Passerini reaction
| Passerini reaction | |
|---|---|
| Named after | Mario Passerini |
| Reaction type | Carbon-carbon bond forming reaction |
| Identifiers | |
| Organic Chemistry Portal | passerini-reaction |
| RSC ontology ID | RXNO:0000244 |
The Passerini reaction is a
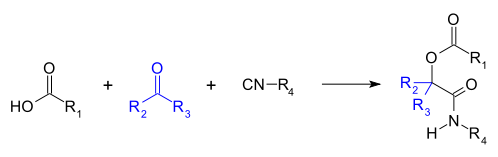
Mechanism
The Passerini reaction has been hypothesized to occur through two mechanistic pathways.[10][7][11] The reaction pathways are dependent on the solvent used.
Concerted mechanism
A

This mechanism involves a
As the Mumm rearrangement requires a second carboxylic acid molecule, this mechanism classifies the Passerini reaction as an organocatalytic reaction.[14][15]
Ionic mechanism
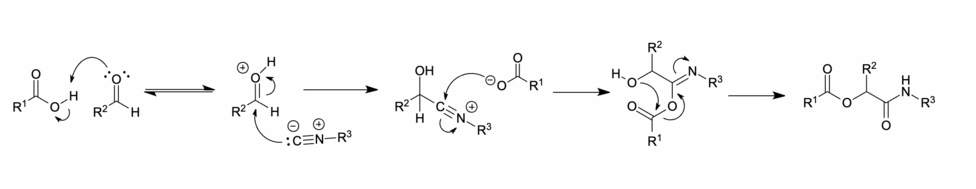
In polar solvents, such as
Reaction control
Molecular weights of polymers synthesized through the Passerini can be controlled through stoichiometric means.[16] For example, polymer chain length and weight can adjusted through isocyanide stoichiometry, and polymer geometry can be influenced through starting reagents.[16][17] To facilitate the Passerini reaction between bulky, sterically hindered reagents, a vortex fluidic device can be used to induce high shear conditions. These conditions emulate the effects of high temperature and pressure, allowing the Passerini reaction to proceed fairly quickly.[18] The Passerini reaction can also exhibit enantioselectivity. Addition of tert-butyl isocyanide to a wide variety of aldehydes (aromatic, heteroaromatic, olefinic, acetylenic, aliphatic) is achieved using a catalytic system of tetrachloride and a chiral bisphosphoramide which provides good yield and good enantioselectivities.[19] For other types of isocyanides, rate of addition of isocyanide to reaction mixture dictates good yields and high selectivities.[19]
Applications
Apart from forming α-
Heterocycles

The original Passerini reaction produces acyclic depsipeptides which are labile in physiological conditions. To increase product stability for medicinal use, post-Passerini cyclization reactions have been used to afford heterocycles such as β-lactams, butenolides, and isocoumarins.[16] To enable these cyclizations, reagents are pre-functionalized with reactive groups (ex. halogens, azides, etc.) and used in tandem with other reactions (ex. Passerini-Knoevenagel, Passerini-Dieckmann) to afford heterocyclic products.[16] Compounds like three membered oxirane and aziridine derivatives, four-membered b-lactams, and five-membered tetrasubstituted 4,5-dihydropyrazoles have been produced through this reaction.[12]
Polymers

This reaction has also been used for polymerization,
Amino acids and pharmaceuticals
Passerini reaction has been employed for the formation of structures like α-amino acids, α-hydroxy-β-amino acids, α-ketoamides, β-ketoamides, α-hydroxyketones and α-aminoxyamides.[12] The Passerini reaction has synthesized α-Acyloxy carboxamides that have demonstrated activity as anti-cancer medications along with functionalized [C60]-fullerenes used in medicinal and plant chemistry.[12][25] This reaction has also been used as a synthetic step in the total synthesis of commercially available pharmaceuticals such as telaprevir (VX-950), an antiviral sold by Vertex Pharmaceuticals and Johnson & Johnson.[12]

See also
References
- ^ Passerini, M.; Simone, L. Gazz. Chim. Ital. 1921, 51, 126–29.
- ^ Passerini, M.; Ragni, G. Gazz. Chim. Ital. 1931, 61, 964–69.
- ).
- .
- PMID 35003575.
- ^ S2CID 236498755.
- ^ PMID 35479468.
- ^ PMID 24994950.
- Angew. Chem. Int. Ed. Engl.2000, 39, 3168–3210. (Review)
- ^ ISBN 0-471-68260-8
- ^ .
- ^ S2CID 245431805.
- ISBN 978-3-030-50865-4, retrieved 24 October 2022
- ^ PMID 25974627.
- PMID 21643563.
- ^ ISSN 1759-9962.
- ^ .
- .
- ^ PMID 16292793.
- ^ PMID 21265532.
- PMID 23945608.
- ISSN 0024-9297.
- S2CID 225447799.
- PMID 24307280.
- S2CID 46969435.
