Phenibut
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Anvifen, Fenibut, Noofen, others[1] |
| Other names | Aminophenylbutyric acid; Fenibut; Fenigam; Phenigam; Phenybut; Phenygam; Phenylgamma; Phenigama; PHG; PhGABA; β-Phenyl-γ-aminobutyric acid; β-Phenyl-GABA[2] |
| Routes of administration | Common: By mouth[3] Uncommon: Rectal[3] |
| Drug class | GABA receptor agonist; Gabapentinoid |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Well-absorbed[6] ≥63% (250 mg)[7] |
| Metabolism | Liver (minimal)[6][7] |
| Metabolites | Inactive[6] |
| Onset of action | Oral: 2–4 hours[3] Rectal: 20–30 minutes[3] |
| Elimination half-life | 5.3 hours (250 mg)[7] |
| Duration of action | 15–24 hours (1–3 g)[3] |
| Excretion | Urine: 63% (unchanged)[7] |
| Identifiers | |
| |
JSmol) | |
| Melting point | 253 °C (487 °F) |
| |
| |
| (verify) | |
Phenibut, sold under the brand names Anvifen, Fenibut, and Noofen among others,
Side effects of phenibut can include
Phenibut was developed in the
Medical uses
Phenibut is used in Russia, Ukraine, Belarus and Latvia as a
Available forms
Phenibut is available as a medication in the form of 250 mg or 500 mg tablets for oral administration and as a solution at a concentration of 10 mg/mL for infusion.[6][8][11] In the US, dietary supplements labeled as containing phenibut have been found to contain zero to greater than 1,100 mg of phenibut per serving.[10]
Contraindications
Contraindications of phenibut include:[6][8]
- Intolerance to phenibut
- Pregnancy and breastfeeding
- Children who are younger than two years of age
- Liver insufficiency or failure
- Ulcerative lesions of the gastrointestinal tract
Phenibut should not be combined with alcohol.[8]
Side effects
Phenibut is generally well-
Overdose
In
Dependency and withdrawal
Interactions
Phenibut may mutually potentiate and extend the duration of the effects of other central nervous system depressants including anxiolytics, antipsychotics, sedatives, opioids, anticonvulsants, and alcohol.[6][8]
Pharmacology
Pharmacodynamics
| Compound | GABAB | GABAA |
|---|---|---|
| GABA | 0.08 | 0.12 |
GHB |
>100 | >100 |
GABOB |
1.10 | 1.38 |
| Phenibut | 9.6 | >100 |
| 4-F-phenibut | 1.70 | >100 |
| Baclofen | 0.13 | >100 |
| (R)-Baclofen | 0.13 | >100 |
| (S)-Baclofen | 74.0 | >100 |
| Values are IC50 (μM) in rat brain. | ||
Phenibut acts as a
| Compound | α2δ |
GABAB |
|---|---|---|
| Phenibut | ND | 177 |
| (R)-Phenibut | 23 | 92 |
| (S)-Phenibut | 39 | >1,000 |
| Baclofen | 156 | 6 |
| Gabapentin | 0.05 | >1,000 |
| Values are Ki (μM) in rat brain. | ||
Phenibut also binds to and blocks
Pharmacokinetics
Little information thus far has been published on the clinical
Some limited information has been described on the pharmacokinetics of phenibut in recreational users taking much higher doses (e.g., 1–3 grams) than typical clinical doses.
Chemistry
Phenibut is a
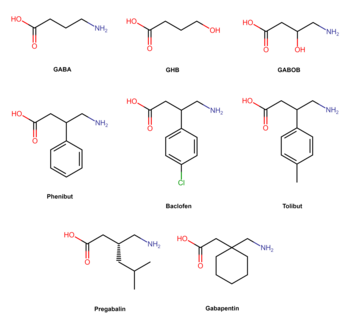
Structure and analogues
Phenibut is a
Phenibut is closely related to a variety of other GABA analogues including
A
Synthesis
A chemical synthesis of phenibut has been published.[11]
History
Phenibut was synthesized at the
Society and culture

Other names
Alternate spellings include fenibut and phenybut.
