Pharmaceutical industry

The pharmaceutical industry is an industry in
History
Mid-1800s – 1945: From botanicals to the first synthetic drugs
The modern era of pharmaceutical industry began with local apothecaries that expanded from their traditional role of distributing botanical drugs such as
Epinephrine, norepinephrine, and amphetamine
By the 1890s, the profound effect of
By 1929 epinephrine had been formulated into an inhaler for use in the treatment of nasal congestion.While highly effective, the requirement for injection limited the use of epinephrine[
Discovery and development of the barbiturates

In 1903,
In 1958, Leo Sternbach discovered the first benzodiazepine, chlordiazepoxide (Librium). Dozens of other benzodiazepines have been developed and are in use, some of the more popular drugs being diazepam (Valium), alprazolam (Xanax), clonazepam (Klonopin), and lorazepam (Ativan). Due to their far superior safety and therapeutic properties, benzodiazepines have largely replaced the use of barbiturates in medicine, except in certain special cases. When it was later discovered that benzodiazepines, like barbiturates, significantly lose their effectiveness and can have serious side effects when taken long-term, Heather Ashton researched benzodiazepine dependence and developed a protocol to discontinue their use.
Insulin
A series of experiments performed from the late 1800s to the early 1900s revealed that diabetes is caused by the absence of a substance normally produced by the pancreas. In 1869, Oskar Minkowski and Joseph von Mering found that diabetes could be induced in dogs by surgical removal of the pancreas. In 1921, Canadian professor Frederick Banting and his student Charles Best repeated this study and found that injections of pancreatic extract reversed the symptoms produced by pancreas removal. Soon, the extract was demonstrated to work in people, but development of insulin therapy as a routine medical procedure was delayed by difficulties in producing the material in sufficient quantity and with reproducible purity. The researchers sought assistance from industrial collaborators at Eli Lilly and Co. based on the company's experience with large scale purification of biological materials. Chemist George B. Walden of Eli Lilly and Company found that careful adjustment of the pH of the extract allowed a relatively pure grade of insulin to be produced. Under pressure from Toronto University and a potential patent challenge by academic scientists who had independently developed a similar purification method, an agreement was reached for non-exclusive production of insulin by multiple companies. Prior to the discovery and widespread availability of insulin therapy the life expectancy of diabetics was only a few months.[13]
Early anti-infective research: Salvarsan, Prontosil, Penicillin and vaccines
The development of drugs for the treatment of infectious diseases was a major focus of early research and development efforts; in 1900, pneumonia, tuberculosis, and diarrhea were the three leading causes of death in the United States and mortality in the first year of life exceeded 10%.[14][15][failed verification]
In 1911 arsphenamine, the first synthetic anti-infective drug, was developed by Paul Ehrlich and chemist Alfred Bertheim of the Institute of Experimental Therapy in Berlin. The drug was given the commercial name Salvarsan.[16] Ehrlich, noting both the general toxicity of arsenic and the selective absorption of certain dyes by bacteria, hypothesized that an arsenic-containing dye with similar selective absorption properties could be used to treat bacterial infections. Arsphenamine was prepared as part of a campaign to synthesize a series of such compounds, and was found to exhibit partially selective toxicity. Arsphenamine proved to be the first effective treatment for syphilis, a disease untl then had been incurable and led inexorably to severe skin ulceration, neurological damage, and death.[17]
Ehrlich's approach of systematically varying the chemical structure of synthetic compounds and measuring the effects of these changes on biological activity was pursued broadly by industrial scientists, including
In 1928, Alexander Fleming discovered the antibacterial effects of penicillin, but its exploitation for the treatment of human disease awaited the development of methods for its large scale production and purification. These were developed by a U.S. and British government-led consortium of pharmaceutical companies during the world war.[23]
There was early progress toward the development of vaccines throughout this period, primarily in the form of academic and government-funded basic research directed toward the identification of the pathogens responsible for common communicable diseases. In 1885,
Unsafe drugs and early industry regulation

Prior to the 20th century, drugs were generally produced by small scale manufacturers with little regulatory control over manufacturing or claims of safety and efficacy. To the extent that such laws did exist, enforcement was lax. In the United States, increased regulation of vaccines and other biological drugs was spurred by tetanus outbreaks and deaths caused by the distribution of contaminated smallpox vaccine and diphtheria antitoxin.
In 1937 over 100 people died after ingesting "
The post-war years, 1945–1970
Further advances in anti-infective research
The aftermath of
A Federal Trade Commission report issued in 1958 attempted to quantify the effect of antibiotic development on American public health. The report found that over the period 1946–1955, there was a 42% drop in the incidence of diseases for which antibiotics were effective and only a 20% drop in those for which antibiotics were not effective. The report concluded that "it appears that the use of antibiotics, early diagnosis, and other factors have limited the epidemic spread and thus the number of these diseases which have occurred". The study further examined mortality rates for eight common diseases for which antibiotics offered effective therapy (syphilis, tuberculosis, dysentery, scarlet fever, whooping cough, meningococcal infections, and pneumonia), and found a 56% decline over the same period.[37] Notable among these was a 75% decline in deaths due to tuberculosis.[38]
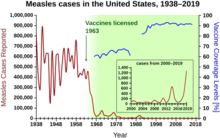
During the years 1940–1955, the rate of decline in the U.S.
Other notable new vaccines of the period include those for measles (1962,
Development and marketing of antihypertensive drugs
Hypertension is a risk factor for atherosclerosis,
Early developments in the field of treating hypertension included quaternary ammonium ion sympathetic nervous system blocking agents, but these compounds were never widely used due to their severe side effects, because the long-term health consequences of high blood pressure had not yet been established, and because they had to be administered by injection.
