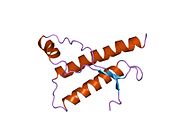
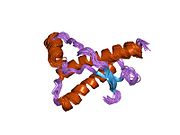
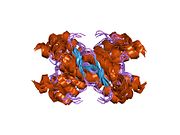
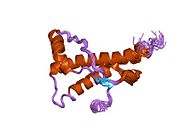
Major prion protein
| View/Edit Human | View/Edit Mouse |
Major prion protein (PrP) is encoded in the human body by the PRNP gene also known as CD230 (cluster of differentiation 230).[5][6][7][8] Expression of the protein is most predominant in the nervous system but occurs in many other tissues throughout the body.[9][10][11]
The protein can exist in multiple
Gene

The human PRNP gene is located on the short (p) arm of
Structure
PrP is highly conserved through mammals, lending credence to application of conclusions from test animals such as mice.
The primary sequence of PrP is 253 amino acids long before post-translational modification. Signal sequences in the amino- and carboxy- terminal ends are removed posttranslationally, resulting in a mature length of 208 amino acids. For human and golden hamster PrP, two glycosylated sites exist on helices 2 and 3 at Asn181 and Asn197. Murine PrP has glycosylation sites as Asn180 and Asn196. A disulfide bond exists between Cys179 of the second helix and Cys214 of the third helix (human PrPC numbering).
PrP messenger RNA contains a pseudoknot structure (prion pseudoknot), which is thought to be involved in regulation of PrP protein translation.[15]
Ligand-binding
The mechanism for conformational conversion to the scrapie isoform is speculated to be an elusive ligand-protein, but, so far, no such compound has been identified. However, a large body of research has developed on candidates and their interaction with the PrPC.[16]
PrPC (normal cellular) isoform
Although the precise function of PrP is not yet known, it is possibly involved in the transport of
PrPSc (scrapie) isoform
PrPSc is a conformational isoform of PrPC, but this orientation tends to accumulate in compact,
The propagation of PrPSc is a topic of great interest, as its accumulation is a pathological cause of
Polymorphisms at sites 136, 154, and 171 are associated with varying susceptibility to ovine scrapie. (These ovine sites correspond to human sites 133, 151, and 168.) Polymorphisms of the PrP-VRQ form and PrP-ARQ form are associated with increased susceptibility, whereas PrP-ARR is associated with resistance. The National Scrapie Plan of the UK aims to breed out these scrapie polymorphisms by increasing the frequency of the resistant allele.[28] However, PrP-ARR polymorphisms are susceptible to atypical scrapie, so this may prove unfruitful.
Function
Nervous system
The strong association to neurodegenerative diseases raises many questions of the function of PrP in the brain. A common approach is using PrP-knockout and
As the null mice age, a marked loss of
Memory
While null mice exhibit normal learning ability and
Further support for PrP's role in memory formation is derived from several population studies. A test of healthy young humans showed increased long-term memory ability associated with an MM or MV genotype when compared to VV.[39] Down syndrome patients with a single valine substitution have been linked to earlier cognitive decline.[40] Several polymorphisms in PRNP have been linked with cognitive impairment in the elderly as well as earlier cognitive decline.[41][42][43] All of these studies investigated differences in codon 129, indicating its importance in the overall functionality of PrP, in particular with regard to memory.
Neurons and synapses
PrP is present in both the pre- and post-synaptic compartments, with the greatest concentration in the pre-synaptic portion.
Some research indicates PrP involvement in neuronal development, differentiation, and neurite outgrowth. The PrP-activated signal transduction pathway is associated with axon and dendritic outgrowth with a series of kinases.[27][48]
Immune system
Though most attention is focused on PrP's presence in the nervous system, it is also abundant in immune system tissue. PrP immune cells include hematopoietic stem cells, mature lymphoid and myeloid compartments, and certain
Muscles, liver, and pituitary
PrP-null mice provide clues to a role in muscular physiology when subjected to a forced swimming test, which showed reduced locomotor activity. Aging mice with an overexpression of PRNP showed significant degradation of muscle tissue.
Though present, very low levels of PrP exist in the liver and could be associated with liver fibrosis. Presence in the pituitary has been shown to affect neuroendocrine function in amphibians, but little is known concerning mammalian pituitary PrP.[16]
Cellular
Varying expression of PrP through the cell cycle has led to speculation on involvement in development. A wide range of studies has been conducted investigating the role in cell proliferation, differentiation, death, and survival.[16] Engagement of PrP has been linked to activation of signal transduction.
