Pancreatic cancer
| Pancreatic cancer | |
|---|---|
 | |
| Diagram showing the position of the pancreas, behind the stomach (which is transparent in this schematic). | |
| Specialty | |
Five year survival rate 13%[6] | |
| Frequency | 393,800 (2015)[7] |
| Deaths | 411,600 (2015)[8] |
Pancreatic cancer arises when
The most common, pancreatic adenocarcinoma, accounts for about 90% of cases,[11] and the term "pancreatic cancer" is sometimes used to refer only to that type.[10] These adenocarcinomas start within the part of the pancreas that makes digestive enzymes.[10] Several other types of cancer, which collectively represent the majority of the non-adenocarcinomas, can also arise from these cells.[10]
About 1–2% of cases of pancreatic cancer are neuroendocrine tumors, which arise from the hormone-producing cells of the pancreas.[10] These are generally less aggressive than pancreatic adenocarcinoma.[10]
Signs and symptoms of the most-common form of pancreatic cancer may include yellow skin, abdominal or back pain, unexplained weight loss, light-colored stools, dark urine, and loss of appetite.[1] Usually, no symptoms are seen in the disease's early stages, and symptoms that are specific enough to suggest pancreatic cancer typically do not develop until the disease has reached an advanced stage.[1][2] By the time of diagnosis, pancreatic cancer has often spread to other parts of the body.[10][12]
Pancreatic cancer rarely occurs before the age of 40, and more than half of cases of pancreatic adenocarcinoma occur in those over 70.
Pancreatic cancer is usually diagnosed by a combination of
The risk of developing pancreatic cancer is lower among non-smokers, and people who maintain a healthy weight and limit their consumption of red or processed meat;[5] however, the risk is greater for men, especially at very high levels of red meat consumption.[14] However, this is in debate, as a study performed by the International Journal of Cancer in 2013 did not find any statistically significant relationship between red meat consumption and pancreatic cancer, finding instead no male connection and only finding positive association of red meat consumption with pancreatic cancer risk in women after restriction to microscopically confirmed cases.[15] Smokers' risk of developing the disease decreases immediately upon quitting, and almost returns to that of the rest of the population after 20 years.[10] Pancreatic cancer can be treated with surgery, radiotherapy, chemotherapy, palliative care, or a combination of these.[1] Treatment options are partly based on the cancer stage.[1] Surgery is the only treatment that can cure pancreatic adenocarcinoma,[12] and may also be done to improve quality of life without the potential for cure.[1][12] Pain management and medications to improve digestion are sometimes needed.[12] Early palliative care is recommended even for those receiving treatment that aims for a cure.[16]
Pancreatic cancer is among the most deadly forms of cancer globally, with one of the lowest survival rates. In 2015, pancreatic cancers of all types resulted in 411,600 deaths globally.[8] Pancreatic cancer is the fifth-most-common cause of death from cancer in the United Kingdom,[17] and the third most-common in the United States.[18] The disease occurs most often in the developed world, where about 70% of the new cases in 2012 originated.[10] Pancreatic adenocarcinoma typically has a very poor prognosis; after diagnosis, 25% of people survive one year and 12% live for five years.[6][10] For cancers diagnosed early, the five-year survival rate rises to about 20%.[19] Neuroendocrine cancers have better outcomes; at five years from diagnosis, 65% of those diagnosed are living, though survival considerably varies depending on the type of tumor.[10]
Types

The many types of pancreatic cancer can be divided into two general groups. The vast majority of cases (about 95%) occur in the part of the pancreas that produces
The small minority of cancers that arise in the
Exocrine cancers
The exocrine group is dominated by pancreatic
The next-most common type, acinar cell carcinoma of the pancreas, arises in the clusters of cells that produce these enzymes, and represents 5% of exocrine pancreas cancers.[25] Like the 'functioning' endocrine cancers described below, acinar cell carcinomas may cause over-production of certain molecules, in this case digestive enzymes, which may cause symptoms such as skin rashes and joint pain.
Cystadenocarcinomas account for 1% of pancreatic cancers, and they have a better prognosis than the other exocrine types.[25]
Pancreatic mucinous cystic neoplasms are a broad group of pancreas tumors that have varying malignant potential. They are being detected at a greatly increased rate as CT scans become more powerful and common, and discussion continues as how best to assess and treat them, given that many are benign.[27]
Neuroendocrine
The small minority of tumors that arise elsewhere in the pancreas are mainly
As with other neuroendocrine tumors, the history of the terminology and classification of PanNETs is complex.
