Peterson olefination
| Peterson olefination | |
|---|---|
| Named after | Donald John Peterson |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | peterson-olefination |
| RSC ontology ID | RXNO:0000080 |
The Peterson olefination (also called the Peterson reaction) is the chemical reaction of α-silyl carbanions (1 in diagram below) with ketones (or aldehydes) to form a β-hydroxysilane (2) which eliminates to form alkenes (3).[1]
Several reviews have been published.[2][3][4][5][6]
Reaction mechanism
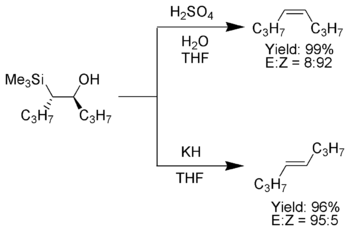
One attractive feature of the Peterson olefination is that it can be used to prepare either cis- or trans-alkenes from the same β-hydroxysilane. Treatment of the β-hydroxysilane with acid will yield one alkene, while treatment of the same β-hydroxysilane with base will yield the alkene of opposite stereochemistry.
Basic elimination
The action of base upon a β-hydroxysilane (1) results in a concerted syn elimination of (2) or (3) to form the desired alkene. The penta-coordinate silicate intermediate (3) is postulated, but no proof exists to date.[when?]
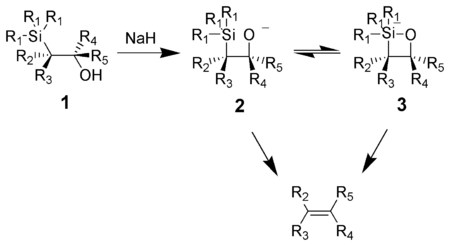
Potassium alkoxides eliminate quickly, while sodium alkoxides generally require heating. Magnesium alkoxides only eliminate in extreme conditions. The order of reactivity of alkoxides, K > Na >> Mg, is consistent with higher electron density on oxygen, hence increasing the alkoxide nucleophilicity.
Acidic elimination
The treatment of the β-hydroxysilane (1) with acid results in protonation and an anti elimination to form the desired alkene.

Alkyl substituents
When the α-silyl carbanion contains only
Once isolated, the diastereomeric β-hydroxysilanes are separated. One diastereomer is treated with acid, while the other is treated with base, thus converted the material to an alkene with the required stereochemistry.[4]
Electron-withdrawing substituents
When the α-silyl carbanion contains electron-withdrawing substituents, the Peterson olefination directly forms the alkene. The intermediate β-hydroxysilane cannot be isolated as it eliminates in-situ. The basic elimination pathway has been postulated in these cases.
Functional group tolerance
Unlike the Wittig reaction, Peterson-type olefinations tolerate nitriles.[8]
Variations
Acidic elimination conditions are sometimes not feasible as the acid also promotes double bond isomerization. Additionally, elimination using sodium or potassium hydride may not be feasible due to incompatible functional groups. Chan et al. have found that acylation of the intermediate silylcarbinol with either acetyl chloride or thionyl chloride gives a β-silyl ester that will eliminate spontaneously at 25 °C giving the desired alkene.[9] Corey and co-workers developed a method (sometimes dubbed the Corey-Peterson olefination[10]) using a silylated imine to yield an α,β-unsaturated aldehyde from a carbonyl compound in one step.[11] For an example for its use in total synthesis see: Kuwajima Taxol total synthesis
See also
- Horner–Wadsworth–Emmons reaction
- Tebbe olefination
- Wittig reaction
References
- .
- ^ Birkofer, L.; Stiehl, O. Top. Curr. Chem. 1980, 88, 58. (Review)
- ^ Ager, D. J. Synthesis 1984, 384–398. (Review)
- ^
- .
- ^
New developments in the Peterson olefination reaction L. Frances van Staden, David Gravestock and David J. Ager Chem. Soc. Rev., 2002,31, 195-200 doi:10.1039/A908402I
- ^ Barrett, A. G. M.; Flygare, J. A.; Hill, J. M.; Wallace, E. M. (1998). "Stereoselective Alkene Synthesis via 1-Chloro-1-[(dimethyl)phenylsilyl]alkanes and α-(Dimethyl)phenylsilyl Ketones: 6-Methyl-6-dodecene". Organic Syntheses; Collected Volumes, vol. 9, p. 580.
- PMID 27187788.
- PMID 4473100.
- PMID 15355023.
- .

