Organic peroxides

In
Types of organic peroxides
Organic peroxides are classified (i) by the presence or absence of a hydroxyl (-OH) terminus and (ii) by the presence of alkyl vs acyl substituents.[2]
- Examples of organic peroxides
-
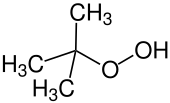
tert-Butyl hydroperoxide, a hydroperoxide (formula: ROOH), which is used to epoxide alkenes.
-
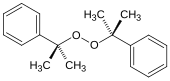
Dicumyl peroxide, a dialkyl peroxide (formula: ROOR), which is used to initiate polymerizations.
-
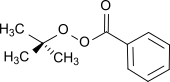
tert-butylperoxybenzoate, a peroxy ester (formula: RCO3R') that used as a radical initiator.
-
dibenzoyl peroxide, a diacyl peroxide (formula: (RCO2)2)) is also used as an initiator for polymerizations.
-
peroxycarboxylic acid (formula: (RCO3H), is a reagent in organic synthesis.
-
Prostaglandin G2, an endo peroxide, the precursor to other prostaglandins
One gap in the classes of organic peroxides is diphenyl peroxide. Quantum chemical calculations predict that it undergoes a nearly barrierless reaction akin to the
Properties
The O−O
Biology

Peroxides play important roles in biology. Hundreds of peroxides and hydroperoxides are known, being derived from fatty acids, steroids, and terpenes.[6] The prostaglandins are biosynthesized by initial formation of a bicyclic peroxide ("endoperoxide") derived from arachidonic acid.[7]
Many aspects of biodegradation or aging are attributed to the formation and decay of peroxides formed from oxygen in air. Countering these effects, an array of biological and artificial antioxidants destroy peroxides.
In

Industrial uses
In polymer chemistry
Many peroxides are used as a radical initiators, e.g., to enable polymerization of acrylates. Industrial resins based on acrylic and/or methacrylic acid esters are invariably produced by radical polymerization with organic peroxides at elevated temperatures.[9] The polymerization rate is adjusted by suitable choice of temperature and type of peroxide.[10]
Thermoplastic production techniques for many industrial polymerization applications include processes which are carried out in bulk, solution, or suspension type batches. Relevant polymers include: polyvinyl chloride (PVC), low-density polyethylene (LDPE), high-density polyethylene (HDPE), polymethyl methacrylate (PMMA), Polystyrene, and
Bleaching and disinfecting agents
Preparation
From hydrogen peroxide
Dialkyl peroxides, e.g., dicumyl peroxide, are synthesized by addition of hydrogen peroxide to alkenes or by O-alkylation of hydroperoxides.
Diacyl peroxides are typically prepared by treating hydrogen peroxide with acid chlorides or acid anhydrides in the presence of base:[1]
- H2O2 + 2 RCOCl → (RCO2)2 + 2 HCl
- H2O2 + (RCO)2O → (RCO2)2 + H2O
The reaction competes with hydrolysis of the acylating agent but the hydroperoxide anion is a superior nucleophile relative to hydroxide. Unsymmetrical diacyl peroxides can be produced by treating acyl chlorides with the peroxy acid.
From O2
Cyclic peroxides can be obtained by
The hazards associated with storage of ethers in air is attributed to the formation of hydroperoxides via the direct albeit slow reaction of triplet oxygen with
Reactions
Homolysis
Organic peroxides are widely used to initiate
- ROOR ⇌ 2 RO.
The tendency to homolyze is also exploited to modify polymers by
Self-oxidation
Especially when in concentrated form, organic peroxides can decompose by self-oxidation, since organic peroxides contain both an oxidizer (the O-O bond) and fuel (C-H and C-C bonds). A "self-accelerating decomposition" occurs when the rate of peroxide decomposition generates heat at a faster rate than it can be dissipated to the environment. Temperature is the main factor in the rate of decomposition. The lowest temperature at which a packaged organic peroxide will undergo a self-accelerating decomposition within a week is defined as the self-accelerating decomposition temperature (SADT). A large fire at the Arkema Chemical Plant in Crosby, Texas (USA) in 2017 was caused by the decomposition of various organic peroxides following power failure and subsequent loss of cooling systems.[14] This occurred due to extreme flooding from Hurricane Harvey, which destroyed main and back-up power generators at the site.[14]
Cumene process
Hydroperoxides are intermediates or reagents in major commercial processes. In the cumene process, acetone and phenol are produced by decomposition of cumene hydroperoxide (Me = methyl):
- C6H5CMe2(O2H) → C6H5OH + O=CMe2
Reduction
Organoperoxides can be reduced to alcohols with lithium aluminium hydride, as described in this idealized equation:
- 4 ROOH + LiAlH4 → LiAlO2 + 2 H2O + 4 ROH
The phosphite esters and tertiary phosphines also effect reduction:
- ROOH + PR3 → P(OR)3 + ROH
Cleavage to ketones and alcohols occurs in the base-catalyzed Kornblum–DeLaMare rearrangement, which involves the breaking of bonds within peroxides to form these products.
Some peroxides are drugs, whose action is based on the formation of radicals at desired locations in the organism. For example, artemisinin and its derivatives, such as artesunate, possess the most rapid action of all current drugs against falciparum malaria.[15] Artesunate is also efficient in reducing egg production in Schistosoma haematobium infection.[16]
Organic synthesis
tert-Butyl hydroperoxide is used for epoxidation and hydroxylation reagents in conjunction with metal catalysts.[17]
Analysis of peroxides
Several analytical methods are used for qualitative and quantitative determination of peroxides.
Quantitative analysis of hydroperoxides can be performed using potentiometric titration with lithium aluminium hydride.[22] Another way to evaluate the content of peracids and peroxides is the volumetric titration with alkoxides such as sodium ethoxide.[23]
Active oxygen in peroxides
Each peroxy group is considered to contain one active oxygen atom. The concept of active oxygen content is useful for comparing the relative
The term active oxygen is used to specify the amount of peroxide present in any organic peroxide formulation. One of the oxygen atoms in each peroxide group is considered "active". The theoretical amount of active oxygen can be described by the following equation:[24]
where p is the number of peroxide groups in the molecule, and m is the molecular mass of the pure peroxide.
Organic peroxides are often sold as formulations that include one or more
Safety