Signs and symptoms

Since pancreatic cancer usually does not cause recognizable symptoms in its early stages, the disease is typically not diagnosed until it has spread beyond the pancreas itself.[4] This is one of the main reasons for the generally poor survival rates. Exceptions to this are the functioning PanNETs, where over-production of various active hormones can give rise to symptoms (which depend on the type of hormone).[31]
Common presenting symptoms of pancreatic adenocarcinoma include:
- Pain in the upper abdomen or back, often spreading from around the stomach to the back. The location of the pain can indicate the part of the pancreas where a tumor is located. The pain may be worse at night and may increase over time to become severe and unremitting.[25] It may be slightly relieved by bending forward. In the UK, about half of new cases of pancreatic cancer are diagnosed following a visit to a hospital emergency department for pain or jaundice. In up to two-thirds of people, abdominal pain is the main symptom, for 46% of the total accompanied by jaundice, with 13% having jaundice without pain.[12]
- Jaundice, a yellow tint to the whites of the eyes or skin, with or without pain, and possibly in combination with darkened urine, results when a cancer in the head of the pancreas obstructs the common bile duct as it runs through the pancreas.[32]
- poor digestion.[12]
- The tumor may compress neighboring organs, disrupting digestive processes and making it difficult for the stomach to empty, which may cause nausea and a feeling of fullness. The undigested fat leads to foul-smelling, fatty feces that are difficult to flush away.[12] Constipation is also common.[33]
- At least 50% of people with pancreatic adenocarcinoma have diabetes at the time of diagnosis.[2] While long-standing diabetes is a known risk factor for pancreatic cancer (see Risk factors), the cancer can itself cause diabetes, in which case recent onset of diabetes could be considered an early sign of the disease.[34] People over 50 who develop diabetes have eight times the usual risk of developing pancreatic adenocarcinoma within three years, after which the relative risk declines.[12]
Other findings
- portal blood vessels (portal vein thrombosis), the deep veins of the extremities (deep vein thrombosis), or the superficial veins (superficial vein thrombosis) anywhere on the body – may be associated with pancreatic cancer, and is found in about 10% of cases.[3]
- Clinical depression has been reported in association with pancreatic cancer in some 10–20% of cases, and can be a hindrance to optimal management. The depression sometimes appears before the diagnosis of cancer, suggesting that it may be brought on by the biology of the disease.[3]
Other common manifestations of the disease include weakness and tiring easily, dry mouth, sleep problems, and a palpable abdominal mass.[33]
Symptoms of spread

The spread of pancreatic cancer to other organs (
Cancers in the pancreas may also be
Risk factors
Risk factors for pancreatic adenocarcinoma include:[2][10][12][37][38][excessive citations]
- Age, sex, and African Americans, though incidence in Africa is low.[10]
- Cigarette smoking is the best-established avoidable risk factor for pancreatic cancer, approximately doubling risk among long-term smokers, the risk increasing with the number of cigarettes smoked and the years of smoking. The risk declines slowly after smoking cessation, taking some 20 years to return to almost that of nonsmokers.[39]
- Obesity – a body mass index greater than 35 increases relative risk by about half.[12][40]
- Family history – 5–10% of pancreatic cancer cases have an inherited component, where people have a family history of pancreatic cancer.lifetime risk of pancreatic cancer of 30–40% to the age of 70.[3] Screening for early pancreatic cancer may be offered to individuals with hereditary pancreatitis on a research basis.[43] Some people may choose to have their pancreas surgically removed to prevent cancer from developing in the future.[3]
- Pancreatic cancer has been associated with these other rare hereditary syndromes:
- Chronic pancreatitis appears to almost triple risk, and as with diabetes, new-onset pancreatitis may be a symptom of a tumor.[3] The risk of pancreatic cancer in individuals with familial pancreatitis is particularly high.[3][42]
- type 3c (pancreatogenic) diabetes.[44]Chronic pancreatitis, pancreatic cancer and diabetes mellitus increased in contaminated populations, particularly children and adolescents, after Fukushima and Chernobyl nuclear incidents. At the same time, worldwide pancreatic diseases, diabetes and environmental radiocesium are increasing.