Modulation of signal transduction pathways has been demonstrated in cross-linking with antibodies and ligand-binding (hop/STI1 or copper).
Diseases caused by PrP misfolding
More than 20 mutations in the PRNP gene have been identified in people with inherited
- Creutzfeldt–Jakob disease – glutamic acid-200 is replaced by lysine while valine is present at amino acid 129
- fatal familial insomnia – aspartic acid-178 is replaced by asparagine while methionine is present at amino acid 129[53]
The conversion of PrPC to PrPSc conformation is the mechanism of transmission of fatal, neurodegenerative transmissible spongiform encephalopathies (TSE). This can arise from genetic factors, infection from external source, or spontaneously for reasons unknown. Accumulation of PrPSc corresponds with progression of neurodegeneration and is the proposed cause. Some PRNP mutations lead to a change in single amino acids (the building-blocks of proteins) in the prion protein. Others insert additional amino acids into the protein or cause an abnormally short protein to be made. These mutations cause the cell to make prion proteins with an abnormal structure. The abnormal protein PrPSc accumulates in the brain and destroys nerve cells, which leads to the mental and behavioral features of prion diseases.
Several other changes in the PRNP gene (called polymorphisms) do not cause prion diseases but may affect a person's risk of developing these diseases or alter the course of the disorders. An allele that codes for a PRNP variant, G127V, provides resistance to kuru.[54]
In addition, some prion diseases can be transmitted from external sources of PrPSc.[55]
- Scrapie – fatal neurodegenerative disease in sheep, not transmissible to humans
- Bovine spongiform encephalopathy (mad-cow disease) – fatal neurodegenerative disease in cows, which can be transmitted to humans by ingestion of brain, spinal, or digestive tract tissue of an infected cow
- Kuru – TSE in humans, transmitted via funerary cannibalism. Generally, affected family members were given, by tradition, parts of the central nervous system according to ritual when consuming deceased family members.
Alzheimer's disease
PrPC protein is one of several cellular receptors of soluble
In humans, the
Research
In 2006 the production of cattle lacking PrPC form of the major prion protein (PrP) protein was reported which were resistant to prion propagation with no apparent developmental abnormalities. Besides the study of bovine products free of prion proteins another use could be so that human pharmaceuticals can be made in their blood without the danger that those products might get contaminated with the infectious agent that causes mad cow.[61][62]
Interactions
A strong
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000171867 - Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000079037 - Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- PMID 3755672.
- PMID 3094007.
- PMID 3014653.
- PMID 2877664.
- PMID 11357156.
- S2CID 20992257.
- ^ PMID 18073096.
- PMID 21987789.
- PMID 7837269.
- S2CID 10064966.
- PMID 11160898.
- ^ PMID 18391177.
- ^ PMID 26104335.
- S2CID 45647133.
- S2CID 24329326.
- S2CID 205383483.
- S2CID 39791520.
- PMID 34433091.
- PMID 35831275.
- PMID 35831291.
- S2CID 4399936.
- PMID 8100741.
- ^ PMID 18558863.
- PMID 11688751.
- PMID 14522848.
- S2CID 23409873.
- S2CID 37511702.
- PMID 10521590.
- PMID 1346338.
- S2CID 42892475.
- S2CID 2618712.
- PMID 20133875.
- S2CID 17164351.
- S2CID 5575951.
- PMID 15987701.
- S2CID 21049364.
- S2CID 11352163.
- S2CID 31538672.
- S2CID 23642220.
- PMID 10516306.
- S2CID 24917999.
- S2CID 2637074.
- S2CID 26307975.
- PMID 19242475.
- PMID 16968391.
- PMID 15354870.
- S2CID 22688729.
- S2CID 31976428.
- S2CID 20822956.
- PMID 19923577.
- "Brain disease 'resistance gene' evolves in Papua New Guinea community; could offer insights into CJD". ScienceDaily (Press release). November 21, 2009.
- PMID 19308092.
- ^ PMID 23948943.
- ^ PMID 25343100.
- ^ PMID 19242475.
- ^ S2CID 31070331.
- ^ S2CID 20553167.
- ^ Weiss R (1 January 2007). "Scientists Announce Mad Cow Breakthrough". The Washington Post. Retrieved 1 January 2007.
- PMID 17195841.
- PMID 17498662.
- PMID 12093732.
External links
- PRNP (PrP) gene at GeneCard
- PRNP+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Susan Lindquist's Seminar: "The Surprising World of Prion Biology"