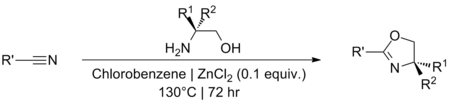Oxazoline

| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
4,5-Dihydro-1,3-oxazole | |
| Other names
Δ2-oxazoline
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.007.274 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3H5NO | |
| Molar mass | 71.079 g·mol−1 |
| Density | 1.075unit?[1] |
| Boiling point | 98 °C (208 °F; 371 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Oxazoline is a five-membered
Oxazoline itself has no applications however oxazolines have been widely investigated for potential applications. These applications include use as
Isomers
 |
Three .
A fourth isomer exists in which the O and N atoms are adjacent, this is known as isoxazoline. |
Synthesis
The synthesis of 2-oxazoline rings is well established and in general proceeds via the cyclisation of a 2-
From carboxylic acids
The usual route to oxazolines entails reaction of acyl chlorides with 2-amino alcohols. Thionyl chloride is commonly used to generate the acid chloride in situ, care being taken to maintain anhydrous conditions, as oxazolines can be ring-opened by chloride if the imine becomes protonated.[8] The reaction is typically performed at room temperature. If reagents milder than SOCl2 are required, oxalyl chloride can be used.[9] Aminomethyl propanol is a popular precursor amino alcohol.[10][11]
Modification of the
From aldehydes
The cyclisation of an amino alcohol and an
From nitriles
The use of catalytic amounts of
Applications
Ligands
Ligands containing a chiral 2-oxazoline ring are used in
2-Substituted oxazolines possess a moderately
Major classes of oxazoline based ligand include:
Notable specialist oxazoline ligands include:
- Tris-oxazolines (TRISOX)
- Bis(oxazolinato)s
- Trisoxazolinylborate ligands
Polymers
Some 2-oxazolines, such as
Analysis of fatty acids
The dimethyloxazoline (DMOX) derivatives of fatty acids are amenable to analysis by gas chromatography.
See also
Structural analogues
- Benzoxazole: where the oxazoline is fused onto a benzene ring.
- Oxazole: which has two double bonds
- Oxazolidine: which has no double bonds
- Thiazoline: where the oxygen is replaced by sulphur
Other pages
- Aminorex a drug bearing an oxazoline ring
References
- ^ .
- .
- .
- PMID 11667224.
- ^ S2CID 95217957.
- .
- .
- .
- .
- .
- S2CID 235855642.
- .
- .
- .
- .
- .
- .
- PMID 12691592.
- doi:10.3987/S-1981-01-0361 (inactive 2024-03-07).)
{{cite journal}}: CS1 maint: DOI inactive as of March 2024 (link - S2CID 95389965.
- PMID 24201227. See the Supplementary Information for details
- PMID 15352789.
- PMID 19378971.
- doi:10.1039/B417389A.
- ^ Greene, T. W. (1991). Protective groups in organic synthesis, 2nd ed. New York: Wiley. pp. 265–266 & 433–436.
- .
- PMID 19768817.
- PMID 17904246.
- PMID 22811405.