Opsin


Animal opsins are
Structure and function
Animal opsins detect light and are the molecules that allow us to see. Opsins are
In the vertebrate photoreceptor cells, all-trans-retinal is released and replaced by a newly synthesized 11-cis-retinal provided from the retinal epithelial cells.
Beside 11-
Functionally conserved residues and motifs
The seven transmembrane α-helical domains in opsins are connected by three extra-cellular and three cytoplasmic loops. Along the α-helices and the loops, many amino acid residues are highly conserved between all opsin groups, indicating that they serve important functions and thus are called functionally conserved residues. Actually, insertions and deletions in the α-helices are very rare and should preferentially occur in the loops. Therefore, different G-protein-coupled receptors have different length and homologous residues may be in different positions. To make such positions comparable between different receptors, Ballesteros and Weinstein introduced a common numbering scheme for G-protein-coupled receptors.[23] The number before the period is the number of the transmembrane domain. The number after the period is set arbitrarily to 50 for the most conserved residue in that transmembrane domain among GPCRs known in 1995. For instance in the seventh transmembrane domain, the proline in the highly conserved NPxxY7.53 motif is Pro7.50, the asparagine before is then Asp7.49, and the tyrosine three residues after is then Tyr7.53.[21] Another numbering scheme is based on cattle rhodopsin. Cattle rhodopsin has 348 amino acids and is the first opsin whose amino acid sequence[24] and whose 3D-structure were determined.[12] The cattle rhodopsin numbering scheme is widespread in the opsin literature.[21] Therefore, it is useful to use both schemes.
The retinal binding lysine
Opsins without the retinal binding lysine are not light sensitive.
Such function does not need to be light detection, as some opsins are also involved in
Beside these functions, an opsin without Lys2967.43, such as a gluopsin, could still be light sensitive, since in cattle rhodopsin, the retinal binding lysine can be shifted from position 296 to other positions, even into other transmembrane domains, without changing light sensitivity.[35]
-
Most known opsins have the retinal binding lysine except some among the tetraopins, The outgroup contains other G protein-coupled receptors.
-
Most tetraopsins have also the retinal binding lysine except some of the chromopsins, which are highlighted by the frame and expanded in the next image. The outgroup contains other G protein-coupled receptors including the other opsins.
-
Most chromopsins have also the retinal binding lysine except the nemopsins, where it is replaced byargenine (R), and the gluopsins, where it is replaced by glutamic acid(E). The astropsins, the nemopsins and the gluopsins are highlighted by the frames. The outgroup contains other G protein-coupled receptors including the other opsins.
In the
The NPxxY motif
The
Also mutations of
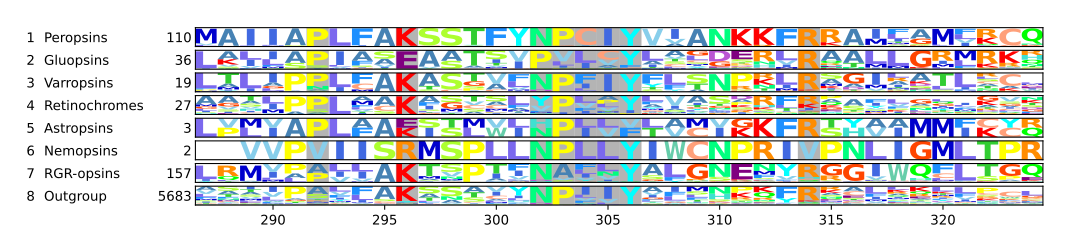
Other residues and motifs
Cys138 and Cys110 form a highly conserved
Spectral tuning sites
Certain amino acid residues, termed spectral tuning sites, have a strong effect on λmax values. Using site-directed mutagenesis, it is possible to selectively mutate these residues and investigate the resulting changes in light absorption properties of the opsin. It is important to differentiate spectral tuning sites, residues that affect the wavelength at which the opsin absorbs light, from functionally conserved sites, residues important for the proper functioning of the opsin. They are not mutually exclusive, but, for practical reasons, it is easier to investigate spectral tuning sites that do not affect opsin functionality. For a comprehensive review of spectral tuning sites see Yokoyama[49] and Deeb.[50] The impact of spectral tuning sites on λmax differs between different opsin groups and between opsin groups of different species.