In 1952 researchers at Ciba discovered the first orally available vasodilator,
A 2009 Cochrane review concluded that thiazide antihypertensive drugs reduce the risk of death (
Oral Contraceptives
Prior to the Second World war, birth control was prohibited in many countries, and in the United States even the discussion of contraceptive methods sometimes led to prosecution under
Thalidomide and the Kefauver-Harris Amendments

In the U.S., a push for revisions of the FD&C Act emerged from Congressional hearings led by Senator Estes Kefauver of Tennessee in 1959. The hearings covered a wide range of policy issues, including advertising abuses, questionable efficacy of drugs, and the need for greater regulation of the industry. While momentum for new legislation temporarily flagged under extended debate, a new tragedy emerged that underscored the need for more comprehensive regulation and provided the driving force for the passage of new laws.
On 12 September 1960, an American licensee, the William S. Merrell Company of Cincinnati, submitted a new drug application for Kevadon (
The
1970–1990s
Statins
In 1971, Akira Endo, a Japanese biochemist working for the pharmaceutical company Sankyo, identified mevastatin (ML-236B), a molecule produced by the fungus Penicillium citrinum, as an inhibitor of HMG-CoA reductase, a critical enzyme used by the body to produce cholesterol. Animal trials showed very good inhibitory effect as in clinical trials, however a long-term study in dogs found toxic effects at higher doses and as a result mevastatin was believed to be too toxic for human use. Mevastatin was never marketed, because of its adverse effects of tumors, muscle deterioration, and sometimes death in laboratory dogs.
In April 1994, the results of a Merck-sponsored study, the
21st Century
Since several decades, biologics have been rising in importance in comparison with small molecules treatments. The biotech subsector, animal health and the Chinese pharmaceutical sector have also grown substantially. On the organisational side, big international pharma corporations have experienced a substantial decline of their value share. Also, the core generic sector (substitutions for off-patent brands) has been downvalued due to competition.[78]
Torreya estimated the pharmaceutical industry to have a market valuation of US$7.03 trillion by February 2021 from which US$6.1 trillion is the value of the publicly traded companies. Small Molecules modality had 58.2% of the valuation share down from 84.6% in 2003. Biologics was up at 30.5% from 14.5%. The valuation share of Chinese Pharma grew from 2003 to 2021 from 1% to 12% overtaking Switzerland who is now ranked number 3 with 7.7%. The United States had still by far the most valued pharmaceutical industry with 40% of global valuation.[79] 2023 was a year of layoffs for at least 10,000 people across 129 public biotech firms globally, albeit most small firms; this was a significant increase in reductions versus 2022 was in part due to worsening global financial conditions and a reduction in investment by "generalist investors".[80] Private firms also saw a significant reduction in venture capital investment in 2023, continuing a downward trend started in 2021, which also led to a reduction in initial public offerings being floated.[80]
Impact of Mergers and Acquisitions
A 2022 article articulated this notion succinctly by saying "In the business of drug development, deals can be just as important as scientific breakthroughs", typically referred to as pharmaceutical M&A (for mergers and acquisitions).
Research and development
Drug discovery is the process by which potential
Drug development refers to activities undertaken after a compound is identified as a potential drug in order to establish its suitability as a medication. Objectives of drug development are to determine appropriate
Often, large multinational corporations exhibit vertical integration, participating in a broad range of drug discovery and development, manufacturing and quality control, marketing, sales, and distribution. Smaller organizations, on the other hand, often focus on a specific aspect such as discovering drug candidates or developing formulations. Often, collaborative agreements between research organizations and large pharmaceutical companies are formed to explore the potential of new drug substances. More recently, multi-nationals are increasingly relying on contract research organizations to manage drug development.[84]
The cost of innovation
Industry-wide research and investment reached a record $65.3 billion in 2009.[88] While the cost of research in the U.S. was about $34.2 billion between 1995 and 2010, revenues rose faster (revenues rose by $200.4 billion in that time).[87]
A study by the consulting firm Bain & Company reported that the cost for discovering, developing and launching (which factored in marketing and other business expenses) a new drug (along with the prospective drugs that fail) rose over a five-year period to nearly $1.7 billion in 2003.[89] According to Forbes, by 2010 development costs were between $4 billion to $11 billion per drug.[90]
Some of these estimates also take into account the
Product approval
In the United States, new pharmaceutical products must be approved by the
A fourth phase of post-approval surveillance is also often required due to the fact that even the largest clinical trials cannot effectively predict the prevalence of rare side-effects. Postmarketing surveillance ensures that after marketing the safety of a drug is monitored closely. In certain instances, its indication may need to be limited to particular patient groups, and in others the substance is withdrawn from the market completely.
The FDA provides information about approved drugs at the Orange Book site.[93]
In the UK, the Medicines and Healthcare products Regulatory Agency approves and evaluates drugs for use. Normally an approval in the UK and other European countries comes later than one in the USA. Then it is the National Institute for Health and Care Excellence (NICE), for England and Wales, who decides if and how the National Health Service (NHS) will allow (in the sense of paying for) their use. The British National Formulary is the core guide for pharmacists and clinicians.
