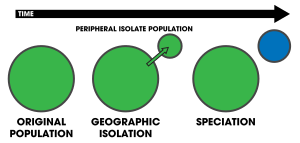
Peripatric speciation
| Part of a series on |
| Evolutionary biology |
|---|
 |
Peripatric speciation is a mode of
The concept of peripatric speciation was first outlined by the evolutionary biologist
The existence of peripatric speciation is supported by observational evidence and laboratory experiments.
History
Peripatric speciation was originally proposed by
In what has been called Mayr's genetic revolutions, he postulated that genetic drift played the primary role that resulted in this pattern.[1]: 389 Seeing that a species cohesion is maintained by conservative forces such as epistasis and the slow pace of the spread of favorable alleles in a large population (based heavily on J. B. S. Haldane's calculations), he reasoned that speciation could only take place in which a population bottleneck occurred.[1]: 389 A small, isolated, founder population could be established on an island for example. Containing less genetic variation from the main population, shifts in allele frequencies may occur from different selection pressures.[1]: 390 This to further changes in the network of linked loci, driving a cascade of genetic change, or a "genetic revolution"—a large-scale reorganization of the entire genome of the peripheral population.[1]: 391 Mayr did recognize that the chances of success were incredibly low and that extinction was likely; though noting that some examples of successful founder populations existed at the time.[7]: 522
Shortly after Mayr, William Louis Brown, Jr. proposed an alternative model of peripatric speciation in 1957 called centrifugal speciation. In 1976 and 1980, the Kaneshiro model of peripatric speciation was developed by Kenneth Y. Kaneshiro which focused on sexual selection as a driver for speciation during population bottlenecks.[9][10][11]
Models
Peripatric
Peripatric speciation models are identical to models of
- The size of the isolated population
- Strong selection caused by the dispersal and colonization of novel environments,
- The effects of genetic drift on small populations.
The size of a population is important because individuals colonizing a new habitat likely contain only a small sample of the genetic variation of the original population. This promotes divergence due to strong selective pressures, leading to the rapid fixation of an allele within the descendant population. This gives rise to the potential for genetic incompatibilities to evolve. These incompatibilities cause reproductive isolation, giving rise to—sometimes rapid—speciation events.[1]: 105 Furthermore, two important predictions are invoked, namely that geological or climatic changes cause populations to become locally fragmented (or regionally when considering allopatric speciation), and that an isolated population's reproductive traits evolve enough as to prevent interbreeding upon potential secondary contact.[13]
The peripatric model results in, what have been called, progenitor-derivative species pairs, whereby the derivative species (the peripherally isolated population)—geographically and genetically isolated from the progenitor species—diverges.
Modern cladistic methods have developed definitions that have incidentally removed derivative species by defining clades in a way that assumes that when a speciation event occurs, the original species no longer exists, while two new species arise; this is not the case in peripatric speciation.[8] Mayr warned against this, as it causes a species to lose their classification status.[17] Loren H. Rieseberg and Luc Brouillet recognized the same dilemma in plant classification.[18]
Quantum and budding speciation
The botanist Verne Grant proposed the term quantum speciation that combined the ideas of J. T. Gulick (his observation of the variation of species in semi-isolation), Sewall Wright (his models of genetic drift), Mayr (both his peripatric and genetic revolution models), and George Gaylord Simpson (his development of the idea of quantum evolution).[19]: 114 Quantum speciation is a rapid process with large genotypic or phenotypic effects, whereby a new, cross-fertilizing plant species buds off from a larger population as a semi-isolated peripheral population.[20][19]: 114 Interbreeding and genetic drift takes place due to the reduced population size, driving changes to the genome that would most likely result in extinction (due to low adaptive value).[19]: 115 In rare instances, chromosomal traits with adaptive value may arise, resulting in the origin of a new, derivative species.[8][21] Evidence for the occurrence of this type of speciation has been found in several plant species pairs: Layia discoidea and L. glandulosa, Clarkia lingulata and C. biloba, and Stephanomeria malheurensis and S. exigua ssp. coronaria.[8]
A closely related model of peripatric speciation is called budding speciation—largely applied in the context of plant speciation.[22] The budding process, where a new species originates at the margins of an ancestral range, is thought to be common in plants[22]—especially in progenitor-derivative species pairs.[23]
Centrifugal speciation
William Louis Brown, Jr. proposed an alternative model of peripatric speciation in 1957 called centrifugal speciation. This model contrasts with peripatric speciation by virtue of the origin of the genetic novelty that leads to reproductive isolation.
