Organophosphate

In
Like most functional groups, organophosphates occur in a diverse range of forms,[2] with important examples including key biomolecules such as DNA, RNA and ATP, as well as many insecticides, herbicides, nerve agents and flame retardants. OPEs have been widely used in various products as flame retardants, plasticizers, and performance additives to engine oil. The low cost of production and compatibility to diverse polymers made OPEs to be widely used in industry including textile, furniture, electronics as plasticizers and flame retardants. These compounds are added to the final product physically rather than by chemical bond.[3] Due to this, OPEs leak into the environment more readily through volatilization, leaching, and abrasion.[4] OPEs have been detected in diverse environmental compartments such as air, dust, water, sediment, soil and biota samples at higher frequency and concentration.[1][4]
The popularity of OPEs as flame retardants came as a substitution for the highly regulated brominated flame retardants.[5]
Forms
Organophosphates are a class of compounds encompassing a number of distinct but closely related
In the context of pesticides, derivatives of organophosphates such as
In biology the esters of
Synthesis
- Alcoholysis of POCl3
- O=PCl3 + 3 ROH → O=P(OR)3 + 3 HCl
When aliphatic alcohols are used the HCl by-product can react with the phosphate esters to give
- O=P(OR)3 + HCl → O=P(OR)2OH + RCl
This reaction is usually undesirable and is exacerbated by high reaction temperatures. It can be inhibited by the use of a base or the removal of HCl through sparging.
- Esterification of phosphoric acid and P2O5
- OP(OH)3 + ROH → OP(OH)2(OR) + H2O
- Oxidation of phosphite esters
- P(OR)3 + [O] → OP(OR)3
- Phosphorylation
The formation of organophosphates is an important part of biochemistry and living systems achieve this using a variety of
Properties
Bonding
The bonding in organophosphates has been a matter of prolonged debate; the phosphorus atom is classically
Acidity
Phosphate esters bearing P-OH groups are acidic. The pKa of the first OH group is typically between 1-2, with the second OH deprotonating at a pKa of 6-7.[20] This is great practical importance as it means that phosphate mono- and di-esters are negatively charged at physiological pH (due to deprotonation).[21] This includes biomolecules such as DNA and RNA. The presence of this negative charge makes these compound much more water soluble, and also more resistant to degradation by hydrolysis or other forms of nucleophilic attack.[22]
Water solubility
The water solubility of organophosphates is an important factor in biological, industrial and environmental settings. The wide variety of substitutes used in organophosphate esters results in great variations in physical properties. OPEs exhibit a wide range of octanol/water
Industrial materials
Pesticides
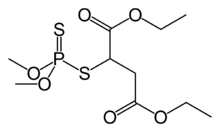
Organophosphates are best known for their use as pesticides. The vast majority are
The development of organophosphate insecticides dates back to the 1930s and is generally credited to
- Acephate
- Azinphos-methyl
- Bensulide
- Chlorethoxyfos
- Coumaphos
- Diazinon
- Dichlorvos
- Dicrotophos
- Dimethoate
- Disulfoton
- Ethion
- Ethoprop
- Ethyl parathion
- Fenamiphos
- Fenitrothion
- Fonofos
- Isoxathion
- Malathion
- Methamidophos
- Methidathion
- Mevinphos
- Naled
- Phosmet
- Profenofos
- Propetamphos
- Quinalphos
- Sulfotep
- Tebupirimfos
- Temephos
- Terbufos
- Tetrachlorvinphos
- Triazofos
Organophosphate insecticides are acetylcholinesterase inhibitors, and when introduced to an organism they act to fatally disrupt the transmission of nerve signals. The risk of human death through organophosphate poisoning[32] was obvious from the start and let to efforts to lower toxicity against mammals while not reducing efficacy against insects.[33][34]
The majority of organophosphate insecticides are
This selectivity is far from perfect and organophosphate insecticides remain acutely toxic to humans, with many thousands estimated to be killed each year due to intentional (suicide)[42] or unintentional poisoning. Beyond their acute toxicity, exposure to organophosphates is associated with a number of heath risks, including organophosphate-induced delayed neuropathy (muscle weakness) and developmental neurotoxicity.[28][43][44] There is limited evidence that certain compounds cause cancer, including malathion and diazinon.[45] Children[46] and farmworkers[47] are considered to be at greater risk.