In many non-US western countries, a 'fourth hurdle' of
Orphan drugs
There are
Global sales
| Company | Pharma revenue ($ million) |
|---|---|
| Pfizer |
100,330 |
| Johnson & Johnson |
94,940 |
| 66,260 | |
| 59,280 | |
| 58,050 | |
| Novartis |
50,540 |
| Bristol Myers Squibb |
46,160 |
| Sanofi |
45,220 |
| AstraZeneca |
44,350 |
| GSK |
36,150 |
| Takeda |
30,000 |
| Eli Lilly and Company |
28,550 |
| Gilead Sciences |
27,280 |
| Bayer |
26,640 |
| Amgen |
26,320 |
| Boehringer Ingelheim |
25,280 |
| Novo Nordisk |
25,000 |
| Moderna |
19,260 |
| 19,160 | |
| BioNTech |
18,200 |
In 2011, global spending on prescription drugs topped $954 billion, even as growth slowed somewhat in Europe and North America. The United States accounts for more than a third of the global pharmaceutical market, with $340 billion in annual sales followed by the EU and Japan.[96] Emerging markets such as China, Russia, South Korea and Mexico outpaced that market, growing a huge 81 percent.[97][98]
The top ten best-selling drugs of 2013 totaled $75.6 billion in sales, with the anti-inflammatory drug
IMS Health publishes an analysis of trends expected in the pharmaceutical industry in 2007, including increasing profits in most sectors despite loss of some patents, and new 'blockbuster' drugs on the horizon.[101]
Patents and generics
Depending on a number of considerations, a company may apply for and be granted a patent for the drug, or the process of producing the drug, granting exclusivity rights typically for about 20 years.[102] However, only after rigorous study and testing, which takes 10 to 15 years on average, will governmental authorities grant permission for the company to market and sell the drug.[103] Patent protection enables the owner of the patent to recover the costs of research and development through high profit margins for the branded drug. When the patent protection for the drug expires, a generic drug is usually developed and sold by a competing company. The development and approval of generics is less expensive, allowing them to be sold at a lower price. Often the owner of the branded drug will introduce a generic version before the patent expires in order to get a head start in the generic market.[104] Restructuring has therefore become routine, driven by the patent expiration of products launched during the industry's "golden era" in the 1990s and companies' failure to develop sufficient new blockbuster products to replace lost revenues.[105]
Prescriptions
In the U.S., the value of prescriptions increased over the period of 1995 to 2005 by 3.4 billion annually, a 61 percent increase. Retail sales of prescription drugs jumped 250 percent from $72 billion to $250 billion, while the average price of prescriptions more than doubled from $30 to $68.[106]
Marketing
Advertising is common in healthcare journals as well as through more mainstream media routes. In some countries, notably the US, they are allowed to advertise directly to the general public. Pharmaceutical companies generally employ salespeople (often called 'drug reps' or, an older term, 'detail men') to market directly and personally to physicians and other healthcare providers. In some countries, notably the US,
To healthcare professionals
The book Bad Pharma also discusses the influence of drug representatives, how ghostwriters are employed by the drug companies to write papers for academics to publish, how independent the academic journals really are, how the drug companies finance doctors' continuing education, and how patients' groups are often funded by industry.[107]
Direct to consumer advertising
Since the 1980s, new methods of marketing for prescription drugs to consumers have become important. Direct-to-consumer media advertising was legalised in the FDA Guidance for Industry on Consumer-Directed Broadcast Advertisements.
Controversies
Drug marketing and lobbying
There has been increasing controversy surrounding pharmaceutical marketing and influence. There have been accusations and findings of influence on doctors and other health professionals through drug reps including the constant provision of marketing 'gifts' and biased information to health professionals;[108] highly prevalent advertising in journals and conferences; funding independent healthcare organizations and health promotion campaigns; lobbying physicians and politicians (more than any other industry in the US[109]); sponsorship of medical schools or nurse training; sponsorship of continuing educational events, with influence on the curriculum;[110] and hiring physicians as paid consultants on medical advisory boards.
Some advocacy groups, such as No Free Lunch and AllTrials, have criticized the effect of drug marketing to physicians because they say it biases physicians to prescribe the marketed drugs even when others might be cheaper or better for the patient.[111]
There have been related accusations of
Meta-analyses have shown that psychiatric studies sponsored by pharmaceutical companies are several times more likely to report positive results, and if a drug company employee is involved the effect is even larger.[115][116][117] Influence has also extended to the training of doctors and nurses in medical schools, which is being fought.
It has been argued that the design of the Diagnostic and Statistical Manual of Mental Disorders and the expansion of the criteria represents an increasing medicalization of human nature, or "disease mongering", driven by drug company influence on psychiatry.[118] The potential for direct conflict of interest has been raised, partly because roughly half the authors who selected and defined the DSM-IV psychiatric disorders had or previously had financial relationships with the pharmaceutical industry.[119]
In the US, starting in 2013, under the Physician Financial Transparency Reports (part of the Sunshine Act), the Centers for Medicare & Medicaid Services has to collect information from applicable manufacturers and group purchasing organizations in order to report information about their financial relationships with physicians and hospitals. Data are made public in the Centers for Medicare & Medicaid Services website. The expectation is that relationship between doctors and Pharmaceutical industry will become fully transparent.[120]
In a report conducted by OpenSecrets, there were more than 1,100 lobbyists working in some capacity for the pharmaceutical business in 2017. In the first quarter of 2017, the health products and pharmaceutical industry spent $78 million on lobbying members of the United States Congress.[121]
Medication pricing
The pricing of pharmaceuticals is becoming a major challenge for health systems.