Opsins in the human eye, brain, and skin
| Abbr. | Name | λmax | Color | Eye | Brain | Skin | Chromosomal location[44] |
|---|---|---|---|---|---|---|---|
| OPN1LW | L-cone (red-cone) opsin | 557 nm | Yellow | Cone | — | — | Xq28[44] |
| OPN1MW | M-cone (green-cone) opsin | 527 nm | Green | Cone | — | — | Xq28[44] |
| OPN1SW | S-cone (blue-cone) opsin | 420 nm | Violet | Cone | — | Melanocytes, keratinocytes[51] | 7q32.1[44] |
| OPN2 (RHO) | Rhodopsin | 505 nm | Blue–green | Rod | — | Melanocytes, keratinocytes[51] | 3q22.1[44] |
| OPN3 | Encephalopsin, panopsin | S-M | Blue–green | Rod, cone, OPL, IPL, GCL[52] | Cerebral cortex, cerebellum, striatum, thalamus, hypothalamus[53][54] | Melanocytes, keratinocytes[51] | 1q43[44] |
OPN4
|
Melanopsin | 480 nm[55] | Sky blue | ipRGC[55] | — | — | 10q23.2[44] |
| OPN5 | Neuropsin | 380 nm[56] | Ultraviolet[56] | Neural retina, RPE[57] | Anterior hypothalamus[58] | Melanocytes, keratinocytes[51] | 6p12.3[44] |
| RRH | Peropsin | RPE cells - microvilli | — | — | 4q25[44] | ||
| RGR | Retinal G protein coupled receptor | RPE cells | — | — | 10q23.1[44] |
RPE,
Cuttlefish
Cuttlefish and octopuses contain opsin in their skin as part of the chromophores. The opsin is part of the sensing network detecting the colour and shape of the cuttlefish's surroundings.[59][60][61]
Phylogeny
Animal opsins (also known as type 2 opsins) are members of the seven-transmembrane-domain proteins of the G protein-coupled receptor (GPCR) superfamily.[1][2]
Animal opsins fall phylogenetically into five groups: The ciliary opsins (cilopsins, c-opsins), the
Animal visual opsins are traditionally classified as either ciliary or rhabdomeric. Ciliary opsins, found in
Ciliary opsins
Ciliary opsins (cilopsins, c-opsins) are expressed in ciliary photoreceptor cells, and include the vertebrate visual opsins and encephalopsins.[66] They convert light signals to nerve impulses via cyclic nucleotide gated ion channels, which work by increasing the charge differential across the cell membrane (i.e. hyperpolarization.[67])
Vertebrate visual opsins
Vertebrate visual opsins are a subclass of ciliary opsins that express in the vertebrate retina and mediate vision. They are further subdivided into:
- cone opsins. Photopsins are further subdivided according to their spectral sensitivity, namely the wavelength at which the highest light absorption is observed (λmax). Vertebrates generally have four (SWS1, SWS2, RH2, LWS) classes of photopsins.[68][69] Mammals lost Rh2 and SWS2 classes during the nocturnal bottleneck, so are generally dichromatic. Primate ancestors later developed two distinct LWS opsins (LWS and MWS), leaving humans with 3 photopsins in 2 classes: SWS1 (OPN1SW) and two forms of LWS (OPN1LW, OPN1MW).
Extraretinal (or extra-ocular) Rhodopsin-Like Opsins (Exo-Rh)
These pineal opsins, found in the Actinopterygii (ray-finned fish) apparently arose as a result of gene duplication from Rh1 (rhodopsin). These opsins appear to serve functions similar to those of pinopsin found in birds and reptiles.[71] [72]
Pinopsins
The first Pineal Opsin (Pinopsin) was found in the chicken pineal gland. It is a blue sensitive opsin (λmax = 470 nm).[73][74]
Pineal opsins have a wide range of expression in the brain, most notably in the pineal region.