Kaneshiro model

The Kaneshiro Model also provides an explanation of the mechanism of speciation during founder events as proposed by Ernst Mayr and Hampton Carson. In most cases, founder events result when single fertilized female is accidentally translocated to an entirely different location, e.g., an adjacent island among a chain of islands such as the Hawaiian Archipelago, and produces a few offspring. Such a founder colony is faced with extremely small population size which as described by the Kaneshiro Model, experiences a shift in the mating system towards and increase in frequency of less choosy females. The resulting destabilization of the genetic system provides the milieu for new genetic variants to arise providing the recipe for speciation to occur. Eventually, a growth in population size paired with novel female mate preferences will give rise to reproductive isolation from the main population-thereby completing the speciation process.[10] Support for this model comes from experiments and observation of species that exhibit asymmetric mating patterns such as the Hawaiian Drosophila species[30][31] or the Hawaiian cricket Laupala.[32] However, while laboratory experiments are ongoing and yet to be completed in support of the model, there are field observations of shifts in the mating systems that undergo population bottlenecks which demonstrate that the dynamics of sexual selection is occurring in nature and therefore, it does represent a plausible process of peripatric speciation that takes place in nature.[11]
Evidence
Observational evidence and laboratory experiments support the occurrence of peripatric speciation. Islands and archipelagos are often the subject of speciation studies in that they represent isolated populations of organisms. Island species provide direct evidence of speciation occurring peripatrically in such that, "the presence of endemic species on oceanic islands whose closest relatives inhabit a nearby continent" must have originated by a colonization event.[1]: 106–107 Comparative phylogeography of oceanic archipelagos shows consistent patterns of sequential colonization and speciation along island chains, most notably on the Azores islands, Canary Islands, Society Islands, Marquesas Islands, Galápagos Islands, Austral Islands, and the Hawaiian Islands—all of which express geological patterns of spatial isolation and, in some cases, linear arrangement.[33] Peripatric speciation also occurs on continents, as isolation of small populations can occur through various geographic and dispersion events. Laboratory studies have been conducted where populations of Drosophila, for example, are separated from one another and evolve in reproductive isolation.
Hawaiian archipelago
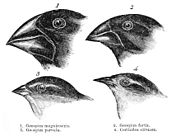
Other endemic species in Hawaii also provide evidence of peripatric speciation such as the endemic flightless crickets (
A host of other Hawaiian endemic arthropod species and genera have had their speciation and phylogeographical patterns studied: the
Other islands
Phylogenetic studies of a species of crab spider (Misumenops rapaensis) in the genus Thomisidae located on the Austral Islands have established the, "sequential colonization of [the] lineage down the Austral archipelago toward younger islands". M. rapaensis has been traditionally thought of as a single species; whereas this particular study found distinct genetic differences corresponding to the sequential age of the islands.[54] The figwart plant species Scrophularia lowei is thought to have arisen through a peripatric speciation event, with the more widespread mainland species, Scrophularia arguta dispersing to the Macaronesian islands.[55][56] Other members of the same genus have also arisen by single colonization events between the islands.[57][58]
Species patterns on continents
The occurrence of peripatry on continents is more difficult to detect due to the possibility of vicariant explanations being equally likely.[1]: 110 However, studies concerning the Californian plant species Clarkia biloba and C. lingulata strongly suggest a peripatric origin.[59] In addition, a great deal of research has been conducted on several species of land snails involving chirality that suggests peripatry (with some authors noting other possible interpretations).[1]: 111