Pesticide regulation in the United States and the regulation of pesticides in the European Union have both been increasing restrictions on organophosphate pesticides since the 1990s, particularly when used for crop protection. The use of organophosphates has decreased considerably since that time, having been replaced by pyrethroids and neonicotinoids, which are effective a much lower levels.[48] Reported cases of organophosphate poisoning in the US have reduced during this period.[49][50] Regulation in the global south can be less extensive.[51][52]
In 2015, only 3 of the 50 most common crop-specific pesticides used in the US were organophosphates (
Flame retardants
Flame retardants are added to materials to prevent combustion and to delay the spread of fire after ignition. Organophosphate flame retardants are part of a wider family of phosphorus-based agents which include organic phosphonate and phosphinate esters, in addition to inorganic salts.[58][59] When some prominent brominated flame retardant were banned in the early 2000s phosphorus-based agents were promoted as safer replacements. This has led to a large increase in their use, with an estimated 1 million tonnes of organophosphate flame retardants produced in 2018.[60] Safety concerns have subsequently been raised about some of these reagents,[61][62] with several under regulatory scrutiny.[63][64]
Organophosphate flame retardants were first developed in the first half of the twentieth century in the from of
which are commonly used to make casing for electrical items like TVs, computers and home appliances.Organophosphates act multifunctionally to retard fire in both the gas phase and condensed (solid) phase. Halogenated organophosphates are more active overall as their degradation products interfere with combustion directly in the gas phase. All organophosphates have activity in the condensed phase, by forming phosphorus acids which promote char formation, insulating the surface from heat and air.
Organophosphates were originally thought to be a safe replacements for brominated flame retardants, however many are now coming under regulatory pressure due to their apparent health risks.[64][67][68] The chlorinated organophosphates may be carcinogenic, while others such as tricresyl phosphate have necrotoxic properties.[69] Bisphenol-A bis(diphenyl phosphate) can hydrolyse to form
Plasticisers

Plasticisers are added to polymers and plastics to improve their flexibility and processability, giving a softer more easily deformable material. In this way brittle polymers can be made more durable. Organophosphates find use because they are multifunctional; primarily plasticising but also imparting flame resistance. The most frequently plasticised polymers are the vinyls (
Pure PVC is more than 60% chlorine by mass and difficult to burn, but its flammability increases the more it is plasticised.
Hydraulic fluids and lubricant additives
Similar to their use as plastisiers, organophosphates are well suited to use as hydraulic fluids due to their low freezing points and high boiling points, fire-resistance, non-corrosiveness, excellent boundary lubrication properties and good general chemical stability. The triaryl phosphates are the most important group, with tricresyl phosphate being the first to be commercialised in the 1940s, with trixylyl phosphate following shortly after. Butylphenyl diphenyl phosphate and propylphenyl diphenyl phosphate became available after 1960.[79]
In addition to their use as hydraulic base-stock, organophosphates (tricresyl phosphate) and metal organothiophosphates (zinc dithiophosphate) are used as both an antiwear additives and extreme pressure additives in lubricants, where they remain effective even at high temperatures.[80][81][82]
Metal extractants
Organophosphates have long been used in the field of extractive metallurgy to liberate valuable rare earths from their ores.[83] Di(2-ethylhexyl)phosphoric acid and tributyl phosphate are used for the liquid–liquid extraction of these elements from the acidic mixtures form by the leaching of mineral deposits.[84] These compounds are also used for the PUREX (plutonium uranium reduction extraction) process, which is used for nuclear reprocessing.[85]
Surfactants
Mono- and di- phosphate esters of alcohols or alcohol
Nerve agents
Although the first phosphorus compounds observed to act as cholinesterase inhibitors were organophosphates,
In nature
The detection of OPEs in the air as far away as Antarctica at concentrations around 1 ng/m3 suggests their persistence in air, and their potential for long-range transport.[24] OPEs were measured in high frequency in air and water and widely distributed in northern hemisphere.[93][94] The chlorinated OPEs (TCEP, TCIPP, TDCIPP) in urban sampling sites and non-halogenated like TBOEP in rural areas respectively were frequently measured in the environment across multiple sites. In the Laurentian Great Lakes total OPEs concentrations were found to be 2–3 orders of magnitude higher than concentrations of brominated flame retardants measured in similar air.[94] Waters from rivers in Germany, Austria, and Spain have been consistently recorded for TBOEP and TCIPP at highest concentrations.[24] From these studies, it is clear that OPE concentrations in both air and water samples are often orders of magnitude higher than other flame retardants, and that concentrations are largely dependent on sampling location, with higher concentrations in more urban, polluted locations.