Vertebrate Ancient (VA) opsin
Vertebrate Ancient (VA) opsin has three isoforms VA short (VAS), VA medium (VAM), and VA long (VAL). It is expressed in the inner retina, within the horizontal and
Parapinopsins
The first parapinopsin (PP) was found in the
Parietopsins
The first parietopsin was found in the photoreceptor cells of the lizard parietal eye. The lizard parietopsin is green-sensitive (λmax = 522 nm), and despite it is a c-opsin, like the vertebrate visual opsins, it does not induce hyperpolarization via a Gt-protein, but induces depolarization via a Go-protein.[80][81]
Encephalopsin or Panopsin
The panopsins are found in many tissues (skin,[51] brain,[53][82] testes,[53] heart, liver,[82] kidney, skeletal muscle, lung, pancreas and retina[82]). They were originally found in the human and mouse brain and thus called encephalopsin.[53]
The first invertebrate panopsin was found in the ciliary photoreceptor cells of the annelid
Panopsins are also expressed in the brains of some insects.[66] The panopsins of mosquito and pufferfish absorb maximally at 500 nm and 460 nm, respectively. Both activate in vitro Gi and Go proteins.[90]
The panopsins are sister to the TMT-opsins.[28][91][47][92]
Teleost Multiple Tissue (TMT) Opsin
The first TMT-opsin was found in many tissues in
Opsins in cnidarians
Rhabdomeric opsins
Rhabdomeric opsins (rhabopsins, r-opsins) are also known as Gq-opsins, because they couple to a Gq-protein. Rhabopsins are used by molluscs and arthropods. Arthropods appear to attain colour vision in a similar fashion to the vertebrates, by using three (or more) distinct groups of opsins, distinct both in terms of phylogeny and spectral sensitivity.[66] The rhabopsin melanopsin is also expressed in vertebrates, where it regulates circadian rhythms and mediates the pupillary reflex.[66]
Unlike cilopsins, rhabopsins are associated with canonical transient receptor potential ion channels; these lead to the electric potential difference across a cell membrane being eradicated (i.e. depolarization).[67]
The identification of the crystal structure of squid rhodopsin[13] is likely to further our understanding of its function in this group.
Arthropods use different opsins in their different eye types, but at least in Limulus the opsins expressed in the lateral and the compound eyes are 99% identical and presumably diverged recently.[98]
Melanopsin
Melanopsin (OPN4) is involved in circadian rhythms, the pupillary reflex, and color correction in high-brightness situations. Phylogenetically, it is a member of the rhabdomeric opsins (rhabopsins, r-opsins) and functionally and structurally a rhabopsin, but does not occur in rhabdomeres.
Tetraopsins
The tetraopsins include the
Neuropsins
Neuropsins are sensitive to UVA, typically at 380 nm. They are found in the brain, testes, skin, and retina of humans and rodents, as well as in the brain and retina of birds. In birds and rodents they mediate ultraviolet vision.[51][56][99] They couple to Gi-proteins.[56][99] In humans, Neuropsin is encoded by the OPN5 gene. In the human retina, its function is unknown. In the mouse, it photo-entrains the retina and cornea at least ex vivo.[100]
Go-opsins
Go-opsins are absent from
RGR-opsins
RGR-opsins, also known as
Peropsin
Other proteins called opsins
Photoreceptors can be classified several ways, including function (vision, phototaxis, photoperiodism, etc.), type of
Beside animal opsins, which are
Microbial and animal opsins are also called type 1 and type 2 opsins respectively. Both types are called opsins, because at one time it was thought that they were related: Both are seven-transmembrane receptors and bind covalently retinal as chromophore, which turns them into photoreceptors sensing light. However, both types are not related on the sequence level.[113]
In fact, the sequence identity between animal and mirobial opsins is no greater than could be accounted for by random chance. However, in recent years new methods have been developed specific to deep
Most microbial opsins are
Microbial opsins are often known by the rhodopsin form of the molecule, i.e., rhodopsin (in the broad sense) = opsin + chromophore. Among the many kinds of microbial opsins are the proton pumps bacteriorhodopsin (BR) and xanthorhodopsin (xR), the chloride pump halorhodopsin (HR), the photosensors sensory rhodopsin I (SRI) and sensory rhodopsin II (SRII), as well as proteorhodopsin (PR), Neurospora opsin I (NOPI), Chlamydomonas sensory rhodopsins A (CSRA), Chlamydomonas sensory rhodopsins B (CSRB), channelrhodopsin (ChR), and archaerhodopsin (Arch).[119]
Several microbal opsins, such as
Microbal opsins (like channelrhodopsin, halorhodopsin, and archaerhodopsin) are used in optogenetics to switch on or off neuronal activity. Microbal opsins are preferred if the neuronal activity should be modulated at higher frequency, because they respond faster than animal opsins. This is because microbal opsins are ion channels or proton/ion pumps and thus are activated by light directly, while animal opsins activate G-proteins, which then activate effector enzymes that produce metabolites to open ion channels.[121]
See also
- Retinylidene protein
- Visual cycle
- Visual phototransduction
- Microbial rhodopsin
- Channelrhodopsins
External links
- Illustration Archived 2020-01-09 at the Baldwin-Wallace College
- Opsin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
References
- ^ S2CID 38970721.
