Pellizzari reaction
The Pellizzari reaction was discovered in 1911 by Guido Pellizzari, and is the
The product is similar to that of the
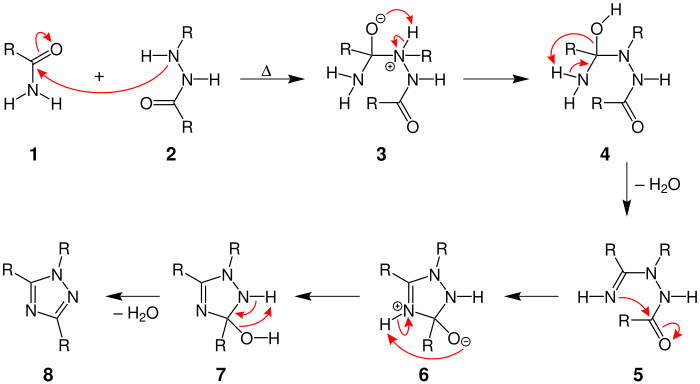
Mechanism
The mechanism begins by the nitrogen in the hydrazide attacking the
Uses
The synthesis of the 1,2,4-triazole has a wide range of biological functions.[
Problems and variations
The Pellizzari reaction is limited in the number of substituents that can be on the ring, so other methods have been developed to incorporate three elements of diversity. Liquid-phase synthesis of 3-alkylamino-4,5-disubstituted-1,2,4-triazoles by PEG support has given moderate yields with excellent purity.[4] In practice, the Pellizzari reaction requires high temperatures, long reaction times, and has an overall low yield. However, adding microwave irradiation shortens the reaction time and increases its yield.[5]
Related reactions
- Einhorn-Brunner reaction
References
- Gazz. Chim. Ital.1911, 41, 20.
- ISBN 9780471704508.
- PMID 15081345.
- .
- doi:10.1016/j.tet.2012.01.003. Archived from the original(PDF) on 2016-03-04. Retrieved 2014-05-20.