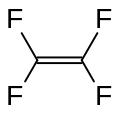
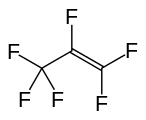
Fluorocarbon

Fluorocarbons are
Nomenclature
Perfluorocarbons or PFCs, are
Perfluoroalkanes
Chemical properties
Perfluoroalkanes are very stable because of the strength of the carbon–fluorine bond, one of the strongest in organic chemistry.[4] Its strength is a result of the electronegativity of fluorine imparting partial ionic character through
Fluorocarbons are colorless and have high density, up to over twice that of water. They are not miscible with most organic solvents (e.g., ethanol, acetone, ethyl acetate, and chloroform), but are miscible with some hydrocarbons (e.g., hexane in some cases). They have very low solubility in water, and water has a very low solubility in them (on the order of 10 ppm). They have low refractive indices.
As the high
- Perfluoroalkanes
-
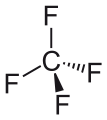
Carbon tetrafluoride, the simplest perfluoroalkane
-
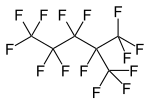
Perfluorooctane, a linear perfluoroalkane
-
Perfluoro-2-methylpentane, a branched perfluoroalkane
-
Perfluoro-1,3-dimethylcyclohexane, a cyclic perfluoroalkane
-
Perfluorodecalin, a polycyclic perfluoroalkane
Flammability
In the 1960s there was a lot of interest in fluorocarbons as anesthetics. The research did not produce any anesthetics, but the research included tests on the issue of flammability, and showed that the tested fluorocarbons were not flammable in air in any proportion, though most of the tests were in pure oxygen or pure nitrous oxide (gases of importance in anesthesiology).[7][8]
| Compound | Test conditions | Result |
|---|---|---|
| Hexafluoroethane | Lower flammability limit in oxygen | None |
| Perfluoropentane | Flash point in air | None |
| Flash point in oxygen | −6 °C | |
| Flash point nitrous oxide | −32 °C | |
| Perfluoromethylcyclohexane | Lower flammability limit in air | None |
| Lower flammability limit in oxygen | 8.3% | |
| Lower flammability limit in oxygen (50 °C) | 7.4% | |
| Lower flammability limit in nitrous oxide | 7.7% | |
| Perfluoro-1,3-dimethylcyclohexane | Lower flammability limit in oxygen (50 °C) | 5.2% |
| Perfluoromethyldecalin | Spontaneous ignition test in oxygen at 127 bar |
No ignition at 500 °C |
| Spontaneous ignition in adiabatic shock wave in oxygen, 0.98 to 186 bar |
No ignition | |
| Spontaneous ignition in adiabatic shock wave in oxygen, 0.98 to 196 bar |
Ignition |
In 1993, 3M considered fluorocarbons as fire extinguishants to replace CFCs.[9] This extinguishing effect has been attributed to their high heat capacity, which takes heat away from the fire. It has been suggested that an atmosphere containing a significant percentage of perfluorocarbons on a space station or similar would prevent fires altogether.[10] [11] When combustion does occur, toxic fumes result, including carbonyl fluoride, carbon monoxide, and hydrogen fluoride.
Gas dissolving properties
Perfluorocarbons dissolve relatively high volumes of gases. The high solubility of gases is attributed to the weak intermolecular interactions in these fluorocarbon fluids.[12]
The table shows values for the mole fraction, x1, of nitrogen dissolved, calculated from the Blood–gas partition coefficient, at 298.15 K (25 °C), 0.101325 MPa.[13]
| Liquid | 104x1 | Concentration ( mM ) |
|---|---|---|
| Water | 0.118 | 0.65 |
| Ethanol | 3.57 | 6.12 |
| Tetrahydrofuran | 5.21 | 6.42 |
| Acetone | 5.42 | 7.32 |
| Cyclohexane | 7.73 | 7.16 |
| Perfluoro-1,3-dimethylcyclohexane | 31.9 | 14.6 |
| Perfluoromethylcyclohexane | 33.1 | 16.9 |
Manufacture
The development of the fluorocarbon industry coincided with World War II.[14] Prior to that, fluorocarbons were prepared by reaction of fluorine with the hydrocarbon, i.e., direct fluorination. Because C-C bonds are readily cleaved by fluorine, direct fluorination mainly affords smaller perfluorocarbons, such as tetrafluoromethane, hexafluoroethane, and octafluoropropane.[15]
Fowler process
A major breakthrough that allowed the large scale manufacture of fluorocarbons was the
- C6H14 + 28 CoF3 → C6F14 + 14 HF + 28 CoF2
The resulting cobalt difluoride is then regenerated, sometimes in a separate reactor:
- 2 CoF2 + F2 → 2 CoF3
Industrially, both steps are combined, for example in the manufacture of the Flutec range of fluorocarbons by F2 chemicals Ltd, using a vertical stirred bed reactor, with hydrocarbon introduced at the bottom, and fluorine introduced halfway up the reactor. The fluorocarbon vapor is recovered from the top.