Regulatory issues
Ben Goldacre has argued that regulators – such as the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK, or the Food and Drug Administration (FDA) in the United States – advance the interests of the drug companies rather than the interests of the public due to revolving door exchange of employees between the regulator and the companies and friendships develop between regulator and company employees.[124] He argues that regulators do not require that new drugs offer an improvement over what is already available, or even that they be particularly effective.[124]
Others have argued that excessive regulation suppresses therapeutic innovation and that the current cost of regulator-required clinical trials prevents the full exploitation of new genetic and biological knowledge for the treatment of human disease. A 2012 report by the President's Council of Advisors on Science and Technology made several key recommendations to reduce regulatory burdens to new drug development, including 1) expanding the FDA's use of accelerated approval processes, 2) creating an expedited approval pathway for drugs intended for use in narrowly defined populations, and 3) undertaking pilot projects designed to evaluate the feasibility of a new, adaptive drug approval process.[125]
Pharmaceutical fraud
The examples and perspective in this section deal primarily with the United States and do not represent a worldwide view of the subject. (August 2015) |
Every major company selling atypical antipsychotics—
On 2 July 2012,
The following is a list of the four largest settlements reached with pharmaceutical companies from 1991 to 2012, rank ordered by the size of the total settlement. Legal claims against the pharmaceutical industry have varied widely over the past two decades, including Medicare and Medicaid fraud, off-label promotion, and inadequate manufacturing practices.[135][136]
| Company | Settlement | Violation(s) | Year | Product(s) | Laws allegedly violated (if applicable) |
|---|---|---|---|---|---|
GlaxoSmithKline[137] |
$3 billion | Off-label promotion/ failure to disclose safety data |
2012 | Avandia/Wellbutrin/Paxil | False Claims Act/FDCA
|
| Pfizer[138] | $2.3 billion | Off-label promotion/kickbacks | 2009 | Bextra/Geodon/ Zyvox/Lyrica |
False Claims Act/FDCA |
| Abbott Laboratories[139] | $1.5 billion | Off-label promotion | 2012 | Depakote |
False Claims Act/FDCA |
| Eli Lilly[140] | $1.4 billion | Off-label promotion | 2009 | Zyprexa | False Claims Act/FDCA |
Physician roles
In May 2015, the
Response to COVID-19
In November 2020 several pharmaceutical companies announced successful trials of COVID-19 vaccines, with efficacy of 90 to 95% in preventing infection. Per company announcements and data reviewed by external analysts, these vaccines are priced at $3 to $37 per dose.[143] The Wall Street Journal ran an editorial calling for this achievement to be recognized with a Nobel Peace Prize.[144]
On 20 February, 46 US lawmakers called for the US government not to grant monopoly rights when giving out taxpayer development money for any coronavirus vaccines and treatments, to avoid giving exclusive control of prices and availability to private manufacturers.[146]
In the United States the government signed agreements in which research and development and/or the building of manufacturing plants for potential COVID-19 therapeutics was subsidized. Typically, the agreement involved the government taking ownership of a certain number of doses of the product without further payment. For example, under the auspices of Operation Warp Speed in the United States, the government subsidized research related to COVID-19 vaccines and therapeutics at Regeneron,[147] Johnson and Johnson, Moderna, AstraZeneca, Novavax, Pfizer, and GSK. Typical terms involved research subsidies of $400 million to $2 billion, and included government ownership of the first 100 million doses of any COVID-19 vaccine successfully developed.[148]
American pharmaceutical company Gilead sought and obtained orphan drug status for remdesivir from the US Food and Drug Administration (FDA) on 23 March 2020. This provision is intended to encourage the development of drugs affecting fewer than 200,000 Americans by granting strengthened and extended legal monopoly rights to the manufacturer, along with waivers on taxes and government fees.[149][150] Remdesivir is a candidate for treating COVID-19; at the time the status was granted, fewer than 200,000 Americans had COVID-19, but numbers were climbing rapidly as the COVID-19 pandemic reached the US, and crossing the threshold soon was considered inevitable.[149][150] Remdesivir was developed by Gilead with over $79 million in U.S. government funding.[150] In May 2020, Gilead announced that it would provide the first 940,000 doses of remdesivir to the federal government free of charge.[151] After facing strong public reactions, Gilead gave up the "orphan drug" status for remdesivir on 25 March.[152] Gilead retains 20-year remdesivir patents in more than 70 countries.[145] In May 2020, the company further announced that it was in discussions with several generics companies to provide rights to produce remdesivir for developing countries, and with the Medicines Patent Pool to provide broader generic access.[153]
Developing world
Patents
Patents have been criticized in the developing world, as they are thought[
In March 2001, 40 multi-national pharmaceutical companies brought litigation against South Africa for its Medicines Act, which allowed the generic production of antiretroviral drugs (ARVs) for treating HIV, despite the fact that these drugs were on-patent.[156] HIV was and is an epidemic in South Africa, and ARVs at the time cost between US$10,000 and US$15,000 per patient per year. This was unaffordable for most South African citizens, and so the South African government committed to providing ARVs at prices closer to what people could afford. To do so, they would need to ignore the patents on drugs and produce generics within the country (using a compulsory license), or import them from abroad. After international protest in favour of public health rights (including the collection of 250,000 signatures by Médecins Sans Frontières), the governments of several developed countries (including The Netherlands, Germany, France, and later the US) backed the South African government, and the case was dropped in April of that year.[157]
In 2016, GlaxoSmithKline (the world's sixth largest pharmaceutical company) announced that it would be dropping its patents in poor countries so as to allow independent companies to make and sell versions of its drugs in those areas, thereby widening the public access to them.[158] GlaxoSmithKline published a list of 50 countries they would no longer hold patents in, affecting one billion people worldwide.
Charitable programs
In 2011 four of the top 20 corporate charitable donations and eight of the top 30 corporate charitable donations came from pharmaceutical manufacturers. The bulk of corporate charitable donations (69% as of 2012) comes by way of non-cash charitable donations, the majority of which again were donations contributed by pharmaceutical companies.[159]
Charitable programs and drug discovery & development efforts by pharmaceutical companies include:
- "river blindness drugs were donated in Africa[160]
- AIDS in South Africa[161]
- GSK's commitment to give free albendazole tablets to the WHO for, and until, the elimination of lymphatic filariasisworldwide.
