Organoaluminium chemistry
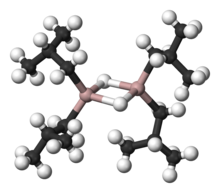
Organoaluminium chemistry is the study of compounds containing bonds between
History
The first organoaluminium compound (C2H5)3Al2I3 was discovered in 1859.
Structure and bonding
Aluminium(III) compounds
Organoaluminium compounds generally feature three- and four-coordinate Al centers, although higher

Ligand exchange in trialkylaluminium compounds
The trialkylaluminium dimers often participate in dynamic equilibria, resulting in the interchange of bridging and terminal ligands as well as ligand exchange between dimers. Even in noncoordinating
Low oxidation state organoaluminium compounds
The first organoaluminium compound with an Al-Al bond was reported in 1988 as (((Me3Si)2CH)2Al)2 (a dialane). They are typically prepared reduction of the dialkylaluminium chlorides by metallic potassium:[6]
- (R2AlCl)2 + 2 K → R2Al-AlR2 + 2 KCl
Another notable group of alanes are tetraalanes containing four Al(I) centres. These compounds adopt a
Preparation
From alkyl halides and aluminium
Industrially, simple aluminium alkyls of the type Al2R6 (R = Me, Et) are prepared in a two-step process beginning with the alkylation of aluminium powder:
- 2 Al + 3 CH3CH2Cl → (CH3CH2)3Al2Cl3
The reaction resembles the synthesis Grignard reagents. The product, (CH3CH2)3Al2Cl3, is called ethylaluminium sesquichloride. The term sesquichloride refers to the fact that, on average, the Cl:Al ratio is 1.5. These sesquichlorides can be converted to the triorganoaluminium derivatives by reduction:
- 2 (CH3CH2)3Al2Cl3 + 6 Na → (CH3CH2)6Al2 + 2 Al + 6 NaCl
This method is used for production of trimethylaluminium and triethylaluminium.[7]
The overall reaction for the production of these simple alkylaluminium compounds is thus as follows:
- 2Al + 6RX + 6M → Al2R6 + 6MX (where M is an alkali metal and X is a halogen)
Hydroalumination
Aluminium powder reacts directly with certain terminal alkenes in the presence of hydrogen. The process entails two steps, the first producing dialkylaluminium hydrides. Such reactions are typically conducted at elevated temperatures and require activation by trialkylaluminium reagents:
- 6 Al + 3 H2 + 12 CH2=CHR → 2 [HAl(CH2CHR)2]3
For nonbulky R groups, the organoaluminium hydrides are typically trimeric. In a subsequent step, these hydrides are treated with more alkene to effect hydroalumiunation:
- 2 [HAl(CH2CHR)2]3 + 3 CH2=CHR → 3 [Al2(CH2CHR)3
Diisobutylaluminium hydride, which is dimeric, is prepared by hydride elimination from triisobutylaluminium:
- 2 i-Bu3Al → (i-Bu2AlH)2 + 2 (CH3)2C=CH2
Carboalumination
Organoaluminum compounds can react with alkenes and alkynes, resulting in the net addition of one organyl group and the metal fragment across the multiple bond (carboalumination). This process can proceed in a purely thermal manner or in the presence of a transition metal catalyst. For the uncatalyzed process, monoaddition is only possible when the alkene is substituted. For ethylene, carboalumination leads to a
The methylalumination of alkynes in the presence of Cp2ZrCl2[10][11] is employed for the synthesis of stereodefined trisubstituted olefin fragments, a common substructure in terpene and polyketide natural products. The synthesis of (E)-4-iodo-3-methylbut-3-en-1-ol[12] shown below is a typical application of this reaction:
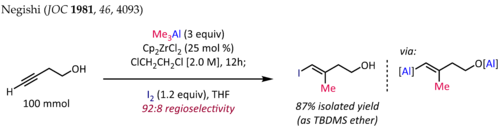
For terminal alkynes, the reaction generally proceeds with good regioselectivity (>90:10 rr) and complete syn selectivity, even in the presence of propargylic or homopropargylic heteroatom substituents. Unfortunately, extension of the zirconocene-catalyzed methylalumination to alkylalumination with higher alkyls results in lower yields and poor regioselectivities.
Laboratory preparations
Although the simple members are commercially available at low cost, many methods have been developed for their synthesis in the laboratory, including metathesis or transmetalation.
- Metathesis of aluminium trichloride with RLi or RMgX gives the trialkyl:
- AlCl3 + 3 BuLi → Bu3Al + 3 LiCl
- Transmetalation:
- 2 Al + 3 HgPh2 → 2 AlPh3 + 3 Hg
Reactions
The high reactivity of organoaluminium compounds toward electrophiles is attributed to the charge separation between aluminium and carbon atom.
Lewis acidity
Organoaluminium compounds are
Electrophiles
The Al–C bond is polarized such that the carbon is highly basic. Acids react to give alkanes. For example, alcohols give alkoxides:
- AlR'3 + ROH → 1/n (R'2Al−OR)n + R'H
A wide variety of acids can be employed beyond the simple mineral acids. Amines give amido derivatives. With carbon dioxide, trialkylaluminium compounds give the dialkylaluminium carboxylate, and subsequently alkyl aluminium dicarboxylates:
- AlR3 + CO2 → R2AlO2CR
- R2AlO2CR + CO2 → RAl(O2CR)2
The conversion is reminiscent of the carbonation of Grignard reagents.[13][14][15]
Similarly, the reaction between trialkylaluminum compounds and carbon dioxide has been used to synthesise alcohols, olefins,[13] or ketones.[16]
With oxygen one obtains the corresponding alkoxides, which can be hydrolysed to the alcohols:
- AlR3 + 3/2 O2 → Al(OR)3
A structurally characterized organo
The reaction between pure trialalkylaluminum compounds and
Applications
Organoaluminium compounds are widely used in the production of alkenes, alcohols, and polymers. Some relevant processes include the
References
- ISBN 978-0199264636.
- ^ M. Witt; H. W. Roesky (2000). "Organoaluminum chemistry at the forefront of research and development" (PDF). Curr. Sci. 78 (4): 410. Archived from the original (PDF) on 2014-10-06.
- .
- ISBN 978-3-527-29390-2.
- ISBN 978-0-471-02775-1.
- ISBN 9780120311514.
- ISBN 9780080405957.
- .
- PMID 20465291.
- ISBN 978-0471984160.
- .
- ^ .
- .
- ^ Zakharkin, L.I.; Gavrilenko, V.V.; Ivanov, L.L. (1967). Zh. Obshch. Khim. 377: 992.
{{cite journal}}: Missing or empty|title=(help) - Continental Oil
- PMID 18283706.
- ^ Cameo Chemicals SDS
- ^ Handling Chemicals Safely 1980. p. 929
- ISBN 9783527306732.
