Periodic trends
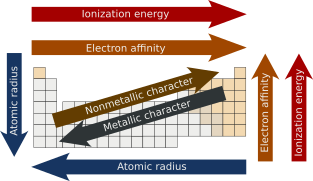
In chemistry, periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of certain elements when grouped by period and/or group. They were discovered by the Russian chemist Dmitri Mendeleev in 1863. Major periodic trends include atomic radius, ionization energy, electron affinity, electronegativity, valency and metallic character. These trends exist because of the similar electron configurations of the elements within their respective groups or periods; they reflect the periodic nature of the elements. These trends give a qualitative assessment of the properties of each element.[1][2]
Summary of trends
| Periodic property | Across the period | Down the group |
|---|---|---|
| Atomic radius | Decreases | Increases |
| Ionization energy | Increases | Decreases |
| Electron affinity | Increases | Decreases |
| Electronegativity | Increases | Decreases |
| Valency | Increases | Constant |
| Nonmetallic character | Increases | Decreases |
| Metallic character | Decreases | Increases |
| Nuclear Charge | Increases | Increases |
Atomic radius
The atomic radius is the distance from the atomic nucleus to the outermost electron orbital in an atom. In general, the atomic radius decreases as we move from left to right in a period, and it increases when we go down a group. This is because in periods, the valence electrons are in the same outermost shell. The atomic number increases within the same period while moving from left to right, which in turn increases the effective nuclear charge. The increase in attractive forces reduces the atomic radius of elements. When we move down the group, the atomic radius increases due to the addition of a new shell.[3][4]
Ionization energy
The ionization energy is the minimum amount of
Trend-wise, as one moves from left to right across a period in the modern periodic table, the ionization energy increases as the nuclear charge increases and the atomic size decreases. The decrease in the atomic size results in a more potent force of attraction between the electrons and the nucleus. However, suppose one moves down in a group. In that case, the ionization energy decreases as atomic size increases due to adding a valence shell, thereby diminishing the nucleus's attraction to electrons.[6][7]
Electron affinity
The
Electronegativity
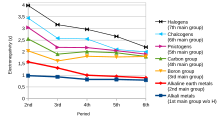
The tendency of an
Trend-wise, as one moves from left to right across a
However, in group XIII (Boron Family), the electronegativity first decreases from boron to aluminium and then increases down the group. It is due to the fact that the atomic size increases as we move down the group, but at the same time the effective nuclear charge increases due to poor shielding of the inner d and f electrons. As a result, the force of attraction of the nucleus for the electrons increases and hence the electronegativity increases from aluminium to thallium.[13][14]
Valency
The valency of an element is the number of electrons that must be lost or gained by an atom to obtain a stable electron configuration. In simple terms, it is the measure of the combining capacity of an element to form chemical compounds. Electrons found in the outermost shell are generally known as valence electrons; the number of valence electrons determines the valency of an atom.[15][16]
Trend-wise, while moving from left to right across a
Metallic and non-metallic properties
Metallic properties generally increase down the groups, as decreasing attraction between the nuclei and outermost electrons causes these electrons to be more loosely bound and thus able to conduct heat and electricity. Across each period, from left to right, the increasing attraction between the nuclei and the outermost electrons causes the metallic character to decrease. In contrast, the nonmetallic character decreases down the groups and increases across the periods.[19][20]
See also
References
- S2CID 211038510.
- S2CID 213379908, retrieved 2022-07-02
- ^ "atomic and ionic radius". www.chemguide.co.uk. Retrieved 2022-06-30.
- .
- ^ "7.4: Ionization Energy". Chemistry LibreTexts. 2014-11-18. Retrieved 2022-07-02.
- ^ "Ionization Energy Trend | Science Trends". sciencetrends.com. 2018-05-18. Retrieved 2022-07-02.
- S2CID 198913558.
- ISBN 978-0-387-30160-0, retrieved 2022-07-02
- ^ "Electron Affinity Trend | Science Trends". sciencetrends.com. 2018-05-14. Retrieved 2022-07-02.
- . Retrieved 2022-06-30.
- ISBN 978-3-319-39312-4, retrieved 2022-06-30
- ISBN 978-3-540-17740-1, retrieved 2022-06-30
- ^ "21.1: The Elements of Group 13". Libretexts. 2013-11-26. Retrieved 2022-06-30.
- PMID 27216700.
- ISBN 978-3-540-72816-0, retrieved 2022-07-02
- ISBN 978-3-642-54596-2, retrieved 2022-07-02
- ISBN 978-0-387-90268-5.
- ISBN 978-1-4612-6262-6, retrieved 2022-07-02
- ISSN 0920-2307.
- ^ "C9.1 – Periodic Trends". IGCSE AID. 2018-03-05. Retrieved 2022-07-02.