- In 2006, Glivec patient assistance programs; and relief to support major humanitarian organisations with emergency medical needs.[162]
See also
- List of industrial complexes– Economic concept
- Big Pharma conspiracy theory– Conspiracy theories about the pharmaceutical industry
- Clinical trial – Phase of clinical research in medicine
- Drug development – Process of bringing a new pharmaceutical drug to the market
- Drug discovery – Pharmaceutical procedure
- Legal drug trade – manufacture and sale of pharmaceutical drugs
- List of pharmaceutical companies
- Licensed production – Production under license of technology developed elsewhere
- Outsourcing – Contracting formerly internal tasks to an external organization
- Pharmaceutical marketing – Advertising by pharmaceutical companies
- Pharmacy – Clinical health science
- Pharmacy benefit management – Administration of prescription drug programs in the United States
- Unitaid – Global health initiative
- Valuation (finance) § Valuation of intangible assets
References
- ISBN 978-3527306732.
- ^ Bozenhardt, Erich H.; Bozenhardt, Herman F. (18 October 2018). "Are You Asking Too Much From Your Filler?". Pharmaceutical Online (Guest column). VertMarkets. Archived from the original on 17 November 2020. Retrieved 30 October 2018.
The core mission of the pharmaceutical industry is to manufacture products for patients to cure them, vaccinate them, or alleviate a symptom, often by manufacturing a liquid injectable or an oral solid, among other therapies.
- ^ Markets, Research and (31 March 2021). "Global Pharmaceuticals Market Report 2021: Market is Expected to Grow from $1228.45 Billion in 2020 to $1250.24 Billion in 2021 - Long-term Forecast to 2025 & 2030". GlobeNewswire News Room (Press release). Archived from the original on 29 November 2021. Retrieved 29 November 2021.
- ) began as local apothecary shops in the mid-1800s.
- ^ "Emergence of Pharmaceutical Science and Industry: 1870-1930". Chem Eng News. 83 (25). 20 June 2005. Archived from the original on 10 November 2018. Retrieved 23 July 2022.
- ISBN 978-0-470-01552-0. Retrieved 23 July 2022.
- ^ S2CID 24974454.
- PMID 18445805.
- S2CID 8934654.
- PMID 18568113.
- .
Officers of the Food and Drug Administration, aware of the seriousness of the problem, estimate that approximately half the 9,000,000,000 barbiturate and amphetamine capsules and tablets manufactured annually in this country are diverted to illegal use. The profits to be gained from the illegal sale of these drugs have proved an attraction to organized crime, for amphetamine can be purchased at wholesale for less than $1 per 1000 capsules, but when sold on the illegal market, it brings $30 to $50 per 1000 and when retailed to the individual buyer, a tablet may bring as much as 10 to 25 cents.
- PMID 13378632.
The barbiturates, introduced into medicine by E. Fischer and J. von Mering in 1903, are certainly among the most widely used and abused drugs in medicine. Approximately 400 tons of these agents are manufactured each year; this is enough to put approximately 9,000,000 people to sleep each night for that period if each were given a 0.1-gm. dose
- PMID 12446492.
- ^ "Leading Causes of Death, 1900-1998" (PDF). CDC.gov. Archived (PDF) from the original on 13 December 2020. Retrieved 23 July 2022.
- PMID 10635683. Archived from the original(PDF) on 25 October 2020. Retrieved 23 July 2022.
- PMID 21793743.
- PMID 19679737.
- PMID 21687759.
- ISBN 978-1-4000-8213-1.
- ^ "All Nobel Prizes in Physiology or Medicine". The Nobel Prize. Retrieved 22 July 2022.
- doi:10.3386/w8556. Retrieved 23 July 2022.
- ^ a b Klein, Herbert (2012). A Population History of the United States. Cambridge University Press. p. 167.
- ISBN 978-0-931292-08-8.
- ^ "Diphtheria — Timelines — History of Vaccines". Archived from the original on 14 May 2016. Retrieved 24 February 2022.
- ^ Ii, Thomas H. Maugh (13 April 2005). "Maurice R. Hilleman, 85; Scientist Developed Many Vaccines That Saved Millions of Lives - Los Angeles Times". Los Angeles Times. Archived from the original on 7 November 2014.
- ^ "Significant Dates in U.S. Food and Drug Law History". Food and Drug Administration. Archived from the original on 6 March 2013. Retrieved 16 December 2019.
- ^ "FDAReview.org, a project of The Independent Institute". Archived from the original on 2 December 2015. Retrieved 24 February 2022.
- ^ "Sulfanilamide Disaster". Food and Drug Administration. Archived from the original on 25 November 2020. Retrieved 16 December 2019.
- ^ "FDA History - Part II". Food and Drug Administration. Archived from the original on 1 January 2018. Retrieved 16 December 2019.
- S2CID 30538825.
- PMID 18248798.
- S2CID 12014890.
- ^ S2CID 27465970.
- S2CID 34647314.
- PMID 12987755.
- ISBN 9780470972458.
- ^ Federal Trade Commission Report of Antibiotics Manufacture, June 1958 (Washington D.C., Government Printing Office, 1958) pages 98-120
- ^ Federal Trade Commission Report of Antibiotics Manufacture, June 1958 (Washington D.C., Government Printing Office, 1958) page 277
- (PDF) from the original on 25 October 2020. Retrieved 8 September 2017.