- Specific types of food (as distinct from obesity) have not been clearly shown to increase the risk of pancreatic cancer.[2][45] Dietary factors for which some evidence shows slightly increased risk include processed meat, red meat, and meat cooked at very high temperatures (e.g. by frying, broiling, or grilling).[45][46]
Alcohol
Drinking alcohol excessively is a major cause of chronic pancreatitis, which in turn predisposes to pancreatic cancer, but considerable research has failed to firmly establish alcohol consumption as a direct risk factor for pancreatic cancer. Overall, the association is consistently weak and the majority of studies have found no association, with smoking a strong confounding factor. The evidence is stronger for a link with heavy drinking, of at least six drinks per day.[3][47]
Pathophysiology

Precancer


Exocrine cancers are thought to arise from several types of
The first is pancreatic
A second type is the intraductal papillary mucinous neoplasm (IPMN). These are macroscopic lesions, which are found in about 2% of all adults. This rate rises to about 10% by age 70. These lesions have about a 25% risk of developing into invasive cancer. They may have KRAS gene mutations (40–65% of cases) and in the GNAS Gs alpha subunit and RNF43, affecting the Wnt signaling pathway.[2] Even if removed surgically, a considerably increased risk remains of pancreatic cancer developing subsequently.[3]
The third type, pancreatic mucinous cystic neoplasm (MCN), mainly occurs in women, and may remain benign or progress to cancer.[49] If these lesions become large, cause symptoms, or have suspicious features, they can usually be successfully removed by surgery.[3]
A fourth type of cancer that arises in the pancreas is the intraductal tubulopapillary neoplasm. This type was recognised by the WHO in 2010 and constitutes about 1–3% of all pancreatic neoplasms. Mean age at diagnosis is 61 years (range 35–78 years). About 50% of these lesions become invasive. Diagnosis depends on histology, as these lesions are very difficult to differentiate from other lesions on either clinical or radiological grounds.[50]
Invasive cancer
The genetic events found in ductal adenocarcinoma have been well characterized, and complete
PanNETs
The genes often found mutated in
Diagnosis
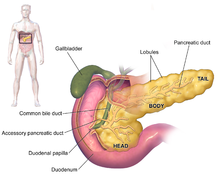


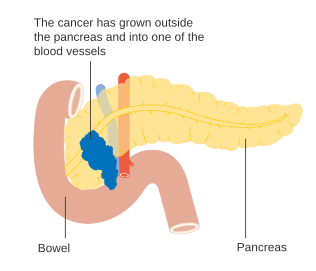
The symptoms of pancreatic adenocarcinoma do not usually appear in the disease's early stages, and they are not individually distinctive to the disease.[3][12][32] The symptoms at diagnosis vary according to the location of the cancer in the pancreas, which anatomists divide (from left to right on most diagrams) into the thick head, the neck, and the tapering body, ending in the tail.
Regardless of a tumor's location, the most common symptom is unexplained weight loss, which may be considerable. A large minority (between 35% and 47%) of people diagnosed with the disease will have had nausea, vomiting, or a feeling of weakness. Tumors in the head of the pancreas typically also cause jaundice, pain, loss of appetite, dark urine, and light-colored stools. Tumors in the body and tail typically also cause pain.[32]
People sometimes have recent onset of atypical type 2 diabetes that is difficult to control, a history of recent but unexplained blood vessel inflammation caused by blood clots (thrombophlebitis) known as Trousseau sign, or a previous attack of pancreatitis.[32] A doctor may suspect pancreatic cancer when the onset of diabetes in someone over 50 years old is accompanied by typical symptoms such as unexplained weight loss, persistent abdominal or back pain, indigestion, vomiting, or fatty feces.[12] Jaundice accompanied by a painlessly swollen gallbladder (known as Courvoisier's sign) may also raise suspicion, and can help differentiate pancreatic cancer from gallstones.[55]
A biopsy by
Histopathology
The most common form of pancreatic cancer (adenocarcinoma) is typically characterized by moderately to
| Cancer type | Relative incidence[11] | Microscopy findings[11] | Micrograph | Immunohistochemistry markers[11] | Genetic alterations[11] |
|---|---|---|---|---|---|
| Pancreatic ductal adenocarcinoma (PDAC) | 90% | Glands and desmoplasia | 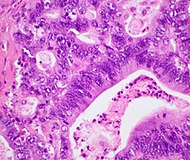 |
||
| Pancreatic acinar cell carcinoma (ACC) | 1% to 2% | Granular appearance | 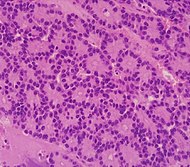
|
||
| Solid pseudopapillary tumor | Discohesive tumor nests surrounded by thin fibrous bands. |  Low and high magnification[58] |
| ||
| Adenosquamous carcinoma | 1% to 4%[59] | Combination of gland-like cells and squamous epithelial cells. |  |
Positive for:
Negative for: |
|
| Pancreatic neuroendocrine tumor | 5% | Multiple nests of tumor cells | 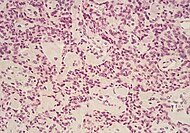 |
|
|
| Pre-cancer below for comparison: | |||||
| Precancer: Intraductal papillary mucinous neoplasm (IPMN) |
3% | Mucinous epithelial cells.[60] Growth within the pancreatic ducts.[61] | 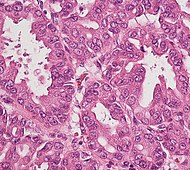
|
||
Staging
Exocrine cancers
Pancreatic cancer is usually staged following a CT scan.[32] The most widely used cancer staging system for pancreatic cancer is the one formulated by the American Joint Committee on Cancer (AJCC) together with the Union for International Cancer Control (UICC). The AJCC-UICC staging system designates four main overall stages, ranging from early to advanced disease, based on TNM classification of Tumor size, spread to lymph Nodes, and Metastasis.[62]