Peroxides are also strong oxidizers and easily react with skin, cotton and wood pulp.[25] For safety reasons, peroxidic compounds are stored in a cool, opaque container, as heating and illumination accelerate their chemical reactions. Small amounts of peroxides, which emerge from storage or reaction vessels are neutralized using reducing agents such as iron(II) sulfate. Safety measures in industrial plants producing large amounts of peroxides include the following:
1) The equipment is located within reinforced concrete structures with foil windows, which would relieve pressure and not shatter in case of explosion.
2) The products are bottled in small containers and are moved to a cold place promptly after the synthesis.
3) The containers are made of non-reactive materials such as stainless steel, some aluminium alloys or dark glass.[26]
For safe handling of concentrated organic peroxides, an important parameter is temperature of the sample, which should be maintained below the self accelerating decomposition temperature of the compound.[27]
The shipping of organic peroxides is restricted. The
| Chemical name | CAS Number
|
Prohibitions |
|---|---|---|
| Acetyl acetone peroxide | 37187-22-7 | > 9% by mass active oxygen
|
Acetyl benzoyl peroxide
|
644-31-5 | solid, or > 40% in solution |
| Ascaridole | 512-85-6 | (organic peroxide) |
| tert-Butyl hydroperoxide | 75-91-2 | > 90% in solution (aqueous) |
| Di-(1-naphthoyl)peroxide | 29903-04-6 | |
| Diacetyl peroxide | 110-22-5 | solid, or > 25% in solution |
| Ethyl hydroperoxide | 3031-74-1 | |
| Methyl ethyl ketone peroxide | 1338-23-4 | > 9% by mass active oxygen in solution |
| Methyl isobutyl ketone peroxide | 37206-20-5 | > 9% by mass active oxygen in solution |
See also
External links
- Organic Peroxide Producers Safety Division
- OSH Answers – organic peroxides
- "The Perils of Peroxides". carolina.com. Burlington, NC: Carolina Biological Supply Company. Archived from the original on 2007-12-18.
- European Organic Peroxide Safety Group
References
- ^ ISBN 978-3527306732.
- ISBN 9780470771730.
- ^ "Henry Rzepa's Blog: Why diphenyl peroxide does not exist".
- .
- ISBN 9780470771730.
- PMID 1522977.
- PMID 12797830.
- ISBN 1-84755-812-7
- ISBN 3-87870-569-7p. 67
- ^ Organische Peroxide für die Polymerisation Archived 2016-06-29 at the Wayback Machine. pergan.com (in German)
- PMID 12974650.
- ISBN 3-527-29985-8, p. 323
- .
- ^ a b USCSB. "Caught in the Storm: Extreme Weather Hazards". Youtube. USCSB. Retrieved 29 June 2023.
- PMID 9210658.
- S2CID 1675813.
- PMID 38259195.
- S2CID 101877371.
- .
- ^ Veibel, S. Analytik organischer Verbindungen, Akademie-Verlag, Berlin, 1960, p. 262
- .
- .
- .
- ^ "ASTM E298, Standard Test Methods for Assay of Organic Peroxides". ASTM. 2010.
- ISBN 3-527-29985-8pp. 741–762
- ^ Ozonelab Peroxide compatibility
- ^ "Product Stewardship". American Chemistry Council. 2021-01-17. Retrieved 2022-01-03.




![{\displaystyle A[\mathrm {O} ]_{\text{theoretical}}(\%)=16{\frac {p}{m}}\times 100,}](https://wikimedia.org/api/rest_v1/media/math/render/svg/176ebe20ba1c311442630b59225f66d03b08fe5f)