- doi:10.3386/w8556. Archived(PDF) from the original on 30 July 2020. Retrieved 24 February 2022.
- S2CID 38744505.
- PMID 174424.
- ^ "Studies:No Evidence That SV40 is Related to Cancer - National Cancer Institute". Archived from the original on 28 October 2014.
- ^ "History of Vaccines — A Vaccine History Project of The College of Physicians of Philadelphia". Archived from the original on 19 February 2022. Retrieved 24 February 2022.
- ^ "Prevention of Measles, Rubella, Congenital Rubella Syndrome, and Mumps, 2013". Archived from the original on 10 November 2014. Retrieved 8 September 2017.
- S2CID 6512947.
- PMID 19110086.
- PMID 19427498. Retrieved 20 June 2009.
- PMID 19363848.
- PMID 19427499. Retrieved 20 June 2009.
- PMID 19393824.
- PMID 19195963. Retrieved 20 June 2009.
- PMID 11842376.
- PMID 18672184. Retrieved 20 June 2009.
- PMID 19008688.
- PMID 19149532. Archived from the originalon 12 January 2013. Retrieved 20 June 2009.
- PMID 20782854.
- ^ Sneader, Walter (2005). Drug Discovery. A History. New York: Wiley. p. 371.
- PMID 8349332.
- PMID 14119789.
- ^ "The Lasker Foundation - Awards". Archived from the original on 23 December 2015. Retrieved 24 February 2022.
- PMID 29667175.
- S2CID 886870.
- PMID 14132613.
- PMID 23235603.
- ^ "A brief history of the birth control pill - The pill timeline | Need to Know | PBS". PBS. 7 May 2010. Archived from the original on 19 January 2018. Retrieved 8 September 2017.
- ^ Magazine, Smithsonian. "Why the Oral Contraceptive Is Just Known as "The Pill"". Smithsonian Magazine. smithsonianmag.com. Archived from the original on 28 April 2021. Retrieved 24 February 2022.
- ^ "BBC News | HEALTH | A short history of the pill". Archived from the original on 6 July 2021. Retrieved 24 February 2022.
- ^ "FDA's Approval of the First Oral Contraceptive, Enovid". Food and Drug Administration. Archived from the original on 23 July 2017. Retrieved 16 December 2019.
- ^ Cafe, Rebecca (4 December 2011). "BBC News - How the contraceptive pill changed Britain". BBC News. Archived from the original on 16 June 2019. Retrieved 21 June 2018.
- ^ "Brochure: The History of Drug Regulation in the United States". Food and Drug Administration. Archived from the original on 23 July 2017. Retrieved 16 December 2019.
- S2CID 3344720.
- PMID 1464741.
- .
- S2CID 5965882.
- ^ "National Inventors Hall of Fame Honors 2012 Inductees". PRNewswire. Archived from the original on 12 May 2014. Retrieved 11 May 2014.
- ^ "How One Scientist Intrigued by Molds Found First Statin". Wall Street Journal. Archived from the original on 24 February 2022. Retrieved 11 May 2014.
- ^ "Top Global Pharmaceutical Company Report - The Pharma 1000" (PDF). Torreya. Retrieved 19 August 2022.
- ^ "Top Global Pharmaceutical Company Report" (PDF). The Pharma 1000. November 2021. Retrieved 29 December 2022.
- ^ BioPharma Dive. Retrieved 10 January 2024.
- ^ a b Bell, Jacob (20 December 2022). "Biotech M&A is picking back up. Here are the latest deal". BiopharmaDive. Archived from the original on 24 December 2022. Retrieved 23 December 2022.
- ^ "Annual Impact Report". Tufts Center for the Study of Drug Development. Archived from the original on 3 February 2022. Retrieved 24 February 2022.
- ^ "The Pharmaceutical Industry in Figures Key Data 2021" (PDF). European Federation of Pharmaceutical Industries and Associations. Retrieved 30 April 2022.
- ^ Outsourcing-Pharma.com (25 May 2011). "Pfizer teams with Parexel and Icon in CRO sector's latest strategic deals". Outsourcing-Pharma.com. Archived from the original on 11 October 2013. Retrieved 24 February 2022.
- ^ "How Many New Drugs Did FDA Approve Last Year?". pharmalot.com. Archived from the original on 8 May 2011. Retrieved 23 April 2011.
- ^ "Research". Archived from the original on 20 July 2011. Retrieved 24 November 2006.
- ^ a b Perry, Susan (8 August 2012). "Donald Light and Joel Lexchin in BMJ 2012;345:e4348, quoted in: Big Pharma's claim of an 'innovation crisis' is a myth, BMJ authors say". MinnPost. Archived from the original on 11 August 2012. Retrieved 8 August 2012.
- ^ "About PhRMA - PhRMA". Archived from the original on 4 January 2013. Retrieved 23 April 2011.
- ^ "Has the Pharmaceutical Blockbuster Model Gone Bust?". bain.com. Archived from the original on 24 March 2016. Retrieved 19 May 2016.
- ^ Harper, Matthew (10 February 2012). "The Truly Staggering Cost Of Inventing New Drugs". Forbes. Archived from the original on 9 December 2020. Retrieved 8 September 2017.
- ^ IMS Health (18 June 2015). "Are European biotechnology companies sufficiently protected?". Portal of Competitive Intelligence. Archived from the original on 30 June 2015. Retrieved 27 June 2015.
- S2CID 45729390. Archived from the originalon 6 February 2012. Retrieved 18 October 2011.
- ^ "Electronic Orange Book". U.S. Food and Drug Administration. Archived from the original on 7 April 2020. Retrieved 31 May 2007.
