Periodic acid
Orthoperiodic acid
| |||
Metaperiodic acid
| |||
 HIO4·2H2O
| |||
| Names | |||
|---|---|---|---|
Other names
| |||
| Identifiers | |||
| |||
3D model (
JSmol ) |
| ||
| ChemSpider | |||
ECHA InfoCard
|
100.030.839 | ||
| EC Number |
| ||
PubChem CID
|
| ||
| UNII |
| ||
| UN number | UN3085 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| HIO4 (metaperiodic) H5IO6 (orthoperiodic) | |||
| Molar mass | 190.91 g/mol (HIO4) 227.941 g/mol (H5IO6) | ||
| Appearance | Colourless crystals | ||
| Melting point | 128.5 °C (263.3 °F; 401.6 K)[1] | ||
| Solubility | soluble in water, alcohols | ||
Conjugate base
|
Periodate | ||
| Hazards[2] | |||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H271, H314, H372, H400 | |||
| P210, P260, P273, P303+P361+P353, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Other anions
|
| ||
Other cations
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Periodic acid (
Periodic acid was discovered by Heinrich Gustav Magnus and C. F. Ammermüller in 1833.[3]
Synthesis
Modern industrial scale production involves the
- IO−3 + 6 HO− - 2 e− → IO5−6 + 3 H2O (
- IO−3 + 6 HO− + Cl2 → IO5−6 + 2 Cl− + 3 H2O
Orthoperiodic acid can be dehydrated to give metaperiodic acid by heating to 100 °C under reduced pressure.
- H5IO6 ⇌ HIO4 + 2 H2O
Further heating to around 150 °C gives
Properties
Orthoperiodic acid has a number of acid dissociation constants.[7][8] The pKa of metaperiodic acid has not been determined.
- H5IO6 ⇌ H4IO−6 + H+, pKa = 3.29
- H4IO−6 ⇌ H3IO2−6 + H+, pKa = 8.31
- H3IO2−6 ⇌ H2IO3−6 + H+, pKa = 11.60
There being two forms of periodic acid, it follows that two types of periodate salts are formed. For example, sodium metaperiodate, NaIO4, can be synthesised from HIO4 while sodium orthoperiodate, Na5IO6 can be synthesised from H5IO6.
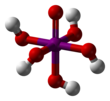
Structure
Orthoperiodic acid forms
Reactions
Like all periodates periodic acid can be used to cleave various 1,2-difunctional compounds. Most notably periodic acid will cleave vicinal diols into two aldehyde or ketone fragments (Malaprade reaction).

This can be useful in determining the structure of
Periodic acid is also used as an
Other oxyacids
Periodate is part of a series of oxyacids in which iodine can assume oxidation states of −1, +1, +3, +5, or +7. A number of neutral iodine oxides are also known.
| Iodine oxidation state | −1 | +1 | +3 | +5 | +7 |
|---|---|---|---|---|---|
| Name | Hydrogen iodide | Hypoiodous acid | Iodous acid
|
Iodic acid | Periodic acid |
| Formula | HI | HIO | HIO2 | HIO3 | HIO4 or H5IO6 |
See also
Compounds with a similar structure:
- Perchloric acid, perbromic acid, the related perhalogenic acids
- perxenic acid, the isoelectronic oxoacids of tellurium and xenon
Compounds with similar chemistry:
- lead tetraacetate (Criegee oxidation)
References
- ISBN 0123526515.)
{{cite book}}: CS1 maint: multiple names: authors list (link - ^ "Periodsaeure zur Synthese". Sigma Aldrich. 8 October 2021. Retrieved 21 November 2021.
- .
- ISBN 978-0-7506-3365-9.
- ^ Parsons, Roger (1959). Handbook of electrochemical constants. Butterworths Scientific Publications Ltd. p. 71.
- ISBN 012126601X.
- ISBN 0123526515.)
{{cite book}}: CS1 maint: multiple names: authors list (link - ISBN 978-1441983824.
- .
- .
- .
- .