To help decide treatment, the tumors are also divided into three broader categories based on whether surgical removal seems possible: in this way, tumors are judged to be "resectable", "borderline resectable", or "unresectable".[63] When the disease is still in an early stage (AJCC-UICC stages I and II), without spread to large blood vessels or distant organs such as the liver or lungs, surgical resection of the tumor can normally be performed, if the patient is willing to undergo this major operation and is thought to be sufficiently fit.[12]
The AJCC-UICC staging system allows distinction between stage III tumors that are judged to be "borderline resectable" (where surgery is technically feasible because the
- Pancreatic cancer staging (TNM classification)
-
Stage T1 pancreatic cancer
-
Stage T2 pancreatic cancer
-
Stage T3 pancreatic cancer
-
Stage T4 pancreatic cancer
-
Pancreatic cancer in nearby lymph nodes – Stage N1

Locally advanced adenocarcinomas have spread into neighboring organs, which may be any of the following (in roughly decreasing order of frequency): the duodenum, stomach, transverse colon, spleen, adrenal gland, or kidney. Very often they also spread to the important blood or lymphatic vessels and nerves that run close to the pancreas, making surgery far more difficult. Typical sites for metastatic spread (stage IV disease) are the liver, peritoneal cavity and lungs, all of which occur in 50% or more of fully advanced cases.[64]
PanNETs
The 2010 WHO classification of tumors of the digestive system grades all the pancreatic neuroendocrine tumors (PanNETs) into three categories, based on their degree of cellular differentiation (from "NET G1" through to the poorly differentiated "NET G3").[22] The U.S. National Comprehensive Cancer Network recommends use of the same AJCC-UICC staging system as pancreatic adenocarcinoma.[65]: 52 Using this scheme, the stage-by-stage outcomes for PanNETs are dissimilar to those of the exocrine cancers.[66] A different TNM system for PanNETs has been proposed by the European Neuroendocrine Tumor Society.[22]
Prevention and screening
Apart from not smoking, the
In the general population, screening of large groups is not considered effective and may be harmful as of 2019,[68] although newer techniques, and the screening of tightly targeted groups, are being evaluated.[69][70] Nevertheless, regular screening with endoscopic ultrasound and MRI/CT imaging is recommended for those at high risk from inherited genetics.[4][57][70][71]
A 2019 meta-analysis found that use of aspirin might be negatively associated with the incidence risk of pancreatic cancer, but found no significant relationship with pancreatic cancer mortality.[72]
Management
Exocrine cancer
A key assessment that is made after diagnosis is whether surgical removal of the tumor is possible (see Staging), as this is the only cure for this cancer. Whether or not surgical resection can be offered depends on how much the cancer has spread. The exact location of the tumor is also a significant factor, and CT can show how it relates to the major blood vessels passing close to the pancreas. The general health of the person must also be assessed, though age in itself is not an obstacle to surgery.[3]
Chemotherapy and, to a lesser extent, radiotherapy are likely to be offered to most people, whether or not surgery is possible. Specialists advise that the management of pancreatic cancer should be in the hands of a
Surgery

Surgery with the intention of a cure is only possible in around one-fifth (20%) of new cases.[12] Although CT scans help, in practice it can be difficult to determine whether the tumor can be fully removed (its "resectability"), and it may only become apparent during surgery that it is not possible to successfully remove the tumor without damaging other vital tissues. Whether or not surgical resection can be offered depends on various factors, including the precise extent of local anatomical adjacency to, or involvement of, the venous or arterial blood vessels,[2] as well as surgical expertise and a careful consideration of projected post-operative recovery.[73][74] The age of the person is not in itself a reason not to operate, but their general performance status needs to be adequate for a major operation.[12]
One particular feature that is evaluated is the encouraging presence, or discouraging absence, of a clear layer or plane of fat creating a barrier between the tumor and the vessels.[3] Traditionally, an assessment is made of the tumor's proximity to major venous or arterial vessels, in terms of "abutment" (defined as the tumor touching no more than half a blood vessel's circumference without any fat to separate it), "encasement" (when the tumor encloses most of the vessel's circumference), or full vessel involvement.[75]: 22 A resection that includes encased sections of blood vessels may be possible in some cases,[76][77] particularly if preliminary neoadjuvant therapy is feasible,[78][79][80] using chemotherapy[74][75]: 36 [81] and/or radiotherapy.[75]: 29–30
Even when the operation appears to have been successful, cancerous cells are often found around the edges ("

For cancers involving the head of the pancreas, the