Electrochemical fluorination
Electrochemical fluorination (ECF) (also known as the Simons' process) involves electrolysis of a substrate dissolved in hydrogen fluoride. As fluorine is itself manufactured by the electrolysis of hydrogen fluoride, ECF is a rather more direct route to fluorocarbons. The process proceeds at low voltage (5 – 6 V) so that free fluorine is not liberated. The choice of substrate is restricted as ideally it should be soluble in hydrogen fluoride. Ethers and tertiary amines are typically employed. To make perfluorohexane, trihexylamine is used, for example:
- N(C6H13)3 + 45 HF → 3 C6F14 + NF3 + 42 H2
The perfluorinated amine will also be produced:
- N(C6H13)3 + 39 HF → N(C6F13)3 + 39 H2
Environmental and health concerns
Fluoroalkanes are generally inert and non-toxic.[16][17][18]
Fluoroalkanes are not ozone depleting, as they contain no chlorine or bromine atoms, and they are sometimes used as replacements for ozone-depleting chemicals.[19] The term fluorocarbon is used rather loosely to include any chemical containing fluorine and carbon, including
Perfluoroalkanes do not bioaccumulate;[citation needed] those used in medical procedures are rapidly excreted from the body, primarily via expiration with the rate of excretion as a function of the vapour pressure; the half-life for octafluoropropane is less than 2 minutes,[20] compared to about a week for perfluorodecalin.[21]

Low-boiling perfluoroalkanes are potent
| Name | Chemical formula | Lifetime (y) | GWP (100 years) |
|---|---|---|---|
| PFC-14 | CF4 | 50000 | 6630 |
| PFC-116 | C2F6 | 10000 | 11100 |
| PFC-c216 | c-C3F6 | 3000 | 9200 |
| PFC-218 | C3F6 | 2600 | 8900 |
| PFC-318 | c-C4F8 | 3200 | 9540 |
The aluminium smelting industry has been a major source of atmospheric perfluorocarbons (
Applications
As they are inert, perfluoroalkanes have essentially no chemical uses, but their physical properties have led to their use in many diverse applications. These include:
- Perfluorocarbon tracer
- Liquid dielectric
- Chemical vapor deposition
- Organic Rankine cycle
- Fluorous biphasic catalysis[26]
- Cosmetics
- Ski waxes
As well as several medical uses:
- Contrast-enhanced ultrasound
- Oxygen Therapeutics
- Blood substitute
- Liquid breathing
- Eye surgery [27]
- Tattoo removal [28]
Fluoroalkenes and fluoroalkynes
- Unsaturated fluorocarbons
-
Perfluoroisobutene, a reactive and highly toxic fluoroalkene gas
-
Tetrafluoroethylene, an important perfluorinated monomer.
-
Hexafluoropropylene, another important perfluoroalkene.
-
Hexafluoro-2-butyne, a perfluoroalkyne.
Polymerization
Fluoroalkenes polymerize more exothermically than normal alkenes.
Environmental and health concerns
Fluoroalkenes and fluorinated alkynes are reactive and many are toxic for example
Perfluoroaromatic compounds
Perfluoroaromatic compounds contain only carbon and fluorine, like other fluorocarbons, but also contain an aromatic ring. The three most important examples are hexafluorobenzene, octafluorotoluene, and octafluoronaphthalene.
- Perfluoroaromatic compounds
Perfluoroaromatic compounds can be manufactured via the Fowler process, like fluoroalkanes, but the conditions must be adjusted to prevent full fluorination. They can also be made by heating the corresponding perchloroaromatic compound with potassium fluoride at high temperature (typically 500 °C), during which the chlorine atoms are replaced by fluorine atoms. A third route is defluorination of the fluoroalkane; for example, octafluorotoluene can be made from perfluoromethylcyclohexane by heating to 500 °C with a nickel or iron catalyst.[31]
Perfluoroaromatic compounds are relatively volatile for their molecular weight, with melting and boiling points similar to the corresponding aromatic compound, as the table below shows. They have high density and are non-flammable. For the most part, they are colorless liquids. Unlike the perfluoralkanes, they tend to be
| Compound | Melting point (°C) | Boiling point (°C) |
|---|---|---|
| Hexafluorobenzene | 5.3 | 80.5 |
| Benzene | 5.5 | 80.1 |
| Octafluorotoluene | <−70 | 102–103 |
| Toluene | −95 | 110.6 |
| Perfluoro(ethylbenzene) | 114–115 | |
| Ethylbenzene | −93.9 | 136.2 |
| Octafluoronaphthalene | 86–87 | 209[32] |
| Naphthalene | 80.2 | 217.9 |
See also
- Category:Fluorocarbons
- Fluorochemical industry
- Hydrofluorocarbon
- Fluorographene
- Perfluorocycloalkene (PFCA)
References
- ^ a b c d e f g h i j
Lemal DM (January 2004). "Perspective on fluorocarbon chemistry". J. Org. Chem. 69 (1): 1–11. PMID 14703372.
