to assess the environmental impact of this process is ongoing. Some complications include toxic chemicals in wastewaters, and reduced DIC in effluents; both of these may negatively impact marine life.[85]
Policies and goals
Global policies
As awareness about ocean acidification grows, policies geared towards increasing monitoring efforts of ocean acidification have been drafted.[156] Previously in 2015, ocean scientist Jean-Pierre Gattuso had remarked that "The ocean has been minimally considered at previous climate negotiations. Our study provides compelling arguments for a radical change at the UN conference (in Paris) on climate change".[157]
International efforts, such as the UN Cartagena Convention (entered into force in 1986),[158] are critical to enhance the support provided by regional governments to highly vulnerable areas to ocean acidification. Many countries, for example in the Pacific Islands and Territories, have constructed regional policies, or National Ocean Policies, National Action Plans, National Adaptation Plans of Action and Joint National Action Plans on Climate Change and Disaster Risk Reduction, to help work towards SDG 14. Ocean acidification is now starting to be considered within those frameworks.[159]
UN Ocean Decade
The UN Ocean Decade has a program called "Ocean acidification research for sustainability". It was proposed by the Global Ocean Acidification Observing Network (GOA-ON) and its partners, and has been formally endorsed as a program of the UN Decade of Ocean Science for Sustainable Development.[160][161] The OARS program builds on the work of GOA-ON and has the following aims: to further develop the science of ocean acidification; to increase observations of ocean chemistry changes; to identify the impacts on marine ecosystems on local and global scales; and to provide decision makers with the information needed to mitigate and adapt to ocean acidification.
Global Climate Indicators
The importance of ocean acidification is reflected in its inclusion as one of seven Global Climate Indicators.
Sustainable Development Goal 14
In 2015, the United Nations adopted the 2030 Agenda and a set of 17 Sustainable Development Goals (SDG), including a goal dedicated to the ocean, Sustainable Development Goal 14,[10] which calls to "conserve and sustainably use the oceans, seas and marine resources for sustainable development". Ocean acidification is directly addressed by the target SDG 14.3. The full title of Target 14.3 is: "Minimize and address the impacts of ocean acidification, including through enhanced scientific cooperation at all levels".[164] This target has one indicator: Indicator 14.3.1 which calls for the "Average marine acidity (pH) measured at agreed suite of representative sampling stations".[165]
The Intergovernmental Oceanographic Commission (IOC) of UNESCO was identified as the custodian agency for the SDG 14.3.1 Indicator. In this role, IOC-UNESCO is tasked with developing the SDG 14.3.1 Indicator Methodology, the annual collection of data towards the SDG 14.3.1 Indicator and the reporting of progress to the United Nations.[166][167]
Policies at country level
United States
In the United States, the Federal Ocean Acidification Research And Monitoring Act of 2009 supports government coordination, such as the National Oceanic Atmospheric Administration's (NOAA) "Ocean Acidification Program".[168][169] In 2015, USEPA denied a citizens petition that asked EPA to regulate CO2 under the Toxic Substances Control Act of 1976 in order to mitigate ocean acidification.[170][171] In the denial, the EPA said that risks from ocean acidification were being "more efficiently and effectively addressed" under domestic actions, e.g., under the Presidential Climate Action Plan, and that multiple avenues are being pursued to work with and in other nations to reduce emissions and deforestation and promote clean energy and energy efficiency.[172]
History
Research into the phenomenon of ocean acidification, as well as awareness raising about the problem, has been going on for several decades. The fundamental research really began with the creation of the
In the early 1970s questions over the long-term impact of the accumulation of fossil fuel CO2 in the sea were already arising around the world and causing strong debate. Researchers commented on the accumulation of fossil CO2 in the atmosphere and sea and drew attention to the possible impacts on marine life. By the mid-1990s, the likely impact of CO2 levels rising so high with the inevitable changes in pH and carbonate ion became a concern of scientists studying the fate of coral reefs.[174]