Brand names
Phenibut is marketed in Russia, Ukraine, Belarus and Latvia under the brand names Anvifen, Fenibut, Bifren and Noofen (Russian: Анвифен, Фенибут, Бифрен and Ноофен, respectively).[1]
Availability
Phenibut is approved in
Recreational use
Phenibut is used
Legal status
As of 2021, phenibut is a controlled substance in Australia,[3] France,[23] Hungary,[24] Italy,[25] and Lithuania.[26][27] In 2015, it was suggested that the legal status of phenibut in Europe should be reconsidered due to its recreational potential.[3] In February 2018, the Australian Therapeutic Goods Administration declared it a prohibited (schedule 9) substance, citing health concerns due to withdrawal and overdose.[28][29]
As of 14 November 2018, Hungary added phenibut and 10 other items to its New Psychoactive Substances ban list.[30]
As of 26 August 2020, Italy added phenibut to its New Psychoactive Substances ban list.[25]
As of 18 September 2020, France added phenibut to the controlled psychoactive substances list, prohibiting production, sale, storage and use.[31]
In the United States, phenibut is not a Controlled Substance. However, Dietary supplements that contain phenibut are unlawful to introduce into interstate commerce, because phenibut is considered a "New Drug" and any food, supplement, cosmetic, or drug that contains phenibut is therefore misbranded. Alabama placed phenibut and tianeptine into Schedule I status on 2021, first by action of the Alabama Department of Public Health and then followed by the state legislature.[5]
References
- ^ ISSN 1382-4368.
- ^ ISBN 978-1-4757-2085-3.
- ^ PMID 26693960.
- ^ Nutrition, Center for Food Safety and Applied (6 March 2023). "Phenibut in Dietary Supplements". FDA.
- ^ a b "HB2, Holmes, Tianeptine and Phenibut added to Schdule I Conrolled Substances". Alabama Pharmacy Association. Archived from the original on 1 June 2021.
- ^ a b c d e f g h i j k l m n o p q r s t Ozon Pharm, Fenibut (PDF), archived from the original (PDF) on 16 September 2017, retrieved 15 September 2017
- ^ PMID 11830761.
- ^ a b c d e f g h i j k l m Регистр лекарственных средств России ([Russian Medicines Register]). "Фенибут (Phenybutum)" [Fenibut (Phenybutum)] (in Russian). Retrieved 15 September 2017.
- PMID 11830761.
- ^ S2CID 237594860.
- ^ a b Sivchik VV, Grygoryan HO, Survilo VL, Trukhachova TV (2012), Синтез γ-амино-β-фенилмасляной кислоты (фенибута) [Synthesis of β-phenyl-γ-aminobutyric acid (phenibut)] (PDF) (in Russian)
- PMID 32881852.
- PMID 23391959.
- PMID 6297646.
- ^ ISBN 978-0-12-378648-7.
- ^ PMID 18275958.
- ISSN 0040-4020.
- ^ S2CID 42606053.
- PMID 26621244.
- PMID 25655145.
- S2CID 4907409.
- S2CID 29071385.
- ^ Par [1] La liste des substances psychotropes
- ^ "39/2018. (XI. 8.) EMMI rendelet Az új pszichoaktív anyaggá minősített anyagokról vagy vegyületcsoportokról szóló 55/2014. (XII. 30.) EMMI rendelet módosításáról" (PDF).
- ^ a b "Gazzetta Ufficiale 11/08/20". Lorenzo Arbolino. 11 August 2020. Retrieved 27 August 2020.
- ^ "RINKOS RIBOJIMO PRIEMONĖS FENIBUTUI!". ntakd.lrv.lt (in Lithuanian). Archived from the original on 6 October 2021. Retrieved 27 January 2020.
- ^ "V-1431 Dėl Lietuvos Respublikos sveikatos apsaugos ministro 2000 m. sausio 6 d. įsakymo Nr. 5 "Dėl Narko..." e-seimas.lrs.lt (in Lithuanian). Retrieved 27 January 2020.
- ^ "3.3 Phenibut". Administration Therapeutic Goods Administration. Australian Government Department of Health. 31 October 2017. Retrieved 6 November 2017.
- ^ "Mass school overdose investigation focuses on banned Russian drug". ABC News. Australian Broadcasting Corporation. 22 February 2018. Retrieved 22 February 2018.
- ^ "EMMI Decree substances or groups of compounds classified as new psychoactive substances". Wolters Kluwer. 1 January 2015. Archived from the original on 8 August 2020. Retrieved 5 August 2020.
- ^ Le phénibut interdit en France | Le Généraliste