- ^ Murphy WJ (March 1947). "Fluorine Nomenclature... A statement by the Editors". Ind. Eng. Chem. 39 (3): 241–242. .
- ^
O'Hagan D (February 2008). "Understanding organofluorichemistry. An introduction to the C–F bond". Chem. Soc. Rev. 37 (2): 308–19. PMID 18197347.
- ^ Kiplinger JL, Richmond TG, Osterberg CE (1994). "Activation of Carbon-Fluorine Bonds by Metal Complexes". Chem. Rev. 94 (2): 373–431. .
- ^ "Archived copy" (PDF). Archived from the original (PDF) on 2008-12-05. Retrieved 2008-11-29.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ Larsen ER (1969). "Fluorine Compounds in Anesthesiology: VI Flammability". Fluorine Chem. Rev. 3: 22–27.
- ^ Flutec (Technical report). ISC Chemicals Limited. 1982.
- ^ John A. Pignato, Jr.; Paul E. Rivers; Myron T. Pike. "Development of Perfluorocarbons As Clean Extinguishing Agents" (PDF). National Institute of Standards and Technology. Archived from the original (PDF) on 2014-05-21. Retrieved 2019-01-03.
- S2CID 111161665.
- .
- ^ "Dissolving gases in FLUTEC liquids" (PDF). F2 Chemicals Ltd. 2005-05-10.
- doi:10.1063/1.555713.
- .
- ^ "EFCTC - Toxicological profiles of PFCS Perfluorocarbons". Archived from the original on 2015-09-24. Retrieved 2014-05-19.
- ^ "HPV Robust Summaries and Test Plan" (PDF). Internet Archive. Archived from the original (PDF) on 2012-12-02. Retrieved 2019-01-03.
- ^ Yamanouchi K; Yokoyama K (1975). "Proceedings of the Xth International Congress for Nutrition: Symposium on Perfluorochemical Artificial Blood, Kyoto": 91.
{{cite journal}}: Cite journal requires|journal=(help) - ^ "Perfluorocarbons (PFCS) definition - ExpertGlossary". Archived from the original on 2014-05-19. Retrieved 2022-12-14.
- PMID 21355829.
- ^ Geyer RP (1975). "Proc. Xth Intern. Congress for Nutr.: Symp on Perfluorochemical Artif. Blood, Kyoto": 3–19.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Change, United Nations Framework Convention on Climate. "Kyoto Protocol". unfccc.int. Retrieved 2017-09-27.
- ^ Myhre, G., D. Shindell, F.-M. Bréon, W. Collins, J. Fuglestvedt, J. Huang, D. Koch, J.-F. Lamarque, D. Lee, B. Mendoza, T. Nakajima, A. Robock, G. Stephens, T. Takemura and H. Zhang (2013) "Anthropogenic and Natural Radiative Forcing" (see Table 8.A.1). In: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Stocker, T.F., D. Qin, G.-K. Plattner, M. Tignor, S.K. Allen, J. Boschung, A. Nauels, Y. Xia, V. Bex and P.M. Midgley (eds.). Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA.
- ^ "The Anode Effect". aluminum-production.com. Archived from the original on 2019-02-22. Retrieved 2014-05-20.
- ^ Leber BP, et al. "Perfluorocarbon (PFC) Generation at Primary Aluminum Smelters" (PDF). Archived from the original (PDF) on 2013-02-16. climatevision.gov
- ^ Flannigan, David J. (21 November 2002). "Fluorous Biphasic Catalysis" (PDF). chemistry.illinois.edu.
- PMID 12714393.
- ^ "Archived copy" (PDF). Archived from the original (PDF) on 2014-05-19. Retrieved 2014-05-19.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ "Schedule 2 of Chemical Weapons Convention". OPCW. Retrieved 2022-01-25.
- ISBN 9780080434056.
- ISBN 978-0-356-02798-2.
- ^ "Octafluoronaphthalene". ChemSpider.
External links
- Fluorocarbons and Sulphur Hexafluoride, proposed by the European Fluorocarbons Technical Committee
- CFCs and Ozone Depletion Freeview video provided by the Vega Science Trust.
- Introduction to fluoropolymers
- Organofluorine chemistry by Graham Sandford[permanent dead link]