By the end of the 20th century the trade-offs between the beneficial role of the ocean in absorbing some 90 % of all heat created, and the accumulation of some 50 % of all fossil fuel CO2 emitted, and the impacts on marine life were becoming more clear. By 2003, the time of planning for the "First Symposium on the Ocean in a High-CO2 World" meeting to be held in Paris in 2004, many new research results on ocean acidification were published.[174]
In 2009, members of the
For example, research in 2010 found that in the 15-year period 1995–2010 alone, acidity had increased 6 percent in the upper 100 meters of the Pacific Ocean from Hawaii to Alaska.[48]
According to a statement in July 2012 by Jane Lubchenco, head of the U.S. National Oceanic and Atmospheric Administration "surface waters are changing much more rapidly than initial calculations have suggested. It's yet another reason to be very seriously concerned about the amount of carbon dioxide that is in the atmosphere now and the additional amount we continue to put out."[177]
A 2013 study found acidity was increasing at a rate 10 times faster than in any of the evolutionary crises in Earth's history.[178]
The "Third Symposium on the Ocean in a High-CO2 World" took place in Monterey, California, in 2012. The summary for policy makers from the conference stated that "Ocean acidification research is growing rapidly".[94]
In a synthesis report published in Science in 2015, 22 leading marine scientists stated that CO2 from burning fossil fuels is changing the oceans' chemistry more rapidly than at any time since the Great Dying (Earth's most severe known extinction event).[157] Their report emphasized that the 2 °C maximum temperature increase agreed upon by governments reflects too small a cut in emissions to prevent "dramatic impacts" on the world's oceans.[157]
See also
- Biological pump – Carbon capture process in oceans
- Free Ocean CO2 Enrichment – technology for studying ocean acidification
- Carbon sink – Reservoir absorbing more carbon from, than emitting to, the air
- Estuarine acidification – Reducing pH values in coastal marine ecosystems
- Holocene extinction – Ongoing extinction event caused by human activity
- Ocean acidification in the Arctic Ocean
- Ocean acidification in the Great Barrier Reef – Threat to the reef which reduces the viability and strength of reef-building corals
- Ocean deoxygenation – Reduction of the oxygen content of the oceans
- Water pollution
References
- ^ Ritchie, Roser, Mispy, Ortiz-Ospina. "SDG 14 – Measuring progress towards the Sustainable Development Goals Archived 22 January 2022 at the Wayback Machine." SDG-Tracker.org, website (2018).
- ^ S2CID 255431338.
Figure 1f
- ^ ISBN 0-85403-617-2.
- ^ PMID 31819102.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - S2CID 214271838.
- S2CID 221369828.
- PMID 18988740.
- ^ a b Cornelia Dean (30 January 2009). "Rising Acidity Is Threatening Food Web of Oceans, Science Panel Says". New York Times.
- ^ PMID 22798578.
- ^ a b "Goal 14 targets". UNDP. Retrieved 24 September 2020.
- ^ IPCC (2022) Chapter 12: Cross sectoral perspectives
- ^ Archived 13 October 2022 at the Wayback Machine in Climate Change 2022: Mitigation of Climate ChangeContribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change]
- ^ Archived 2 August 2022 at the Wayback Machine, Cambridge University Press, Cambridge, United Kingdom and New York, NY, US: 12–36
- ^ Wikidata Q28261134.
- ^ "The Carbon Cycle". earthobservatory.nasa.gov. 16 June 2011. Retrieved 16 November 2022.
- ^ doi:10.5194/essd-14-4811-2022.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - ^ S2CID 225741986.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - Encyclopædia Britannica Online. Retrieved 11 February 2010.
- ISBN 978-0-613-91814-5.
- ^ a b c d e Cooley, S., D. Schoeman, L. Bopp, P. Boyd, S. Donner, D.Y. Ghebrehiwet, S.-I. Ito, W. Kiessling, P. Martinetto, E. Ojea, M.-F. Racault, B. Rost, and M. Skern-Mauritzen, 2022: Chapter 3: Oceans and Coastal Ecosystems and Their Services Archived 21 October 2022 at the Wayback Machine. In: Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 28 February 2022 at the Wayback Machine [H.-O. Pörtner, D.C. Roberts, M. Tignor, E.S. Poloczanska, K. Mintenbeck, A. Alegría, M. Craig, S. Langsdorf, S. Löschke, V. Möller, A. Okem, B. Rama (eds.)]. Cambridge University Press, Cambridge, UK and New York, NY, US, pp. 379–550.
- ^ Nina Notman (29 July 2014). "The other carbon dioxide problem". Chemistry World.
- ^ Alex Rogers (9 October 2013). "Global warming's evil twin: ocean acidification". The Conversation.
- ^ "Ocean acidification (Issues Brief)" (PDF). IUCN (International Union for Conservation of Nature). November 2017. Retrieved 3 November 2020.
- hdl:2262/27779.