Although curative surgery no longer entails the very high death rates that occurred until the 1980s, a high proportion of people (about 30–45%) still have to be treated for a post-operative sickness that is not caused by the cancer itself. The most common complication of surgery is difficulty in emptying the stomach.[3] Certain more limited surgical procedures may also be used to ease symptoms (see Palliative care): for instance, if the cancer is invading or compressing the duodenum or colon. In such cases, bypass surgery might overcome the obstruction and improve quality of life but is not intended as a cure.[12]
Chemotherapy
After surgery,
Gemcitabine was approved by the United States
The FOLFIRINOX chemotherapy regimen using four drugs was found more effective than gemcitabine, but with substantial side effects, and is thus only suitable for people with good performance status. This is also true of protein-bound paclitaxel (nab-paclitaxel), which was licensed by the FDA in 2013 for use with gemcitabine in pancreas cancer.[89] By the end of 2013, both FOLFIRINOX and nab-paclitaxel with gemcitabine were regarded as good choices for those able to tolerate the side-effects, and gemcitabine remained an effective option for those who were not. A head-to-head trial between the two new options is awaited, and trials investigating other variations continue. However, the changes of the last few years have only increased survival times by a few months.[86] Clinical trials are often conducted for novel adjuvant therapies.[4]
Radiotherapy
The role of radiotherapy as an auxiliary (adjuvant) treatment after potentially curative surgery has been controversial since the 1980s.[3] In the early 2000s the European Study Group for Pancreatic Cancer Research (ESPAC) showed prognostic superiority of adjuvant chemotherapy over chemoradiotherapy.[90][91][4] The European Society for Medical Oncology recommends that adjuvant radiotherapy should only be used for people enrolled in clinical trials.[57] However, there is a continuing tendency for clinicians in the US to be more ready to use adjuvant radiotherapy than those in Europe. Many clinical trials have tested a variety of treatment combinations since the 1980s, but have failed to settle the matter conclusively.[3][4]
Radiotherapy may form part of treatment to attempt to shrink a tumor to a resectable state, but its use on unresectable tumors remains controversial as there are conflicting results from clinical trials. The preliminary results of one trial, presented in 2013, "markedly reduced enthusiasm" for its use on locally advanced tumors.[2]
PanNETs
Treatment of PanNETs, including the less common
For localized tumors, the surgical procedure may be much less extensive than the types of surgery used to treat pancreatic adenocarcinoma described above, but otherwise surgical procedures are similar to those for exocrine tumors. The range of possible outcomes varies greatly; some types have a very high survival rate after surgery while others have a poor outlook. As all this group are rare, guidelines emphasize that treatment should be undertaken in a specialized center.[22][29] Use of liver transplantation may be considered in certain cases of liver metastasis.[95]
For functioning tumors, the
Radiation therapy is occasionally used if there is pain due to anatomic extension, such as
Palliative care
Palliative care is medical care which focuses on treatment of symptoms from serious illness, such as cancer, and improving quality of life.[106] Because pancreatic adenocarcinoma is usually diagnosed after it has progressed to an advanced stage, palliative care as a treatment of symptoms is often the only treatment possible.[107]
Palliative care focuses not on treating the underlying cancer, but on treating symptoms such as
Other symptoms or complications that can be treated with palliative surgery are obstruction by the tumor of the intestines or
Both surgery and advanced inoperable tumors often lead to
can also be used to clear stomach contents.Outcomes
| Clinical stage | U.S. five-year survival (%) for 1992–1998 diagnoses | |
|---|---|---|
| Exocrine pancreatic cancer | Neuroendocrine treated with surgery | |
| IA / I | 14 | 61 |
| IB | 12 | |
| IIA / II | 7 | 52 |
| IIB | 5 | |
| III | 3 | 41 |
| IV | 1 | 16 |
Pancreatic adenocarcinoma and the other less common exocrine cancers have a very poor prognosis, as they are normally diagnosed at a late stage when the cancer is already locally advanced or has spread to other parts of the body.[2] Outcomes are much better for PanNETs: Many are benign and completely without clinical symptoms, and even those cases not treatable with surgery have an average five-year survival rate of 16%,[63] although the outlook varies considerably according to the type.[31]
For locally advanced and
About 1500 genes are linked to outcomes in pancreatic adenocarcinoma. These include both unfavorable genes, where high expression is related to poor outcome, for example
Distribution

In 2015, pancreatic cancers of all types resulted in 411,600 deaths globally.[8] In 2014, an estimated 46,000 people in the US are expected to be diagnosed with pancreatic cancer and 40,000 to die of it.[2] Although it accounts for only 2.5% of new cases, pancreatic cancer is responsible for 6% of cancer deaths each year.[111] It is the seventh-highest cause of death from cancer worldwide.[10] Pancreatic cancer is the fifth most-common cause of death from cancer in the United Kingdom,[17] and the third most-common in the United States.[18]
Globally, pancreatic cancer is the 11th most-common cancer in women and the 12th most-common in men.
