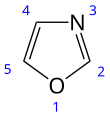
Oxazole
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,3-Oxazole[1] | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 103851 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.005.474 | ||
| EC Number |
| ||
| 485850 | |||
| MeSH | D010080 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H3NO | |||
| Molar mass | 69.06 g/mol | ||
| Density | 1.050 g/cm3 | ||
| Boiling point | 69.5 °C (157.1 °F; 342.6 K) | ||
| Acidity (pKa) | 0.8 (of conjugate acid)[2] | ||
| Hazards | |||
| GHS labelling:[3] | |||
 
| |||
| Danger | |||
| H225, H318 | |||
| P210, P233, P240, P241, P242, P243, P264+P265, P280, P303+P361+P353, P305+P354+P338, P317, P370+P378, P403+P235, P501 | |||
| Supplementary data page | |||
| Oxazole (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Oxazole is the parent compound for a vast class of
Preparation
The classic synthetic route the Robinson–Gabriel synthesis by dehydration of 2-acylaminoketones:

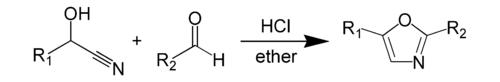
The Fischer oxazole synthesis from cyanohydrins and aldehydes is also widely used:
Other methods are known including the reaction of α-
.Biosynthesis
In

Where X = H, CH
3 for serine and threonine respectively, B = base.
(1) Enzymatic cyclization. (2) Elimination. (3) [O] = enzymatic oxidation.
Oxazoles are not as abundant in biomolecules as the related thiazoles with oxygen replaced by a sulfur atom.
Reactions
With a pKa of 0.8 for the conjugate acid (oxazolium salts), oxazoles are far less basic than imidazoles (pKa = 7).
Nucleophilic aromatic substitution takes place with leaving groups at C2.
Diels–Alder reactions involving oxazole (as dienes) and electrophilic alkenes has been well developed as a route to pyridines. In this way, alkoxy-substituted oxazoles serve a precursors to the pyridoxyl system, as found in vitamin B6. The initial cycloaddition affords a bicyclic intermediate, with an acid-sensitive oxo bridgehead.

In the Cornforth rearrangement of 4-acyloxazoles is a thermal rearrangement reaction with the organic acyl residue and the C5 substituent changing positions.
- Various oxidation reactions. One study[7] reports on the oxidation of 4,5-diphenyloxazole with 3 equivalents of CAN to the corresponding imide and benzoic acid:
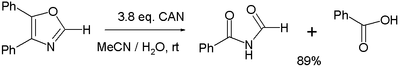
- In the balanced half-reaction three equivalents of water are consumed for each equivalent of oxazoline, generating 4 protons and 4 electrons (the latter derived from CeIV).
See also
- Isoxazole, an analog with the nitrogen atom in position 2.
- Thiazole, an analog with the oxygen replaced by a sulfur.
- Benzoxazole, where the oxazole is fused to a benzene ring.
- Oxazoline, which has one double bond reduced.
- Oxazolidine, which has both double bonds reduced.
- Oxazolone, an analog with a carbonyl group
Additional reading
- Fully Automated Continuous Flow Synthesis of 4,5-Disubstituted Oxazoles Marcus Baumann, Ian R. Baxendale,
References
- ISBN 978-0-85404-182-4.
- ^ Zoltewicz, J. A. & Deady, L. W. Quaternization of heteroaromatic compounds. Quantitative aspects. Adv. Heterocycl. Chem. 22, 71-121 (1978).
- ^ "Oxazole". pubchem.ncbi.nlm.nih.gov.
- ^ ISBN 0-582-01421-2.
- PMID 10331285.
- ISBN 978-3-527-30673-2.