- S2CID 135282569.
- ^ Paul Freund; Stefan Bachu; Dale Simbeck; Kelly (Kailai) Thambimuthu; Murlidhar Gupta (2005). "Annex I: Properties of CO2 and carbon-based fuels". In Bert Metz; Ogunlade Davidson; Heleen de Coninck; Manuela Loos; Leo Meyer (eds.). IPCC Special Report on Carbon Dioxide Capture and Storage (PDF). IPCC. p. 390. Archived from the original (PDF) on 10 February 2010. Retrieved 1 November 2014.
- ^ S2CID 84841808.
- .
- PMID 30700764.
- .
- ^ Gies, E. (11 January 2018). "Like Oceans, Freshwater Is Also Acidifying". Scientific American. Retrieved 13 January 2018.
- PMID 29337079.
- ^ a b IPCC, 2021: Annex VII: Glossary Archived 5 June 2022 at the Wayback Machine [Matthews, J.B.R., V. Möller, R. van Diemen, J.S. Fuglestvedt, V. Masson-Delmotte, C. Méndez, S. Semenov, A. Reisinger (eds.)]. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 9 August 2021 at the Wayback Machine [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, US
- ^ "What is alkalinity?". Water Research Center. 2014. Retrieved 5 February 2018.
- .
- S2CID 31054160.
- .
- ISBN 978-0-13-143888-0.
- S2CID 4306199.
- .
- ^ S2CID 35487689.
- ^ S2CID 53062358.
- PMID 30373837.
- .
- ^ "Atmospheric CO2 and Ocean pH". cleanet.org. Retrieved 17 November 2022.
- ^ "Quality of pH Measurements in the NODC Data Archives". www.pmel.noaa.gov. Retrieved 18 December 2023.
- ^ "The pH Scale". Woods Hole Oceanographic Institution. Retrieved 29 March 2023.
- ^ a b Marah J. Hardt; Carl Safina (9 August 2010). "How Acidification Threatens Oceans from the Inside Out". Scientific American. Archived from the original on 26 December 2010.
- ^ a b c d Arias, P.A., N. Bellouin, E. Coppola, R.G. Jones, G. Krinner, J. Marotzke, V. Naik, M.D. Palmer, G.-K. Plattner, J. Rogelj, M. Rojas, J. Sillmann, T. Storelvmo, P.W. Thorne, B. Trewin, K. Achuta Rao, B. Adhikary, R.P. Allan, K. Armour, G. Bala, R. Barimalala, S. Berger, J.G. Canadell, C. Cassou, A. Cherchi, W. Collins, W.D. Collins, S.L. Connors, S. Corti, F. Cruz, F.J. Dentener, C. Dereczynski, A. Di Luca, A. Diongue Niang, F.J. Doblas-Reyes, A. Dosio, H. Douville, F. Engelbrecht, V. Eyring, E. Fischer, P. Forster, B. Fox-Kemper, J.S. Fuglestvedt, J.C. Fyfe, et al., 2021: Technical Summary Archived 21 July 2022 at the Wayback Machine. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 9 August 2021 at the Wayback Machine [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, US, pp. 33–144.
- ^ a b c d Canadell, J.G., P.M.S. Monteiro, M.H. Costa, L. Cotrim da Cunha, P.M. Cox, A.V. Eliseev, S. Henson, M. Ishii, S. Jaccard, C. Koven, A. Lohila, P.K. Patra, S. Piao, J. Rogelj, S. Syampungani, S. Zaehle, and K. Zickfeld, 2021: Chapter 5: Global Carbon and other Biogeochemical Cycles and Feedbacks Archived 27 July 2022 at the Wayback Machine. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 9 August 2021 at the Wayback Machine [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, US, pp. 673–816.
- ^ .
- .
- .
- .
- .
- .
- PMID 19666624.
- .
- S2CID 129883047.
- .
- ^ Josep G. Canadell, J. G., P. M. S. Monteiro, M. H. Costa, L. Cotrim da Cunha, P. M. Cox, A. V. Eliseev, S. 3 Henson, M. Ishii, S. Jaccard, C. Koven, A. Lohila, P. K. Patra, S. Piao, J. Rogelj, S. Syampungani, S. Zaehle, 4 K. Zickfeld, 2021, Global Carbon and other Biogeochemical Cycles and Feedbacks Supplementary Material. Chapter 5 Archived 18 November 2022 at the Wayback Machine In: Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth 6 Assessment Report of the Intergovernmental Panel on Climate Change Archived 9 August 2021 at the Wayback Machine [Masson-Delmotte, V., P. Zhai, A. 7 Pirani, S. L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M. I. Gomis, M. Huang, K. 8 Leitzell, E. Lonnoy, J.B.R. Matthews, T. K. Maycock, T. Waterfield, O. Yelekçi, R. Yu and B. Zhou (eds.)]. 9 Available from https://ipcc.ch/static/ar6/wg1.
