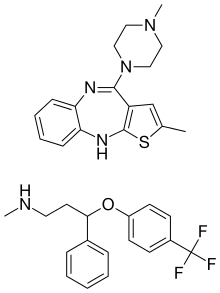
Olanzapine/fluoxetine
 | |
| Combination of | |
|---|---|
| Olanzapine | Atypical antipsychotic |
| Fluoxetine | Selective serotonin reuptake inhibitor |
| Clinical data | |
| Trade names | Symbyax, Cinol Forte, Olapin Forte, others |
| AHFS/Drugs.com | Professional Drug Facts |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| KEGG | |
| | |
Olanzapine/fluoxetine (trade name Symbyax, created by
Medical uses
Olanzapine/fluoxetine was approved by the U.S. Food and Drug Administration (FDA) to treat the depressive episodes of bipolar I disorder in 2003.[1] In 2009, it was granted approval for the treatment of treatment-resistant depression.[4]
Olanzapine/fluoxetine, or other
Side effects
Possible side effects of olanzapine/fluoxetine include all those of the two component drugs:
Olanzapine/fluoxetine could produce a severe allergic reaction and should not be used if the patient has previously experienced an allergic reaction to either fluoxetine or olanzapine.[9]
Olanzapine is correlated with an increase in blood sugar. Patients with diabetes, or those at risk for developing it, require careful monitoring.[9]
In rare cases, olanzapine/fluoxetine may cause neuroleptic malignant syndrome.[1]
Like other SSRIs, olanzapine/fluoxetine carries a boxed warning stating that it could increase the risk of suicidal thoughts and behaviors in patients aged 24 and under. The warning also states that olanzapine/fluoxetine may increase the risk of death in elderly patients with dementia-related psychosis.[1]
See also
- Amitriptyline/perphenazine
- Aripiprazole/sertraline
- "California rocket fuel"
- Flupentixol/melitracen
- Tranylcypromine/trifluoperazine
References
- ^ a b c d e f "Symbyax- olanzapine and fluoxetine hydrochloride capsule". DailyMed. 21 April 2020. Retrieved 30 September 2020.
- ^ "Tratamento medicamentoso dos transtornos bipolares - Transtornos psiquiátricos". Manuais MSD edição para profissionais (in Brazilian Portuguese). Retrieved 2022-10-02.
- PMID 19906346.
- ^ Grohol, J. "FDA Approves Symbyax for Treatment Resistant Depression". Psych Central Blog. Archived from the original on 2017-12-26. Retrieved 2010-07-17.
- PMID 14700010.
- S2CID 38506292.
- PMID 10937610.
- PMID 12359687.
- ^ a b "Symbyax". Drugs.com.
External links
- "Fluoxetine hydrochloride mixture with Olanzapine". Drug Information Portal. U.S. National Library of Medicine.
