Phenacemide
Source: Wikipedia, the free encyclopedia.
Anticonvulsant
 | |
| Clinical data | |
|---|---|
| Trade names | Phenurone |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| ATC code | |
| Pharmacokinetic data | |
| Elimination half-life | 22–25 hours |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
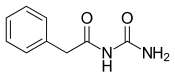
Phenacemide (
ureide (acetylurea) class.[1] It is a congener and ring-opened analogue of phenytoin (a hydantoin),[2][3] and is structurally related to the barbiturates and to other hydantoins.[4] Phenacemide was introduced in 1949 for the treatment of epilepsy, but was eventually withdrawn due to toxicity.[2][3]
See also
References
- ISBN 978-0-412-46630-4.
- ^ ISBN 978-81-7371-679-9.
- ^ )
- ISBN 978-81-85790-03-9.
Further reading
- Coker SB (1986). "The use of phenacemide for intractable partial complex epilepsy in children". Pediatric Neurology. 2 (4): 230–232. PMID 3508693.
- Coker SB, Holmes EW, Egel RT (December 1987). "Phenacemide therapy of complex partial epilepsy in children: determination of plasma drug concentrations". Neurology. 37 (12): 1861–1866. S2CID 219205208.
External links
| GABAergics | |||||||
|---|---|---|---|---|---|---|---|
| Channel modulators |
| ||||||
| Others |
| ||||||
| |||||||
VGKCsTooltip Voltage-gated potassium channels
| Blockers |
|
|---|---|
| Activators |
|
IRKsTooltip Inwardly rectifying potassium channel
| Blockers |
|
|---|---|
| Activators |
|
| Blockers |
|
|---|---|
| Activators |
K2PsTooltip Tandem pore domain potassium channel
| Blockers |
|
|---|---|
| Activators |
VGSCsTooltip Voltage-gated sodium channels
| Blockers |
|
|---|---|
| Activators |
|
| Blockers | |
|---|---|
| Activators |
| Blockers |
|---|
CaCCsTooltip Calcium-activated chloride channel
| Blockers |
|
|---|---|
| Activators |
| Blockers | |
|---|---|
| Activators |
| Blockers |
|---|
TRPs Tooltip Transient receptor potential channels |
|
|---|---|
LGICs Tooltip Ligand gated ion channels |
|
This anticonvulsant-related article is a stub. You can help Wikipedia by expanding it. |