- ^ Bond, D.P.G., Wignall, P.B., Joachimski, M.M., Sun, Y., Savov, I., Grasby, S.E., Beauchamp, B. and Blomeier, D.P. 2015. An abrupt extinction in the Middle Permian (Capitanian) of the Boreal Realm (Spitsbergen) and its link to anoxia and acidification. Geological Society of America Bulletin, 127 (9–10): 1411–1421.
- ^ Hand, Eric (16 April 2015). "Sixth extinction, rivaling that of the dinosaurs, should join the big five, scientists say". Science.
- ^ Berezow, Alex (21 April 2015). "New mass extinction event identified by geologists". BBC.
- ^ S2CID 28891777.
- ^ hdl:11511/35436.
- PMID 20421502.
- S2CID 130658467.
- ^ .
- ^ .
- ^ PMID 31636204.
- ^ S2CID 133715470.
- ^ S2CID 26909706.
- S2CID 53590993.
- S2CID 133353132.
- S2CID 42675849.
- S2CID 130434497
- S2CID 28535145.
- .
- PMID 28858305.
- ^ Joel, Lucas (21 October 2019). "The Dinosaur-Killing Asteroid Acidified the Ocean in a Flash – The Chicxulub event was as damaging to life in the oceans as it was to creatures on land, a study shows". The New York Times. Retrieved 22 October 2019.
- ^ "The Geological Record of Ocean Acidification". JournalistsResource.org, retrieved 14 March 2012
- ^ "An Ominous Warning on the Effects of Ocean Acidification by Carl Zimmer: Yale Environment 360". e360.yale.edu. Archived from the original on 16 February 2014. Retrieved 25 January 2014.
- ^ Newspapers, Les Blumenthal-McClatchy (22 April 2010). "Report: Ocean acidification rising at unprecedented rate". mcclatchydc.
- ^ S2CID 245089649.
- ^ PMID 24143135.
- ^ Lee, J.-Y., J. Marotzke, G. Bala, L. Cao, S. Corti, J.P. Dunne, F. Engelbrecht, E. Fischer, J.C. Fyfe, C. Jones, A. Maycock, J. Mutemi, O. Ndiaye, S. Panickal, and T. Zhou, 2021: Chapter 4: Future Global Climate: Scenario-Based Projections and Near- Term Information Archived 9 November 2022 at the Wayback Machine. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 18 October 2022 at the Wayback Machine [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, US, pp. 553–672.
- ^ "PMEL CO2 – Carbon Dioxide Program". www.pmel.noaa.gov. Retrieved 6 September 2021.
- ^ a b National Research Council. Overview of Climate Changes and Illustrative Impacts. Climate Stabilization Targets: Emissions, Concentrations, and Impacts over Decades to Millennia Archived 6 September 2015 at the Wayback Machine. Washington, DC: The National Academies Press, 2011. 1. Print.
- .
- PMID 18460426.
- ^ .
- S2CID 133783884.
- ^ a b "Ocean Acidification Summary for Policymakers". IGBP. 14 November 2013.
- ^ "Special Report on the Ocean and Cryosphere in a Changing Climate — Special Report on the Ocean and Cryosphere in a Changing Climate". IPCC. 25 September 2019. Retrieved 12 November 2019.
- .
- .
- .
- S2CID 4426501.
- S2CID 130190489.
- ^ S2CID 206511068.
- .
- S2CID 14774230.
- .
- ^ Petrou, Katherina; Nielsen, Daniel (27 August 2019). "Acid oceans are shrinking plankton, fueling faster climate change". phys.org. Retrieved 12 November 2019.
- ^ "Acidification Of Oceans May Contribute To Global Declines Of Shellfish, Study By Stony Brook Scientists Concludes" (Press release). School of Marine and Atmospheric Sciences at Stony Brook University. 27 September 2010. Archived from the original on 3 September 2012. Retrieved 4 June 2012.
- .
- .
