Penicillamine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Cuprimine, Cuprenyl, Depen, others |
| Other names | D-penicillamine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618021 |
| License data | |
| Pregnancy category |
|
By mouth (capsules) | |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Variable |
| Metabolism | Liver |
| Elimination half-life | 1 hour |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Penicillamine, sold under the brand name of Cuprimine among others, is a
Penicillamine was approved for medical use in the United States in 1970.
Medical uses
It is used as a chelating agent:
- In Wilson's disease, a rare genetic disorder of copper metabolism, penicillamine treatment relies on its binding to accumulated copper and elimination through urine.[4]
- Penicillamine was the second line treatment for arsenic poisoning, after dimercaprol (BAL).[5] It is no longer recommended.[6]
In
Penicillamine has been used to treat scleroderma.[8]
Penicillamine can be used as a disease-modifying antirheumatic drug (DMARD) to treat severe active rheumatoid arthritis in patients who have failed to respond to an adequate trial of conventional therapy,[9] although it is rarely used today due to availability of TNF inhibitors and other agents, such as tocilizumab and tofacitinib. Penicillamine works by reducing numbers of T-lymphocytes, inhibiting macrophage function, decreasing IL-1, decreasing rheumatoid factor, and preventing collagen from cross-linking.
Adverse effects
Common side effects include rash, loss of appetite, nausea, diarrhea, and
Bone marrow suppression, dysgeusia, anorexia, vomiting, and diarrhea are the most common side effects, occurring in ~20–30% of the patients treated with penicillamine.[10][11]
Other possible adverse effects include:
- Hepatotoxicity[12]
- Membranous glomerulonephritis[13]
- Aplastic anemia (idiosyncratic)[12]
- Antibody-mediated myasthenia gravis[10] and Lambert–Eaton myasthenic syndrome, which may persist even after its withdrawal
- Drug-induced systemic lupus erythematosus[14]
- Elastosis perforans serpiginosa[15]
- Toxic myopathies[16]
- Unwanted breast growth[17]
- Oligospermia
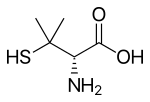
Chemistry

Penicillamine is a tri
Penicillamine is a chiral drug with one stereogenic center and exist as a pair of enantiomers. Refer the image for the structure of penicillamine enantiomers. The (S)-enantiomer, the
History
John Walshe first described the use of penicillamine in Wilson's disease in 1956.
Penicillamine was first synthesized by
Penicillamine has been used in rheumatoid arthritis since the first successful case in 1964.[25]
Cost
In the United States,
References
- ^ a b c d e f "Penicillamine". The American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 8 December 2016.
- ^ ISBN 9789241547659.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.)
{{cite book}}: CS1 maint: location missing publisher (link - PMID 4306792.
- PMID 908992.
- PMID 11869818.
- ^ PMID 6071831.
- PMID 7137731.
- ^ "Cuprimine (penicillamine) Capsules for Oral Use. U.S. Full Prescribing Information" (PDF). Archived (PDF) from the original on 8 September 2015. Retrieved 29 April 2016.
- ^ PMID 859814.
- PMID 3063003.
- ^ PMID 2774703.
- ISBN 978-1-4160-2973-1.
- PMID 6958210.
- ISBN 978-1-4160-2999-1.2nd edition.
- ISBN 978-0-443-06889-8.
- PMID 6780026.
- ^ Ariens EJ (1989). Chiral Separations by HPLC. Chichester: Ellis Horwwod, Chichester. pp. 31–68.
- ISBN 9780080932941. Archivedfrom the original on 10 September 2017.
- PMID 14483916.
- PMID 903530.
- PMID 13279157.
- S2CID 11406561.
- ISBN 9781438118826.
- PMID 14218939.
- ^ Petersen M. "How 4 drug companies rapidly raised prices on life-saving drugs". Los Angeles Times. Retrieved 27 March 2019.
External links
- "Penicillamine". Drug Information Portal. U.S. National Library of Medicine.
