Period 2 element
| Part of a series on the |
| Periodic table |
|---|
A period 2 element is one of the
The second period contains the elements
Periodic trends
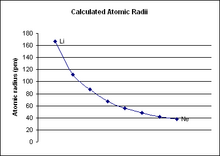
Period 2 is the first period in the periodic table from which periodic trends can be drawn. Period 1, which only contains two elements (hydrogen and helium), is too small to draw any conclusive trends from it, especially because the two elements behave nothing like other s-block elements.[1][2] Period 2 has much more conclusive trends. For all elements in period 2, as the atomic number increases, the atomic radius of the elements decreases, the electronegativity increases, and the ionization energy increases.[3]
Period 2 only has two metals (lithium and beryllium) of eight elements, less than for any subsequent period both by number and by proportion. It also has the most number of nonmetals, namely five, among all periods. The elements in period 2 often have the most extreme properties in their respective groups; for example, fluorine is the most reactive halogen, neon is the most inert noble gas,[4] and lithium is the least reactive alkali metal.[5]
All period 2 elements completely obey the
Chemical element Block Electron configuration 3 Li Lithium s-block[He] 2s1 4 Be Beryllium s-block[He] 2s2 5 B Boron p-block[He] 2s2 2p1 6 C Carbon p-block[He] 2s2 2p2 7 N Nitrogen p-block[He] 2s2 2p3 8 O Oxygen p-block[He] 2s2 2p4 9 F Fluorine p-block[He] 2s2 2p5 10 Ne Neon p-block[He] 2s2 2p6
Lithium

Lithium (Li) is an
Lithium is one of the few elements synthesized in the Big Bang. Lithium is the 31st most abundant element on earth,[7] occurring in concentrations of between 20 and 70 ppm by weight,[8] but due to its high reactivity it is only found naturally in compounds.[8]
Lithium
Beryllium

Beryllium (Be) is the chemical element with atomic number 4, occurring in the form of 9Be. At standard temperature and pressure, beryllium is a strong, steel-grey, light-weight,
Small amounts of beryllium were
Due to its stiffness, light weight, and dimensional stability over a wide temperature range, beryllium metal is used in as a structural material in aircraft, missiles and
Beryllium and beryllium compounds are classified by the
Boron

Boron (B) is the chemical element with atomic number 5, occurring as 10B and 11B. At standard temperature and pressure, boron is a
Boron does not occur naturally as a free element, but in compounds such asBoron is an essential plant
Carbon
Carbon is the chemical element with atomic number 6, occurring as 12C, 13C and 14C.
Carbon is the fourth most abundant element in the universe by mass after
Nitrogen

Nitrogen is the chemical element with atomic number 7, the symbol N and
Many industrially important compounds, such as ammonia, nitric acid, organic nitrates (propellants and explosives), and cyanides, contain nitrogen. The extremely strong bond in elemental nitrogen dominates nitrogen chemistry, causing difficulty for both organisms and industry in breaking the bond to convert the N
2 molecule into useful compounds, but at the same time causing release of large amounts of often useful energy when the compounds burn, explode, or decay back into nitrogen gas.
Nitrogen occurs in all living organisms, and the nitrogen cycle describes movement of the element from air into the biosphere and organic compounds, then back into the atmosphere. Synthetically produced nitrates are key ingredients of industrial fertilizers, and also key pollutants in causing the eutrophication of water systems. Nitrogen is a constituent element of amino acids and thus of proteins, and of nucleic acids (DNA and RNA). It resides in the chemical structure of almost all neurotransmitters, and is a defining component of alkaloids, biological molecules produced by many organisms.[38]
Oxygen
Oxygen is the chemical element with atomic number 8, occurring mostly as 16O, but also 17O and 18O.
Oxygen is the third-most common element by mass in the universe (although there are more carbon atoms, each carbon atom is lighter). It is highly electronegative and non-metallic, usually diatomic, gas down to very low temperatures. Only fluorine is more reactive among non-metallic elements. It is two electrons short of a full octet and readily takes electrons from other elements. It reacts violently with
Oxygen is essential to all life. Plants and phytoplankton photosynthesize water and carbon dioxide and water, both oxides, in the presence of sunlight to form sugars with the release of oxygen. The sugars are then turned into such substances as cellulose and (with nitrogen and often sulfur) proteins and other essential substances of life. Animals especially but also fungi and bacteria ultimately depend upon photosynthesizing plants and phytoplankton for food and oxygen.
Fire uses oxygen to oxidize compounds typically of carbon and hydrogen to water and carbon dioxide (although other elements may be involved) whether in uncontrolled conflagrations that destroy buildings and forests or the controlled fire within engines or that supply electrical energy from turbines, heat for keeping buildings warm, or the motive force that drives vehicles.
Oxygen forms roughly 21% of the Earth's atmosphere; all of this oxygen is the result of photosynthesis. Pure oxygen has use in medical treatment of people who have respiratory difficulties. Excess oxygen is toxic.
Oxygen was originally associated with the formation of acids—until some acids were shown to not have oxygen in them. Oxygen is named for its formation of acids, especially with non-metals. Some oxides of some non-metals are extremely acidic, like sulfur trioxide, which forms sulfuric acid on contact with water. Most oxides with metals are alkaline, some extremely so, like potassium oxide. Some metallic oxides are amphoteric, like aluminum oxide, which means that they can react with both acids and bases.
Although oxygen is normally a diatomic gas, oxygen can form an allotrope known as ozone. Ozone is a triatomic gas even more reactive than oxygen. Unlike regular diatomic oxygen, ozone is a toxic material generally considered a pollutant. In the upper atmosphere, some oxygen forms ozone which has the property of absorbing dangerous ultraviolet rays within the ozone layer. Land life was impossible before the formation of an ozone layer.
Fluorine