- S2CID 3497477.
- PMID 29378969.
- S2CID 205247928.
- S2CID 3935534.
- ^ PMID 19188596.
- S2CID 9375062.
- S2CID 85749253.
- ^ "Effects of Ocean Acidification on Marine Species & Ecosystems". Report. OCEANA. Retrieved 13 October 2013.
- .
- JSTOR 24861875.
- S2CID 131084581.
- S2CID 219729997.
- ^ "Comprehensive study of Arctic Ocean acidification". Study. CICERO. Archived from the original on 10 December 2013. Retrieved 14 November 2013.
- ^ Woods Hole Oceanographic Institution (August 2016). "Changes in Aragonite Saturation of the World's Oceans, 1880–2015". epa.gov.
- .
- PMID 19075232.
- doi:10.3354/ab00037.
- ^ Kwok, Roberta (4 June 2013). "Ocean acidification could make squid develop abnormally". University of Washington. Retrieved 24 August 2013.
- ^ "Swiss marine researcher moving in for the krill". The Australian. 2008. Archived from the original on 11 December 2008. Retrieved 28 September 2008.
- .
- ^ Acid In The Oceans: A Growing Threat To Sea Life Archived 12 May 2018 at the Wayback Machine by Richard Harris. All Things Considered, 12 August 2009.
- ^ "Ocean Acidification Promotes Disruptive and Harmful Algal Blooms on Our Coasts". 2014.
- ^ .
- PMID 35113875.
- ^ Pike C, Crook V, GollockM. Anguilla anguilla. The IUCN Red List ofThreatened Species 2020: e. T60344A152845178. International Union for Conservation ofNature; 2020. https://doi.org/10.2305/ IUCN.UK.2020-2.RLTS.T60344A152845178.en
- ^ Brauner CJ, Shartau RB, Damsgaard C, EsbaughAJ, Wilson RW, Grosell M. Acid-base physiology and CO2 homeostasis: Regulationand compensation inresponse toelevated environmental CO2. Fish Physiology.Elsevier ;2019. pp. 69–132.https://doi.or g/10.1016/bs.fp.2019.08.003
- ^ Sganga, Daniela E., et al. "CO2 Induced Seawater Acidification Impacts Survival and Development of European Eel Embryos." PLOS ONE, edited by Hans G. Dam, vol. 17, no. 4, Apr. 2022, p. e0267228. DOI.org (Crossref), https://doi.org/10.1371/journal.pone.0267228.
- S2CID 202922296..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - S2CID 4358903.
- hdl:11329/1892.
- .
- PMID 12048249.
- S2CID 836308.
- PMC 3597261.
- S2CID 8015698.
- PMID 26559327.
- ^ "Coral reefs". WWF. Retrieved 6 May 2019.
- .
- S2CID 12607336.
- ^ "Antarctic marine wildlife is under threat, study finds". BBC Nature. Retrieved 13 October 2013.
- ^ V. J. Fabry; C. Langdon; W. M. Balch; A. G. Dickson; R. A. Feely; B. Hales; D. A. Hutchins; J. A. Kleypas & C. L. Sabine. "Present and Future Impacts of Ocean Acidification on Marine Ecosystems and Biogeochemical Cycles" (PDF). Report of the Ocean Carbon and Biogeochemistry Scoping Workshop on Ocean Acidification Research. Archived from the original (PDF) on 17 November 2010. Retrieved 14 November 2013.
- ^ "Canada's State of the Oceans Report, 2012". Report. Fisheries and Oceans Canada. 2012. Archived from the original on 6 November 2013. Retrieved 21 October 2013.
- ^ Robert J. Foy; Mark Carls; Michael Dalton; Tom Hurst; W. Christopher Long; Dusanka Poljak; André E. Punt; Michael F. Sigler; Robert P. Stone; Katherine M. Swiney (Winter 2013). "CO 2 , pH, and Anticipating a Future under Ocean Acidification" (PDF). Oncorhynchus. Vol. XXXIII, no. 1. Retrieved 14 November 2013.
- ^ "Bering Sea Crab Fishery". Report. Seafood Market Bulletin. November 2005. Archived from the original on 11 December 2013. Retrieved 10 November 2013.
