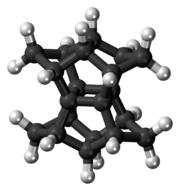
Pagodane

| |
| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C20H20 | |
| Molar mass | 260.380 g·mol−1 |
| Density | 1.629 g/ml |
| Structure | |
| D2h | |
| 0 D | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Pagodane is an
The name pagodane is used more generally for any member of a family of compounds whose molecular skeletons have the same 16-carbon central cage as the basic compound. Each member can be seen as the result of connecting eight atoms of this cage in pairs by four alkane chains. The general member is denoted [m.n.p.q]pagodane where m, n, p and q are the number of carbons of those four chains. The general formula is then C
16+sH
12+2s where s= m+n+p+q. In particular, the basic compound C
20H
20 has those carbons connected by four methylene bridges (m=n=p=q=1), and its name within that family is therefore [1.1.1.1]pagodane.[2]
Synthesis and structure
The compound was first synthesized by Horst Prinzbach and his associates in 1987, by a 14-step sequence starting from isodrin.[2] In the process they also synthesized [2.2.1.1]pagodane C
22H
24 and several derivatives.
Prinzbach remarked that "the obvious need for [the short name 'pagodane'] can be readily understood in view of the von Baeyer/
In carbon skeleton of pagodane, there can be distinguished many propellane-type fragments.[2]
The overall synthesis can be summarized as follows:[2][3]
 |
The scheme depicted here may be shortened to 14 one-pot operations with 24% overall yield. Yet, this variation requires the use of tetrachlorothiophenedioxide instead of tetrachloro-dimethoxycyclopentadiene in two of the early steps. While fewer steps and higher yield look attractive at first sight, this approach had to be given up due to high cost and restricted availability of the dioxide.[2]
Derivatives
Several derivatives are available, such as the
20H
16O
2 (melting point about 322 °C).[2]
Both [1.1.1.1]pagodane and [2.2.1.1]pagodane form
Pagodane is an isomer of dodecahedrane and can be chemically converted to it.[7][8]
References
- ^ Elegant Solutions: Ten Beautiful Experiments in Chemistry Philip Ball RSC 2005
- ^ a b c d e f g h Wolf-Dieter Fessner, Gottfried Sedelmeier, Paul R. Spurr, Grety Rihs, H. Prinzbach (1987), "Pagodane": the efficient synthesis of a novel, versatile molecular framework.
- ^ G. K. Surya Prakash (1998), Investigations on intriguing long lived carbodications. Pure & Appl. Chem., volume 70 issue 10, pp. 2001–06. Online version at iupac.org. Retrieved 2010-01-14.
- ^ G.K.S. Prakash, V.V. Krishnamurthy, R. Herges, R. Bau, H. Yuan, G.A Olah, W.-D. Fessner, H. Prinzbach: [1.1.1.1]- and [2.2.1.1]Pagodane Dications: Frozen Two-Electron Woodward-Hoffmann Transition State Models. J. Am. Chem. Soc. 1988, 110, 7764-7772
- ^ Wolf-Dieter Fessner, Bulusu A. R. C. Murty, Horst Prinzbach (1987), The Pagodane Route to Dodecahedranes – Thermal, Reductive, and Oxidative Transformations of Pagodanes
- ^ Wolf-Dieter Fessner, Bulusu A. R. C. Murty, Jürgen Wörth, Dieter Hunkler, Hans Fritz, Horst Prinzbach, Wolfgang D. Roth, Paul von Ragué Schleyer, Alan B. McEwen, Wilhelm F. Maier (1987), Dodecahedranes from [1.1.1.1]Pagodanes.
