Oxidizing agent

An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or
In one sense, an oxidizing agent is a chemical species that undergoes a chemical reaction in which it gains one or more electrons. In that sense, it is one component in an oxidation–reduction (redox) reaction. In the second sense, an oxidizing agent is a chemical species that transfers electronegative atoms, usually oxygen, to a substrate. Combustion, many explosives, and organic redox reactions involve atom-transfer reactions.
Electron acceptors
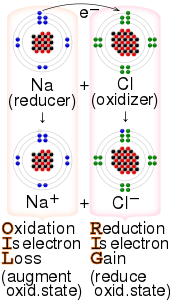
Extensive tabulations of ranking the electron accepting properties of various reagents (redox potentials) are available, see Standard electrode potential (data page).
Atom-transfer reagents
In more common usage, an oxidizing agent transfers oxygen atoms to a substrate. In this context, the oxidizing agent can be called an oxygenation reagent or oxygen-atom transfer (OAT) agent.
4 (perchlorate). Notice that these species are all oxides
In some cases, these oxides can also serve as electron acceptors, as illustrated by the conversion of MnO−
4 to MnO2−
4,ie permanganate to manganate.
Common oxidizing agents
- Oxygen (O2)
- Ozone (O3)
- Hydrogen peroxide (H2O2) and other inorganic peroxides, Fenton's reagent
- Fluorine (F2), chlorine (Cl2), and other halogens
- black powder
- Potassium chlorate (KClO3)
- Peroxydisulfuric acid (H2S2O8)
- Peroxymonosulfuric acid (H2SO5)
- Hypochlorite, chlorite, chlorate, perchlorate, and other analogous halogen oxyanions
- Fluorides of chlorine, bromine, and iodine
- Hexavalent chromium compounds such as chromic and dichromic acids and chromium trioxide, pyridinium chlorochromate (PCC), and chromate/dichromate compounds such as Sodium dichromate (Na2Cr2O7)
- Permanganate compounds such as potassium permanganate (KMnO4)
- Sodium perborate ([Na+
]
2·[B
2O
4(OH)
4]2−
) - Nitrous oxide (N2O), Nitrogen dioxide/Dinitrogen tetroxide (NO2 / N2O4)
- Sodium bismuthate (NaBiO3)
- Cerium (IV) compounds such as ceric sulfate
- Lead dioxide (PbO2)
Dangerous materials definition
The dangerous goods definition of an oxidizing agent is a substance that can cause or contribute to the combustion of other material.[4] By this definition some materials that are classified as oxidizing agents by analytical chemists are not classified as oxidizing agents in a dangerous materials sense. An example is potassium dichromate, which does not pass the dangerous goods test of an oxidizing agent.
The
Common oxidizing agents and their products
| Agent | Product(s) |
|---|---|
| O2 oxygen | Various, including the oxides H2O and CO2 |
| O3 ozone | Various, including ketones, aldehydes, and H2O; see ozonolysis |
| F2 fluorine | F− |
| Cl2 chlorine | Cl− |
| Br2 bromine | Br− |
| I2 iodine | I−, I− 3 |
| ClO− hypochlorite | Cl−, H2O |
| ClO− 3 chlorate |
Cl−, H2O |
| HNO3 nitric acid | NO nitric oxide NO2 nitrogen dioxide |
| SO2 sulfur dioxide | S sulfur (Claus process, ultramarine production, more commonly reducing agent) |
| Hexavalent chromium CrO3 dichromate
|
Cr3+, H2O |
| MnO− 4 permanganate MnO2− 4 manganate |
Mn2+ (acidic) or MnO2 (basic) |
| SbF5 antimony pentafluoride | SbF6- hexafluoroantimonate or SbF3 antimony trifluoride
|
| PtF6 platinum hexafluoride | PtF6- hexafluoroplatinate |
| RuO 4 ruthenium tetroxide OsO 4 osmium tetroxide |
in organic lab scale synthesis |
| H2O2, other peroxides | Various, including oxides and H2O |
| Tl(III) thallic compounds | Tl(I) thallous compounds, in organic lab scale synthesis |
See also
- Combustion – Chemical reaction between a fuel and oxygen
- Dye – Soluble chemical substance or natural material which can impart color to other materials
- Electron acceptor – Chemical entity capable of accepting electrons
- Electron donor – Chemical entity capable of donating electrons to another entity
- Electrosynthesis – Synthesis of chemical compounds in an electrochemical cell
- Organic oxidation– Redox reaction that takes place with organic compounds
- Organic redox reaction – Redox reaction that takes place with organic compounds
- Reducing agent – Chemical species that donates an electron to another species in a redox reaction
- Solvated electron – Free electron in a solution, often liquid ammonia