- ^ a b c IPCC (2022) Chapter 12: Cross sectoral perspectives Archived 13 October 2022 at the Wayback Machine in Climate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 2 August 2022 at the Wayback Machine, Cambridge University Press, Cambridge, United Kingdom and New York, NY, US
- ^ a b IPCC (2022) Technical Summary Archived 23 September 2022 at the Wayback Machine. In Climate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 2 August 2022 at the Wayback Machine, Cambridge University Press, Cambridge, United Kingdom and New York, NY, US
- ^ Cornwall, Warren (30 November 2023). "An Alkaline Solution". Science Magazine.
- ^ "Action Plans". OA Alliance. Retrieved 4 November 2022.
- ^ a b c Harrabin, Roger (3 July 2015). "CO2 emissions threaten ocean crisis". BBC News.
- ^ "Cartagena Convention | The Caribbean Environment Programme (CEP)". www.unep.org. Retrieved 4 November 2022.
- ^ Turner, J; McIntosh, R. Duncan (2019). Mainstreaming Ocean Acidification into National Policies: A Handbook for Pacific Islands (PDF). Apia, Samoa: Secretariat of the Pacific Regional Environment Programme.
- ^ "The Ocean Decade – The Science we need for the Ocean we want". Ocean Decade. Retrieved 4 November 2022.
- ^ "Overview". OARS: Ocean Acidification Research for Sustainability. GOA-ON.
- ^ "Global Climate Indicators". GCOS. WMO.
- ^ "Home | GCOS". gcos.wmo.int. Retrieved 4 November 2022.
- ^ United Nations (2017) Resolution adopted by the General Assembly on 6 July 2017, Work of the Statistical Commission pertaining to the 2030 Agenda for Sustainable Development (A/RES/71/313 Archived 23 October 2020 at the Wayback Machine)
- ^ "Goal 14: Sustainable Development Knowledge Platform". sustainabledevelopment.un.org. Retrieved 5 September 2020.
- ^ "Update on IOC Custodianship Role in Relation to SDG 14 Indicators". unesdoc.unesco.org. Retrieved 4 November 2022.
- ^ "SDG 14.3.1 data portal". oa.iode.org. Retrieved 4 November 2022.
- S2CID 218961473.
- ^ "OAP Home". oceanacidification.noaa.gov. Retrieved 4 November 2022.
- ^ Center for Biological Diversity; Donn J. Viviani. "TSCA Section 21 Petition Requesting EPA to Regulate Anthropogenic Emissions Carbon Dioxide" (PDF). US EPA.
- ^ "Carbon Dioxide Emissions and Ocean Acidification; TSCA Section 21 Petition; Reasons for Agency Response". Environmental Protection Agency (EPA). 7 October 2015.
- ^ "The President's Climate Action Plan" (PDF). obamawhitehouse.archives.gov. June 2013.
- ^ Sørensen, S. P. L. (1909). "Über die Messung und die Bedeutung der Wasserstoffionenkonzentration bei enzymatischen Prozessen" (PDF). Biochem. Z. 21: 131–304. Archived (PDF) from the original on 15 April 2021. Retrieved 22 March 2021.
Original German: Für die Zahl p schlage ich den Namen Wasserstoffionenexponent und die Schreibweise pH• vor. Unter dem Wasserstoffionexponenten (pH•) einer Lösungwird dann der Briggsche Logarithmus des reziproken Wertes des auf Wasserstoffionenbezagenen Normalitäts faktors de Lösungverstanden.
Two other publications appeared in 1909, one in French and one in Danish. - ^ ISSN 1726-4170.
 Text was copied from this source, which is available under a Creative Commons Attribution 3.0 Unported License
Text was copied from this source, which is available under a Creative Commons Attribution 3.0 Unported License - ^ IAP (June 2009). "Interacademy Panel (IAP) Member Academies Statement on Ocean Acidification"., Secretariat: TWAS (the Academy of Sciences for the Developing World), Trieste, Italy.
- ^ Harvey, Fiona (4 December 2019). "Tackling degraded oceans could mitigate climate crisis – report". The Guardian. Retrieved 7 December 2019.
- Huffington Post. 9 July 2012. Archived from the originalon 12 July 2012. Retrieved 9 July 2012.
- ^ Fiona Harvey (25 August 2013). "Rising levels of acids in seas may endanger marine life, says study". The Guardian. Retrieved 29 August 2013.

![{\displaystyle {\Omega }={\frac {\left[{\ce {Ca^2+}}\right]\left[{\ce {CO3^2-}}\right]}{K_{sp}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/0422ec8bbb280ae8547a20d486b928b0c21846f4)