- ^ "The Orphan Drug Act (as amended)". U.S. Food and Drug Administration. Archived from the original on 7 April 2020. Retrieved 24 September 2007.
- ^ "The top 20 pharma companies by 2022 revenue".
- ^ "Standardseite der Domain www.vfa.de" (PDF). Archived from the original (PDF) on 11 August 2016. Retrieved 24 March 2008.
- ^ Herper, Matthew & Kang, Peter (22 March 2006). "The World's Ten Best-Selling Drugs". Forbes. Archived from the original on 5 May 2006. Retrieved 31 May 2007.
- ^ "Creating Connected Solutions for Better Healthcare Performance". IMS Health. Archived from the original (PDF) on 10 May 2020. Retrieved 23 February 2022.
- ^ Kollewe, Julia (27 March 2014). "World's 10 bestselling prescription drugs made $75bn last year". the Guardian. Archived from the original on 7 April 2020. Retrieved 12 December 2016.
- ^ "Top 100 Drugs for 2013 by Sales - U.S. Pharmaceutical Statistics". Archived from the original on 31 March 2014. Retrieved 23 January 2018.
- ^ "IMS Health Forecasts 5 to 6 Percent Growth for Global Pharmaceutical Market in 2007". IMS Health. 24 October 2006. Archived from the original on 3 May 2011. Retrieved 19 June 2007.
- ^ Frequently Asked Questions (FAQs) Archived 25 February 2013 at the Wayback Machine
- ^ "New Drug Approvals in 2006" (PDF). March 2007. Archived from the original (PDF) on 28 February 2008. Retrieved 23 February 2008.
- ^ "Assessment of Authorized Generics in the U.S" (PDF). IMS Consulting. June 2006. Archived from the original (PDF) on 28 February 2008. Retrieved 23 February 2008.
- ^ "Sanofi Laying Off 1,700 in US". Drug Discovery & Development. Archived from the original on 11 September 2011. Retrieved 24 February 2022.
- ^ "2007 Health and Nutrition - Census" (PDF). U.S. Census Bureau. Archived (PDF) from the original on 31 July 2020. Retrieved 19 May 2016.
- ISBN 9780865478060.
- Washington Post. Archivedfrom the original on 9 November 2020. Retrieved 23 May 2007.
- ^ "Drug Lobby Second to None: How the pharmaceutical industry gets its way in Washington". publicintegrity.org. 7 July 2005. Archived from the original on 9 June 2007. Retrieved 23 May 2007.
- PMID 12775595.
- ^ "Dr. No Free Lunch". Mother Jones. Archived from the original on 16 November 2020. Retrieved 19 May 2016.
- ISBN 978-1-74114-579-3.
- ^ "A Collection of Articles on Disease Mongering". Public Library of Science. Archived from the original on 7 June 2007. Retrieved 23 May 2007.
- ^ "Pharmaceutical Market Research, Trends And Analysis Reports". literated.com. Archived from the original on 19 January 2016. Retrieved 17 January 2016.
- S2CID 43544256.
- PMID 16199844.
- PMID 17602130.
- PMID 16597178.
- S2CID 11909535.
- ^ "Open Payments". February 2019. Archived from the original on 22 February 2022. Retrieved 24 February 2022.
- New York Times. Archivedfrom the original on 20 November 2020. Retrieved 30 May 2017.
- from the original on 27 November 2020. Retrieved 14 November 2020.
- ^ "High Drug Prices and Patient Costs: Millions of Lives and Billions of Dollars Lost". www.cidsa.org. West Health Council for Informed Drug Spending Analysis. 18 November 2020. Retrieved 20 February 2023.
- ^ ISBN 9780865478060.
- )
- ^ "Financial Crimes to the Public Report 2006". FBI. 2006. Archived from the original on 29 May 2016. Retrieved 28 July 2016.
- ^ "FBI-Health Care Fraud". FBI. Archived from the original on 2 July 2016. Retrieved 28 July 2016.
- ^ "Department of Justice". Department of Justice. 19 March 2015. Archived from the original on 9 November 2014. Retrieved 22 November 2020.
- ^ Wilson, Duff (2 October 2010). "Side Effects May Include Lawsuits". The New York Times. Archived from the original on 23 June 2017. Retrieved 25 February 2017.
- ^ "GlaxoSmithKline". BBC News. 4 July 2012. Archived from the original on 17 November 2020. Retrieved 21 June 2018.
- ^ "GlaxoSmithKline Agrees to Pay $3 Billion in U.S. Drug Settlement". Bloomberg. 2 July 2012. Archived from the original on 14 May 2014. Retrieved 11 March 2017.
- ^ Mogul, Fred (2 July 2012). "NY to Get Millions in GlaxoSmithKlein Settlement". WNYC. Archived from the original on 19 April 2013. Retrieved 2 July 2012.
- ^ "BBC News -GlaxoSmithKline to pay $3bn in US drug fraud scandal". BBC Online. 2 July 2012. Archived from the original on 17 November 2020. Retrieved 2 July 2012.
- ^ Thomas, Katie & Schmidt, Michael S. (2 July 2012). "Glaxo Agrees to Pay $3 Billion in Fraud Settlement". The New York Times. Archived from the original on 2 March 2017. Retrieved 3 July 2012.
- ^ "Rapidly Increasing Criminal and Civil Penalties Against the Pharmaceutical Industry: 1991-2010". citizen.org. Archived from the original on 16 May 2016. Retrieved 19 May 2016.
- ^ Thomas, Katie; Schmidt, Michael S. (2 July 2012). "GlaxoSmithKline Agrees to Pay $3 Billion in Fraud Settlement". The New York Times. Archived from the original on 12 November 2020. Retrieved 25 February 2017.