This section needs expansion. You can help by adding to it. (May 2011) |
Fluorine is the chemical element with atomic number 9. It occurs naturally in its only stable form 19F.[39]
Fluorine is a pale-yellow, diatomic gas under normal conditions and down to very low temperatures. Short one electron of the highly stable octet in each atom, fluorine molecules are unstable enough that they easily snap, with loose fluorine atoms tending to grab single electrons from just about any other element. Fluorine is the most reactive of all elements, and it even attacks many oxides to replace oxygen with fluorine. Fluorine even attacks silica, one of the favored materials for transporting strong acids, and burns asbestos. It attacks common salt, one of the most stable compounds, with the release of chlorine. It never appears uncombined in nature and almost never stays uncombined for long. It burns hydrogen simultaneously if either is liquid or gaseous—even at temperatures close to absolute zero.[40] It is extremely difficult to isolate from any compounds, let alone keep uncombined.
Fluorine gas is extremely dangerous because it attacks almost all organic material, including live flesh. Many of the binary compounds that it forms (called fluorides) are themselves highly toxic, including soluble fluorides and especially
Neon
This section needs expansion. You can help by adding to it. (May 2011) |
Neon is the chemical element with atomic number 10, occurring as 20Ne, 21Ne and 22Ne.[41]
Neon is a monatomic gas. With a complete octet of outer electrons it is highly resistant to removal of any electron, and it cannot accept an electron from anything. Neon has no tendency to form any normal compounds under normal temperatures and pressures; it is effectively inert. It is one of the so-called "noble gases".
Neon is a trace component of the atmosphere without any biological role.
Notes
References
- S2CID 93781427.
- ^ "International Union of Pure and Applied Chemistry > Periodic Table of the Elements". IUPAC. Retrieved 2011-05-01.
- ISBN 978-0-495-12671-3.
- .
- ^ ISBN 978-1-57912-814-2.
- ^ a b Lithium at WebElements.
- ISBN 0-313-33438-2.
- ^
- PMID 18142718.
- PMID 10885179.
- PMID 17042835.
- ^ a b c d e Beryllium at WebElements.
- ^ Standards and properties of beryllium copper.
- ^ Information about beryllium tweeters.
- ^ "IARC Monograph, Volume 58". International Agency for Research on Cancer. 1993. Retrieved 2008-09-18.
- ^ Information about chronic beryllium disease.
- ^ a b c Boron at WebElements.
- ^ a b c Properties of boron.
- ^ W.T.M.L. Fernando; L.C. O'Brien; P.F. Bernath. "Fourier Transform Spectroscopy: B4Σ−−X4Σ−" (PDF). University of Arizona, Tucson. Retrieved 2007-12-10.[permanent dead link]
- ^ K.Q. Zhang, B.Guo, V. Braun, M. Dulick, P.F. Bernath. "Infrared Emission Spectroscopy of BF and AIF" (PDF). Retrieved 2007-12-10.
{{cite web}}: CS1 maint: multiple names: authors list (link)[permanent dead link] - ^ "Compound Descriptions: B2F4". Landol Börnstein Substance/Property Index. Retrieved 2007-12-10.
- PMID 15012243.
- ^ Zook EG, Lehman J (1965). "850-5". J. Assoc. Off Agric. Chem. 48.
- ^ "Boron". PDRhealth. Archived from the original on October 11, 2007. Retrieved 2008-09-18.
- ^ a b c d Carbon at WebElements.
- ^ "Amorphous carbon". IUPAC Compendium of Chemical Terminology (2nd ed.). International Union of Pure and Applied Chemistry. 1997. Retrieved 2008-09-24.
- ^ Vander Wal, R. (May 1996). "Soot Precursor Material: Spatial Location via Simultaneous LIF-LII Imaging and Characterization via TEM" (PDF). NASA Contractor Report (198469). Retrieved 2008-09-24.[dead link]
- ^ "diamond-like carbon films". IUPAC Compendium of Chemical Terminology (2nd ed.). International Union of Pure and Applied Chemistry. 1997. Retrieved 2008-09-24.
- ^ a b Presentation about isotopes Archived 2008-07-19 at the Wayback Machine by Mahananda Dasgupta of the Department of Nuclear Physics at Australian National University.
- .
- ^ Ten most abundant elements in the universe, taken from The Top 10 of Everything, 2006, Russell Ash, page 10. Retrieved October 15, 2008. Archived February 10, 2010, at the Wayback Machine
- ISBN 978-0-07-110595-8.
- ISBN 1-57059-680-8.
- ^ a b c "Structure and Nomenclature of Hydrocarbons". Purdue University. Retrieved 2008-03-23.
- ^ a b c d Alberts, Bruce; Alexander Johnson; Julian Lewis; Martin Raff; Keith Roberts; Peter Walter (2002). Molecular Biology of the Cell. Garland Science.
- ISBN 0-486-64624-6.
- ^ Nitrogen at WebElements.
- ISBN 978-0-521-03541-5.
- ^ National Nuclear Data Center. "NuDat 2.1 database – fluorine-19". Brookhaven National Laboratory. Retrieved 2011-05-01.
- ^ "WebElements Periodic Table » Fluorine » the essentials". www.webelements.com.
- ^ "Neon: Isotopes". Softciências. Archived from the original on 2012-11-15. Retrieved 2011-05-01.
External links
 Media related to Periodic table row 2 at Wikimedia Commons
Media related to Periodic table row 2 at Wikimedia Commons