PanNETs
The annual
History
Recognition and diagnosis
The earliest recognition of pancreatic cancer has been attributed to the 18th-century Italian scientist
Regarding the recognition of PanNETs, the possibility of cancer of the islet cells was initially suggested in 1888. The first case of
Small precancerous neoplasms for many pancreatic cancers are being detected at greatly increased rates by modern medical imaging. One type, the intraductal papillary mucinous neoplasm (IPMN) was first described by Japanese researchers in 1982. It was noted in 2010 that: "For the next decade, little attention was paid to this report; however, over the subsequent 15 years, there has been a virtual explosion in the recognition of this tumor."[64]
Surgery
The first reported partial pancreaticoduodenectomy was performed by the Italian surgeon Alessandro Codivilla in 1898, but the patient only survived 18 days before succumbing to complications. Early operations were compromised partly because of mistaken beliefs that people would die if their duodenum were removed, and also, at first, if the flow of pancreatic juices stopped. Later it was thought, also mistakenly, that the pancreatic duct could simply be tied up without serious adverse effects; in fact, it will very often leak later on. In 1907–1908, after some more unsuccessful operations by other surgeons, experimental procedures were tried on corpses by French surgeons.[115]
In 1912 the German surgeon
The first operation was unplanned, as cancer was only discovered in the operating theater. Whipple's success showed the way for the future, but the operation remained a difficult and dangerous one until recent decades. He published several refinements to his procedure, including the first total removal of the duodenum in 1940, but he only performed a total of 37 operations.[115]
The discovery in the late 1930s that vitamin K prevented bleeding with jaundice, and the development of blood transfusion as an everyday process, both improved post-operative survival,[115] but about 25% of people never left hospital alive as late as the 1970s.[116] In the 1970s a group of American surgeons wrote urging that the procedure was too dangerous and should be abandoned. Since then outcomes in larger centers have improved considerably, and mortality from the operation is often less than 4%.[24]
In 2006 a report was published of a series of 1,000 consecutive pancreatico-duodenectomies performed by a single surgeon from Johns Hopkins Hospital between 1969 and 2003. The rate of these operations had increased steadily over this period, with only three of them before 1980, and the median operating time reduced from 8.8 hours in the 1970s to 5.5 hours in the 2000s, and mortality within 30 days or in hospital was only 1%.[115][116] Another series of 2,050 operations at the Massachusetts General Hospital between 1941 and 2011 showed a similar picture of improvement.[117]
Research directions
This section needs to be updated. (May 2023) |
Early-stage research on pancreatic cancer includes studies of genetics and early detection, treatment at different cancer stages, surgical strategies, and targeted therapies, such as inhibition of growth factors, immune therapies, and vaccines.[42][118][119][120][121] Bile acids may have a role in the carcinogenesis of pancreatic cancer.[122]
A key question is the timing of events as the disease develops and progresses – particularly the role of
Keyhole surgery (
Efforts are underway to develop new drugs, including those targeting
The nanoparticles assist in the sustained and targeted release of a drug regimen to cancer/tumor-specific sites rather than affecting healthy cells, leading to negligible or no toxicity.[134]
See also
- Gastrointestinal cancer
- Pancreatic Cancer Action Network (organization in the US)
- Lustgarten Foundation for Pancreatic Cancer Research (organization in the US)
- List of people diagnosed with pancreatic cancer
References
- ^ a b c d e f g "Pancreatic Cancer Treatment (PDQ®) Patient Version". National Cancer Institute. National Institutes of Health. 17 April 2014. Archived from the original on 5 July 2014. Retrieved 8 June 2014.
- ^ PMID 25207767.
- ^ PMID 23856911.
- ^ PMID 21620466. Archived from the original(PDF) on 12 January 2015.
- ^ a b "Can pancreatic cancer be prevented?". American Cancer Society. 11 June 2014. Archived from the original on 13 November 2014. Retrieved 13 November 2014.
- ^ a b "Five-Year Pancreatic Cancer Survival Rate Increases to 12%". PANCAN. 2023. Archived from the original on 4 March 2023. Retrieved 3 March 2023.
- PMID 27733282.
- ^ PMID 27733281.
- ^ "What Is Cancer? Defining Cancer". National Cancer Institute, National Institutes of Health. 7 March 2014. Archived from the original on 25 June 2014. Retrieved 5 December 2014.
- ^ ISBN 978-92-832-0429-9.
- ^ PMID 28299752.
- ^ S2CID 206894869.
- ^ "Draft Recommendation Statement: Pancreatic Cancer: Screening". US Preventive Services Task Force. Retrieved 11 February 2019.
- PMID 22240790.
- S2CID 2613568.
- PMID 24182608.
- ^ a b "Cancer facts and figures – Why we exist". Pancreatic Cancer Research Fund. Retrieved 5 April 2019.
- ^ a b "Pancreatic Cancer – Cancer Stat Facts". SEER. Retrieved 4 April 2019.
- ^ a b "Pancreatic Cancer Treatment (PDQ®) Health Professional Version". National Cancer Institute. National Institutes of Health. 21 February 2014. Archived from the original on 22 October 2014. Retrieved 24 November 2014. "The highest cure rate occurs if the tumor is truly localized to the pancreas; however, this stage of disease accounts for less than 20% of cases. In cases with localized disease and small cancers (<2 cm) with no lymph-node metastases and no extension beyond the capsule of the pancreas, complete surgical resection is still associated with a low actuarial five-year survival rate of 18% to 24%."