The chestnut-tailed antbird (Sciaphylax hemimelaena) is located within the Noel Kempff Mercado National Park (Serrania de Huanchaca) in Bolivia. Within this region exists a forest fragment estimated to have been isolated for 1000–3000 years. The population of S. hemimelaena antbirds that reside in the isolated patch express significant song divergence; thought to be an "early step" in the process of peripatric speciation. Further, peripheral isolation "may partly explain the dramatic diversification of suboscines in Amazonia".[13]
The montane spiny throated reed frog species complex (genus: Hyperolius) originated through occurrences of peripatric speciation events. Lucinda P. Lawson maintains that the species' geographic ranges within the Eastern Afromontane Biodiversity Hotspot support a peripatric model that is driving speciation; suggesting that this mode of speciation may play a significant role in "highly fragmented ecosystems".[2]
In a study of the phylogeny and biogeography of the land snail genus Monacha, the species M. ciscaucasica is thought to have speciated peripatrically from a population of M. roseni. In addition, M. claussi consists of a small population located on the peripheral of the much larger range of M. subcarthusiana suggesting that it also arose by peripatric speciation.[60]
Red spruce (
Using a phylogeographic approach paired with
Laboratory experiments
| Species | Replicates | Year |
|---|---|---|
| Drosophila adiastola | 1 | 1979[69] |
| Drosophila silvestris | 1 | 1980[70] |
| Drosophila pseudoobscura | 8 | 1985[71] |
| Drosophila simulans | 8 | 1985[72] |
Musca domestica
|
6 | 1991[73] |
| Drosophila pseudoobscura | 42 | 1993[74] |
| Drosophila melanogaster | 50 | 1998[75] |
| Drosophila melanogaster | 19; 19 | 1999[76] |
| Drosophila grimshawi | 1 | N/A[11] |
Peripatric speciation has been researched in both laboratory studies and nature. Jerry Coyne and H. Allen Orr in Speciation suggest that most laboratory studies of allopatric speciation are also examples of peripatric speciation due to their small population sizes and the inevitable divergent selection that they undergo.[1]: 106 Much of the laboratory research concerning peripatry is inextricably linked to founder effect research. Coyne and Orr conclude that selection's role in speciation is well established, whereas genetic drift's role is unsupported by experimental and field data—suggesting that founder-effect speciation does not occur.[1]: 410 Nevertheless, a great deal of research has been conducted on the matter, and one study conducted involving bottleneck populations of Drosophila pseudoobscura found evidence of isolation after a single bottleneck.[77][78]
The table is a non-exhaustive table of laboratory experiments focused explicitly on peripatric speciation. Most of the studies also conducted experiments on vicariant speciation as well. The "replicates" column signifies the number of lines used in the experiment—that is, how many independent populations were used (not the population size or the number of generations performed).[11]
References
![]() This article was submitted to WikiJournal of Science for external academic peer review in 2018 (reviewer reports). The updated content was reintegrated into the Wikipedia page under a CC-BY-SA-3.0 license (2018). The version of record as reviewed is:
Andrew Z Colvin; et al. (14 August 2018). "Peripatric speciation" (PDF). WikiJournal of Science. 1 (2): 008.
This article was submitted to WikiJournal of Science for external academic peer review in 2018 (reviewer reports). The updated content was reintegrated into the Wikipedia page under a CC-BY-SA-3.0 license (2018). The version of record as reviewed is:
Andrew Z Colvin; et al. (14 August 2018). "Peripatric speciation" (PDF). WikiJournal of Science. 1 (2): 008. {{cite journal}}: CS1 maint: unflagged free DOI (link
- ^ ISBN 978-0-87893-091-3
- ^ )
- ^ a b Ernst Mayr. (1954). Change of genetic environment and evolution. In J. Huxley, A. C. Hardy & E. B. Ford. (eds) Evolution as a Process, Unwin Brothers, London. Pp. 157–180.
