Pentene
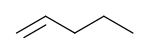 1-Pentene
| |
 cis-2-Pentene
| |
 trans-2-Pentene
| |
| Names | |
|---|---|
| IUPAC names
Pent-1-ene
cis-Pent-2-ene trans-Pent-2-ene | |
| Other names
amylene, n-amylene, n-pentene, beta-n-amylene, sym-methylethylethylene, pentylene
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.042.636 |
| EC Number |
|
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H10 | |
| Molar mass | 70.135 g·mol−1 |
| Density | 0.64 g/cm3 (1-pentene)[1] |
| Melting point | −165.2 °C (−265.4 °F; 108.0 K) (1-pentene)[1] |
| Boiling point | 30 °C (86 °F; 303 K) (1-pentene)[1] |
| -53.7·10−6 cm3/mol | |
| Hazards | |
| Safety data sheet (SDS) | MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Pentenes (also called Pentylenes) are alkenes with the chemical formula C
5H
10. Each molecule contains one double bond within its molecular structure. Six different compounds are in this class, differing from each other by whether the carbon atoms are attached linearly or in a branched structure and whether the double bond has a cis or trans form.
Straight-chain isomers
1-Pentene is an
via thermal cracking of hydrocarbon fractions.The only commercial manufacturer of 1-pentene is
2-Pentene has two geometric isomers: cis-2-pentene and trans-2-pentene. Cis-2-Pentene is used in olefin metathesis.
Branched-chain isomers
The branched isomers are 2-methylbut-1-ene, 3-methylbut-1-ene (isopentene), and
Isoamylene is one of the three main byproducts of
Production of fuels
Propylene, isobutene, and amylenes are feedstocks in the alkylation units of refineries. Using isobutane, blendstocks are generated with high branching for good combustion characteristics. Amylenes are valued as precursors to fuels, especially aviation fuels of relatively low volatility, as required by various regulations.[3]
References
- ^ a b c Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ "RSA Olefins | cChange". www.cchange.ac.za. Retrieved 2017-10-19.
- ISBN 0-471-23896-1.