- PMID 21721197.). Diagram by Mikael Häggström, M.D.
{{cite journal}}: CS1 maint: multiple names: authors list (link - ISBN 978-0-7637-8047-0. Archivedfrom the original on 24 June 2016.
- ^ outlines the proposed TNM staging system for PanNETs.)
- ISBN 978-0-387-77497-8. Archivedfrom the original on 10 September 2017. Retrieved 12 June 2016.
- ^ ISBN 978-1-4511-0545-2. Online edition, with updates to 2014
- ^ ISBN 978-1-118-46871-5.
- ^ a b c "Types of Pancreas Tumors". The Sol Goldman Pancreas Cancer Research Center. Johns Hopkins Medicine. 2012. Archived from the original on 8 October 2014. Retrieved 18 November 2014.
- PMID 23622140.
- ^ S2CID 3735444.
- ^ S2CID 7329783.
- ^ The Medical Subject Headings indexing system refers to "islet cell carcinoma", which is subdivided into gastrinoma, glucagonoma, somatostatinoma, and VIPoma. See: 2014 MeSH tree at "Pancreatic Neoplasms [C04.588.322.475]" Archived 19 March 2016 at the Wayback Machine 16 October 2014
- ^ a b "Islet Cell Tumors of the Pancreas / Endocrine Neoplasms of the Pancreas". The Sol Goldman Pancreas Cancer Research Center. Johns Hopkins Medicine. 2012. Archived from the original on 5 January 2015. Retrieved 5 January 2015.
- ^ PMID 24784121.
- ^ ISBN 978-0-7817-6884-9.
- ^ PMID 19111249.
- ^ "Chapter 15; Pancreas" (PDF). Manual for Staging of Cancer (2nd ed.). American Joint Committee on Cancer. pp. 95–98. Archived (PDF) from the original on 29 November 2014. See p. 95 for citation regarding "... lesser degree of involvement of bones and brain and other anatomical sites."
- PMID 25320654.
- ^ "Causes of pancreatic cancer". NHS Choices. National Health Service, England. 7 October 2014. Archived from the original on 6 November 2014. Retrieved 5 December 2014.
- PMID 19194662.
- PMID 22104574.
- PMID 31230151.
- PMID 27041411.
- ^ PMID 24624093.
- S2CID 29100310.
- PMID 34501532..
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ a b "Cancer Facts and Figures 2014" (PDF). American Cancer Society. Archived (PDF) from the original on 18 December 2014. Retrieved 5 January 2015., p. 19, "Though evidence is still accumulating, consumption of red or processed meat, or meat cooked at very high temperatures, may slightly increase risk."
- PMID 22240790.
- ^ PMID 24403441.
- PMID 27267993.)), Image title and optimization: Mikael Häggström, M.D.
{{cite journal}}: CS1 maint: multiple names: authors list (link) (distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/ - PMID 21886451.
- PMID 27684978.
- ^ a b "The human pathology proteome in pancreatic cancer – The Human Protein Atlas". www.proteinatlas.org. Retrieved 28 September 2017.
- ^ PMID 28818916.
- ^ S2CID 31094880.
- PMID 22723327.
- S2CID 21799234.
- S2CID 38963685.
- ^ PMID 22997452.
- ^ a b c d Image by Mikael Häggström, MD.
Reference for features: Pooja Navale, M.D., Omid Savari, M.D., Joseph F. Tomashefski, Jr., M.D., Monika Vyas, M.D. "Solid pseudopapillary neoplasm".{{cite web}}: CS1 maint: multiple names: authors list (link) Last author update: 4 March 2022 - PMID 20205828.
- ^ Diana Agostini-Vulaj. "Pancreas – Exocrine tumors / carcinomas – Intraductal papillary mucinous neoplasm (IPMN)". Pathology Outlines. Topic Completed: 1 July 2018. Revised: 9 March 2020
- PMID 25775066.
- PMID 20555103.
- ^ a b c "Staging of pancreatic cancer". American Cancer Society. 11 June 2014. Archived from the original on 10 August 2020. Retrieved 29 September 2014.
- ^ ISBN 978-0-7817-6546-6. Archived from the originalon 6 February 2015. Retrieved 3 November 2014.
- ^ a b c d "Neuroendocrine tumors, NCCN Guidelines Version 1.2015" (PDF). National Comprehensive Cancer Network, Inc. NCCN). 11 November 2014. Retrieved 25 December 2014.
- ^ National Cancer Institute. Pancreatic Neuroendocrine Tumors (Islet Cell Tumors) Treatment (PDQ®) Incidence and Mortality "Pancreatic Neuroendocrine Tumors (Islet Cell Tumors) Treatment (PDQ®)–Health Professional Version". Archived from the original on 4 January 2015. Retrieved 29 December 2014.
