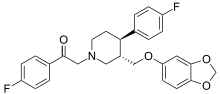
Omiloxetine
Source: Wikipedia, the free encyclopedia.
Chemical compound
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
JSmol) | |
| Density | 1.3±0.1 g/cm3 |
| Melting point | 228.65 °C (443.57 °F) |
| Boiling point | 587.2 °C (1,089.0 °F) |
| Solubility in water | 0.0015 mg/mL (20 °C) |
| |
| |
Omiloxetine (omiloextinum, omiloxetino
INN)[1] was a selective serotonin reuptake inhibitor drug candidate that underwent preclinical development by the Spanish pharmaceutical company, Ferrer Internacional, until 2005, when it was abandoned.[2][3]
Rafael Foguet also patented Abaperidone.
References
- ^ "Proposed INN: List 76" (PDF). WHO Drug Information. 10 (4): 212–213. 1996.
- ^ Terencio J (2005-12-05). "Omiloxetine - Ferrer licensing offer, worldwide". R & D Focus Drug News. Archived from the original on 2016-03-07. Retrieved 2012-05-19.
- ^ De Lecea C (2003-12-08). "Omiloxetine - Ferrer partnering opportunity, worldwide Ferrer preclinical data". R & D Focus Drug News. Archived from the original on 2016-03-07. Retrieved 2012-05-19.
| ||
|---|---|---|
SNRIs Tooltip Serotonin–norepinephrine reuptake inhibitors | ||
NRIs Tooltip Norepinephrine reuptake inhibitors | ||
NDRIs Tooltip Norepinephrine–dopamine reuptake inhibitors | ||
NaSSAs Tooltip Noradrenergic and specific serotonergic antidepressants | ||
SARIs Tooltip Serotonin antagonist and reuptake inhibitors | ||
SMS Tooltip Serotonin modulator and stimulators | ||
| Others |
| |
TCAs Tooltip Tricyclic antidepressants |
| |
|---|---|---|
TeCAs Tooltip Tetracyclic antidepressants | ||
| Others | ||
| |||||||
|
Miscellaneous | |
|---|---|
|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