- ^ PMID 8170923.
- S2CID 4324074.
- S2CID 4352920.
- S2CID 4022911.
- PMID 16589696.
- PMID 13367054.
- .
- PMID 16589909.
- S2CID 1657759.
- S2CID 20407577.
- ^ PMID 10926528.
- ^ S2CID 4339970.
- S2CID 4152360.
- PMID 14351151.
- PMID 16590155.
- S2CID 45830716.
- ^ S2CID 4302421.
- ^ PMID 4877437.
- doi:10.1002/wmts.6.
- ^ PMID 35954284..
 Material was copied and adapted from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied and adapted from this source, which is available under a Creative Commons Attribution 4.0 International License - PMID 18370404.
- ISBN 978-0-12-185295-5.
- ^ S2CID 85819100.
- ^ S2CID 201420079.
- ^ PMID 32243853.
- ^ PMID 22012981.
- ^ PMID 28172965.
- ^ S2CID 17819587.
- PMID 26472700.
- S2CID 206530389.
- S2CID 1422764.
- PMID 35143663.
- ^ PMID 28598695.
- ^ PMID 23904486.
- S2CID 14008354.
- ^ PMID 12601165.
- PMID 31855179.
- PMID 7679072.
- PMID 10748160.
- ^ PMID 10037690.
- S2CID 4184319.
- ^ PMID 20852774.
- ^ PMID 15774036.
- S2CID 22879394.
- S2CID 36881435.
- ^ PMID 27861495.
- S2CID 6202186.
- S2CID 28746630.
- S2CID 24105079.
- ^ PMID 25267311.
- PMID 18344558.
- ^ PMID 10234000.
- PMID 22991144.
- ^ PMID 23554393.
- ^ PMID 22043319.
- PMID 14623103.
- PMID 24403072.
- PMID 20392722.
- ^ Yong E (20 May 2015). "Octopuses, and Maybe Squid, Can Sense Light With Their Skin". National Geographic. Archived from the original on February 23, 2021.
- PMID 25136094.
- ^ PMID 26154478.
- PMID 29303477.
- ^ PMID 31635694.
- ^ PMID 23112152.
- ^ PMID 19720651.
- ^ PMID 20219739.
- PMID 19720655.
- PMID 16213808.
- PMID 28289214.
- PMID 10581404.
- PMID 21416149.
- S2CID 4301315.
- S2CID 52984435.
- PMID 10821749.
- S2CID 41895317.
- PMID 9334384.
- PMID 15096614.
- PMID 26370232.
- S2CID 28604455.
- PMID 24041647.
- ^ PMID 11401433.
- S2CID 2583520.
- PMID 28623234.
- PMID 29669554.
- PMID 29809157.
- ^ PMID 26255845.
- PMID 19720648.
- PMID 23578808.
- PMID 23479626.
- ^ PMID 26491964.
- ^ PMID 23776409.
- ^ PMID 12670711.
- PMID 18832159.
- PMID 23227169.
- PMID 18160295.
- PMID 25062921.
- PMID 8327495.
- ^ PMID 21135214.
- PMID 26392540.
- PMID 25193307.
- PMID 9287291.
- S2CID 11669142.
- PMID 8258527.
- ^ Nagata T, Koyanagi M, Terakita A (20 October 2010). "Molecular Evolution and Functional Diversity of Opsin-Based Photopigments". Retrieved 7 May 2018.
- PMID 15961402.
- PMID 18474598.
- PMID 8639565.
- PMID 9275222.
- ISBN 978-1-4939-1468-5. Retrieved 3 September 2015.
- S2CID 84439732.
- PMID 15860584.
- PMID 2948499.
- PMID 23476135.
- PMID 25075528.
- PMID 21402729.
- PMID 23981446.
- PMID 24706784.
- PMID 23994288.
- PMID 17339603.
- PMID 22196724.