- ^ "GlaxoSmithKline to Plead Guilty and Pay $3 Billion to Resolve Fraud Allegations and Failure to Report Safety Data". 2 July 2012. Archived from the original on 9 September 2014. Retrieved 22 November 2020.
- ^ "JUSTICE DEPARTMENT ANNOUNCES LARGEST HEALTH CARE FRAUD" (PDF). US department of Justice. Archived (PDF) from the original on 2 December 2020. Retrieved 19 May 2016.
- ^ "Abbott Labs to Pay $1.5 Billion to Resolve Criminal & Civil Investigations of Off-label Promotion of Depakote". 7 May 2012. Archived from the original on 21 August 2014. Retrieved 22 November 2020.
- ^ "#09-038: Eli Lilly and Company Agrees to Pay $1.415 Billion to Resolve Allegationsof Off-label Promotion of Zyprexa (2009-01-15)". Archived from the original on 13 March 2016. Retrieved 22 November 2020.
- PMID 25946285.
- PMID 25946288.
- ^ "Covid vaccine front-runners: How much they cost, who's bought them and how they're stored". CNBC. 17 November 2020. Archived from the original on 1 December 2020. Retrieved 2 December 2020.
- ^ Henninger, Daniel (2 December 2020). "Opinion | Pharma Deserves the Nobel Peace Prize for the Covid Vaccines". Wall Street Journal. Archived from the original on 3 February 2021. Retrieved 4 December 2020.
- ^ a b "No profiteering on COVID-19 drugs and vaccines, says MSF". Médecins Sans Frontières (MSF) International. Archived from the original on 26 November 2021. Retrieved 22 April 2020.
- ^ Mazzucato, Mariana; Momenghalibaf, Azzi (18 March 2020). "Drug Companies Will Make a Killing From Coronavirus". The New York Times. Archived from the original on 23 November 2020. Retrieved 22 April 2020.
- ^ "HHS, DOD Collaborate with Regeneron on Large-Scale Manufacturing Demonstration Project of COVID-19 Investigational Therapeutic Treatment". 6 July 2020. Archived from the original on 18 December 2020. Retrieved 2 December 2020.
- ^ "COVID-19 Vaccines". 12 December 2020. Archived from the original on 19 December 2020. Retrieved 2 December 2020.
- ^ a b Lupkin, Sydney (24 March 2020). "FDA Grants Experimental Coronavirus Drug Benefits For Rare Disease Treatments". NPR. Archived from the original on 14 December 2020. Retrieved 24 March 2020.
- ^ a b c Conley, Julia (24 March 2020). "'This Is a Massive Scandal': Trump FDA Grants Drug Company Exclusive Claim on Promising Coronavirus Drug". Common Dreams. Archived from the original on 8 December 2020. Retrieved 22 April 2020.
- ^ Boodman, Eric (18 May 2020). "Gilead ups its donation of the Covid-19 drug remdesivir for U.S. hospitals". statnews.com. Archived from the original on 20 December 2020. Retrieved 19 July 2020.
- ^ Lawson, Alex (25 March 2020). "Don't Let Big Pharma Make a Killing by Profiteering Off COVID-19 Treatments". Common Dreams. Archived from the original on 9 November 2020. Retrieved 22 April 2020.
- ^ "Gilead Sciences Statement on Expanding Global Supply of Investigational Antiviral Remdesivir | Gilead". Archived from the original on 11 December 2020. Retrieved 16 July 2020.
- ^ See for example: 't Hoen, Ellen. "TRIPS, Pharmaceutical Patents, and Access to Essential Medicines: A Long Way from Seattle to Doha". Chicago Journal of International Law, 27(43), 2002; Musungu, Sisule F., and Cecilia Oh. "The Use of Flexibilities in TRIPS by Developing Countries: Can They Provide Access to Medicines?" Commission on Intellectual Property Rights, Innovation and Public Health, The World Health Organization, 2005.
- ^ "Declaration on the TRIPS agreement and public health". World Trade Organization. Archived from the original on 20 June 2016. Retrieved 19 May 2016.
- ^ Pharmaceutical Manufacturer's Association v. The President of South Africa (PMA), 2002 (2) SA 674 (CC) (S. Africa).
- ISBN 978-1-139-49691-9. Archivedfrom the original on 16 November 2020. Retrieved 28 July 2016.
- ^ "GlaxoSmithKline to 'drop patents in poor countries for better drug access'". BBC News. Archived from the original on 9 November 2020. Retrieved 19 May 2016.
- ^ "Bumper Year for Corporate Donations Reveals Profit Motives". corpwatch.org. Archived from the original on 11 November 2017. Retrieved 24 February 2022.
- ^ "Merk". Archived from the original on 26 August 2006. Retrieved 30 August 2006.
- ^ "Pfizer Will Donate Fluconazole to South Africa". Archived from the original on 24 October 2010. Retrieved 30 August 2006.
- ^ "Corporate Responsibility: Novartis". novartis.com. Archived from the original on 1 September 2017. Retrieved 19 May 2016.
External links
 Quotations related to Pharmaceutical industry at Wikiquote
Quotations related to Pharmaceutical industry at Wikiquote- "Global Medicines Use in 2020". IMS Institute for Healthcare Informatics. November 2015.
- "The pharmaceutical industry and global health – Facts & figures 2017" (PDF). International Federation of Pharmaceutical Manufacturers & Associations. February 2017.
- "Licensing Agreements in the Pharmaceutical Industry".
- "The Pharmaceutical Industry in Figures – Key Data 2018" (PDF). European Federation of Pharmaceutical Industries and Associations.