- ^ Ernst Mayr. (1982). Processes of speciation in animals. In A. R. I. Liss. (eds) Mechanisms of Speciation, Alan R. Liss Inc., New York. Pp. 1–19.
- PMID 15280221.
- PMID 6777243.
- ^ a b Ernst Mayr (1963), Animal Species and Evolution, Harvard University Press, pp. 1–797
- ^
- S2CID 205773169
- ^ S2CID 28701838
- ^ S2CID 82095639
- )
- ^
- ISBN 978-0-520-26868-5.
- ^ S2CID 32546573
- JSTOR 2445111
- JSTOR 1223457
- ^ ISBN 978-0231083263
- S2CID 84541831
- PMID 11403867
- ^ PMID 24452025
- ^ )
- ^ )
- S2CID 225071133
- ISSN 1948-6596
- ^ S2CID 86734208
- S2CID 27865356
- S2CID 45100702
- S2CID 38590572
- PMID 27432948
- ISBN 9783527600908)
{{citation}}: CS1 maint: multiple names: authors list (link - ^ DeSalle R. (1995). Molecular approaches to biogeographic analysis of Hawaiian Drosophilidae. Pp. 72-89 in W.L. Wagner and V.A. Funk (eds.) Hawaiian Biogeography: Evolution on a Hot-Spot Archipeligo. Smithsonian Institution Press, Washington DC.
- ^ T. J. Givnish (1998). "Adaptive plant evolution on islands: classical patterns, molecular data, new insights". Evolution on Islands. 281: 304.
- ^ T. J. Givnish, K. J. Sytsma, W. J. Hahn, and J. F. Smith. (1995). Molecular evolution, adaptive radiation, and geographic speciation in Cyanea (Campanulaceae, Lobeliodeae). Pp. 259-301 in W.L. Wagner and V.A. Funk (eds.) Hawaiian Biogeography: Evolution on a Hot-Spot Archipeligo. Smithsonian Institution Press, Washington DC.
- )
- PMID 12451181
- JSTOR 2399630
- S2CID 32193624
- )
- ^ Rosemary G. Gillespie & H. B. Croom. (1995). Comparison of speciation mechanisms in web-building and non-web-building groups within a lineage of spiders. In W.L. Wagner & V.A. Funk (eds.) Hawaiian Biogeography: Evolution on a Hot-Spot Archipeligo, Smithsonian Institution Press, Washington DC. Pp. 121-146.
- S2CID 11856750
- S2CID 7748888
- )
- ^ Rosemary G. Gillespie, Malia A. J. Rivera, & Jessica E. Garb. (1998). Sun, surf and spiders: taxonomy and phylogeography of Hawaiian Araneae. Proceedings of the 17th European Colloquium of Arachnology.
- ^ Jessica E. Garb (1999), "An Adaptive Radiation of Hawaiian Thomisidae: Biogreographic and Genetic Evidence", The Journal of Arachnology, 27: 71–78
- ^ W. J. Gertsch (1973), "The cavernicolous fauna of Hawaiian lava tubes. 3. Araneae (Spiders)", Pacific Insects, 15: 163–180
- )
- )
- )
- S2CID 29359389
- S2CID 43087290
- )
- )
- doi:10.1016/j.ppees.2015.05.002)
{{citation}}: CS1 maint: multiple names: authors list (link - PMID 24096055
- JSTOR 2405888
- S2CID 88655961
- doi:10.1139/b94-219
- ^ PMID 21653356
- )
- )
- )
- ^ J. W. Wright (1955), "Species crossability in Spruce in relation to distribution and taxonomy", Forest Science, 1 (4): 319–349
- ^ )
- )
- ^ Lorna H. Arita & Kenneth Y. Kaneshiro (1979), "Ethological Isolation Between Two Stocks of Drosophila Adiastola Hardy", Proc. Hawaii. Entomol. Soc., 13: 31–34
- S2CID 43809774
- S2CID 34137489
- )
- S2CID 13379387
- )
- )
- )
- S2CID 30943286
- S2CID 34137489