References
- ^ PMID 27322497.
- .
- ^ PMID 30690428.
- ^ PMID 25290907.
- ^ PMID 22537891.
- .
- S2CID 105424570.
- S2CID 96234775.
- S2CID 105424570.
- S2CID 58665691.
- PMID 37307603.
- S2CID 73451989.
- PMID 24013652.
- .
- ^ PMID 19378976.
- S2CID 234836388.
- .
- .
- .
- .
- .
- .
- PMID 22332897.
- ^ S2CID 19824807.
- S2CID 206579583.
- ^ "Status and Trends of Pesticide Use". United Nations Environment Programme. World Health Organization, & Food and Agriculture Organization of the United Nations. 2022.
- ^ PMID 34903838.
- ^ PMID 29228398.
- ISSN 0009-2347.
- )
- ISBN 978-3-030-81952-1.
- PMID 25425841.
- .
- PMID 27976502.
- PMID 13058919.
- S2CID 84496732.
- .
- PMID 14389245.
- .
- .
- PMID 13730019.
- PMID 28535450.
- S2CID 254871617.
- ^ "The environmental, human health and economic impacts of pesticides" (PDF). United Nations Environment Programme [UNEP]. Retrieved 2 January 2024.
- PMID 31829533.
- PMID 24121005.
- PMID 27128815.
- ^ "Status and Trends of Pesticide Use". United Nations Environment Programme. World Health Organization, & Food and Agriculture Organization of the United Nations. 2022.
- PMID 22251442.
- PMID 19379510.
- S2CID 258033572.
- .
- PMID 31515508.
- ^ "Tolerance Revocations: Chlorpyrifos". www.regulations.gov. Retrieved 2 January 2024.
Federal Register Number: 2021-18091
- PMID 33132734.
- S2CID 10379060.
- ISBN 978-3-540-67625-6.
- ^ PMID 22537891.
- .
- PMID 28942249.
- PMID 32494578.
- S2CID 195694322.
- ^ "ECHA identifies certain brominated flame retardants as candidates for restriction". echa.europa.eu. European Chemicals Agency. Retrieved 3 January 2024.
- ^ doi:10.2823/854233
- ISBN 978-0-471-48494-3.
- doi:10.1002/pi.2290.
- PMID 32494578.
- S2CID 195694322.
- PMID 10596299.
- S2CID 24611076.
- S2CID 257638565.
- PMID 26851706.
- ^ ISBN 3527306730.
- .
- ISBN 978-3-446-40801-2.
- S2CID 247663661.
- ISBN 978-0-470-25354-0.
- doi:10.1002/pat.645.
- S2CID 235095497. Retrieved 2024-01-07.
- S2CID 93027894.
- .
- S2CID 246434462.
- S2CID 104245067.
- .
- S2CID 94173845.
- ^ ISBN 978-1-4051-7179-3. Retrieved 2023-05-27.
- S2CID 105424570.
- ISBN 978-0-8031-0413-6. Retrieved 2023-05-27.
- .
- PMID 21105582.
- S2CID 49661943.
- S2CID 21659617.
- .
- ^ PMID 25045802.