- ^ "Diet and activity factors that affect risks for certain cancers: Pancreatic cancer section". American Cancer Society. 20 August 2012. Archived from the original on 4 November 2014. Retrieved 4 November 2014.
- PMID 31386141.
- PMID 25170208.
- ^ PMID 25170207.
- PMID 31672839.
- PMID 31860953.
- PMID 24578248.
- ^ S2CID 42685174.
- ^ a b c "Pancreatic adenocarcinoma. NCCN Guidelines Version 1.2015" (PDF). NCCN Guidelines. National Comprehensive Cancer Network, Inc. 4 December 2014. Retrieved 26 December 2014.
- PMID 25339810.
- PMID 25152577.
- PMID 25071332.)
{{cite journal}}: CS1 maint: DOI inactive as of January 2024 (link - PMID 20422030.
- PMID 25519932.
- PMID 25089113.
- PMID 25499079.
- ^ PMID 25152582.
- PMID 27383694.
- PMID 23852311.
- ^ PMID 24683721.
- Wolters Kluwer Health. Archivedfrom the original on 8 December 2014. Retrieved 18 November 2014.
- ^ "Cancer Drug Information: FDA Approval for Erlotinib Hydrochloride". National Cancer Institute. National Institutes of Health. 3 July 2013. Archived from the original on 29 November 2014. Retrieved 5 December 2014.
- S2CID 31633898.
- S2CID 23803289.
- PMID 15028824.
- S2CID 6985904.
- PMID 22261919.
- S2CID 2097604.
- PMID 25332984.
- ^ Nick Mulcahy (17 December 2014). "FDA Approves Lanreotide for Neuroendocrine Tumors". Medscape Medical News. WebMD LLC. Archived from the original on 18 January 2015. Retrieved 25 December 2014.
- The ASCO Post. 15 May 2011, Volume 2, Issue 8
- ^ National Cancer Institute. Cancer Drug Information. FDA Approval for Sunitinib Malate Archived 5 January 2015 at the Wayback Machine. Pancreatic Neuroendocrine Tumors
- ISBN 978-0-615-41824-7. Archivedfrom the original on 15 May 2011.
- PMID 22221516.
- PMID 25297671.
- PMID 20113677.
- PMID 25065935.
- S2CID 39434924.
- S2CID 8129133.
- ^ "Palliative or Supportive Care". American Cancer Society. 2014. Archived from the original on 21 August 2014. Retrieved 20 August 2014.
- PMID 25132756.
- ^ "If treatment for pancreatic cancer stops working". American Cancer Society. 11 June 2014. Archived from the original on 22 October 2014. Retrieved 20 August 2014.
- PMID 21412903.
- ^ "Cancer Facts and Figures 2014" (PDF). American Cancer Society. Archived (PDF) from the original on 18 December 2014. Retrieved 5 January 2015., Table, p. 18, rates adjusted for normal life expectancy
- S2CID 22305510.
- ^ "What are the key statistics about pancreatic cancer?". American Cancer Society. 11 June 2014. Archived from the original on 11 November 2014. Retrieved 11 November 2014.
- ^ "Pancreatic cancer statistics". Cancer Research UK. Archived from the original on 18 December 2014. Retrieved 18 December 2014.; "In 2010, in the UK, the lifetime risk of developing pancreatic cancer is 1 in 73 for men and 1 in 74 for women", noting "The lifetime risk ... has been calculated ... using the 'Current Probability' method; this is a different method used from most other cancer sites since the possibility of having more than one diagnosis of pancreatic cancer over the course of their lifetime is very low"
- ^ PMID 6356946.
- ^ PMID 21609369.
- ^ PMID 16794383.
- PMID 22770961.
- ^ a b c "What's new in pancreatic cancer research and treatment?". American Cancer Society. 2019. Retrieved 2 May 2019.
- ^ "Pancreatic cancer research". Cancer Research UK. Archived from the original on 18 February 2014. Retrieved 17 July 2014.
- ^ "Australian Pancreatic Genome Initiative". Garvan Institute. Archived from the original on 26 July 2014. Retrieved 17 July 2014.
- PMID 23103869.
- PMID 27672269.
- S2CID 26414866.
- PMID 25479908.
- ^ "About EUROPAC". European Registry of Hereditary Pancreatitis and Familial Pancreatic Cancer (EUROPAC). University of Liverpool. Archived from the original on 26 July 2014. Retrieved 17 July 2014.
- PMID 24485260.
- S2CID 3137300.
- PMID 25307210.
- PMID 24631947.
- ^ PMID 25152585.
- PMID 25170201.
- PMID 27512661.
- PMID 30060755.
- ISBN 978-981-19-5557-0.)
{{cite book}}:|journal=ignored (help)CS1 maint: multiple names: authors list (link
External links
- Pancreatic cancer at Curlie