Orcinol
| |
| Names | |
|---|---|
| Preferred IUPAC name
5-Methylbenzene-1,3-diol | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.007.259 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H8O2 | |
| Molar mass | 124.139 g·mol−1 |
| Appearance | Crystalline solid |
| Melting point | 109.0 °C (228.2 °F; 382.1 K)[2] |
| Boiling point | 291 °C (556 °F; 564 K)[2] |
| Miscible | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
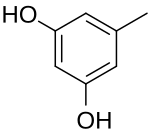
Orcinol is an organic compound with the formula CH3C6H3(OH)2. It occurs in many species of
Camponotus saundersi. It is a colorless solid. It is related to resorcinol
, 1,3-C6H4(OH)2.
Synthesis and reactions
Orcinol was first prepared by dehydroacetic acid, a conversion that involved ring-opening of the pyrone to a triketone. This early experiment helped establish the rich condensation chemistry of polyketides.[4] It can be obtained by fusing extract of aloes with potash,[5] followed by acidification.
It undergoes O-
dimethylsulfate.[6]
It is used in the production of the dye
Ferric chloride gives a bluish-violet coloration with the aqueous solution. Unlike resorcinol it does not give a fluorescein with phthalic anhydride. Oxidation of the ammoniacal solution gives orcein, C28H24N2O7, the chief constituent of the natural dye archil. 4-Methylcatechol is an isomer, found as its methyl ether (creosol) in beech-wood tar.[5]
Production from shale oil
Orcinol is also found in shale oil produced from Kukersite oil shale.[2] It is the main water-soluble phenol in the oil, and has been extracted and refined industrially by Viru Keemia Grupp.[7]
See also
References
- ^ Merck Index, 11th Edition, 6819.
- ^ ISSN 0021-9568.
- ^ Robiquet: „Essai analytique des lichens de l'orseille", Annales de chimie et de physique, 1829, 42, p. 236–257.
- PMID 11548049.
- ^ a b One or more of the preceding sentences incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Orcin". Encyclopædia Britannica. Vol. 20 (11th ed.). Cambridge University Press. p. 173.
- .
- ^ "Fine chemicals". Viru Keemia Grupp. Retrieved 2020-10-23.

