Phenol
| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Phenol[1] | |||
| Systematic IUPAC name
Benzenol | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.003.303 | ||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 2821 (solution) 2312 (molten) 1671 (solid) | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H6O | |||
| Molar mass | 94.113 g/mol | ||
| Appearance | Transparent crystalline solid | ||
| Odor | Sweet and tarry | ||
| Density | 1.07 g/cm3 | ||
| Melting point | 40.5 °C (104.9 °F; 313.6 K) | ||
| Boiling point | 181.7 °C (359.1 °F; 454.8 K) | ||
| 8.3 g/100 mL (20 °C) | |||
| log P | 1.48[2] | ||
| Vapor pressure | 0.4 mmHg (20 °C)[3] | ||
| Acidity (pKa) |
| ||
Conjugate base
|
Phenoxide | ||
| UV-vis (λmax) | 270.75 nm[5] | ||
| 1.224 D | |||
| Pharmacology | |||
| C05BB05 (WHO) D08AE03 (WHO), N01BX03 (WHO), R02AA19 (WHO) | |||
| Hazards | |||
| GHS labelling: | |||
   [6] [6]
| |||
| Danger | |||
| H301, H311, H314, H331, H341, H373[6] | |||
| P261, P280, P301+P310, P305+P351+P338, P310[6] | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 79 °C (174 °F; 352 K) | ||
Explosive limits
|
1.8–8.6%[3] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
| ||
LDLo (lowest published)
|
| ||
LC50 (median concentration)
|
| ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 5 ppm (19 mg/m3) [skin][3] | ||
REL (Recommended)
|
| ||
IDLH (Immediate danger) |
250 ppm[3] | ||
| Safety data sheet (SDS) | [1] | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an
Phenol was first extracted from
Properties
Phenol is an organic compound appreciably soluble in water, with about 84.2 g dissolving in 1000 ml (0.895 M). Homogeneous mixtures of phenol and water at phenol to water mass ratios of ~2.6 and higher are possible. The sodium salt of phenol, sodium phenoxide, is far more water-soluble. It is a combustible solid (NFPA rating = 2). When heated, phenol produces flammable vapors that are explosive at concentrations of 3 to 10% in air. Carbon dioxide or dry chemical extinguishers should be used to fight phenol fires.[5]
Acidity
Phenol is a weak acid (pH 6.6). In aqueous solution in the pH range ca. 8 - 12 it is in equilibrium with the phenolate
Phenol is more acidic than aliphatic alcohols. Its enhanced acidity is attributed to
Hydrogen bonding
In
Tautomerism

Phenol exhibits
For substituted phenols, several factors can favor the keto tautomer: (a) additional hydroxy groups (see resorcinol) (b) annulation as in the formation of naphthols, and (c) deprotonation to give the phenolate.[18]
Phenoxides are enolates stabilised by aromaticity. Under normal circumstances, phenoxide is more reactive at the oxygen position, but the oxygen position is a "hard" nucleophile whereas the alpha-carbon positions tend to be "soft".[19]
Reactions

Phenol is highly reactive toward
Phenol is so strongly activated that bromination and chlorination lead readily to polysubstitution.
Aqueous solutions of phenol are weakly acidic and turn blue litmus slightly to red. Phenol is neutralized by sodium hydroxide forming sodium phenate or phenolate, but being weaker than carbonic acid, it cannot be neutralized by sodium bicarbonate or sodium carbonate to liberate carbon dioxide.
- C6H5OH + NaOH → C6H5ONa + H2O
When a mixture of phenol and benzoyl chloride are shaken in presence of dilute sodium hydroxide solution, phenyl benzoate is formed. This is an example of the Schotten–Baumann reaction:
- C6H5COCl + HOC6H5 → C6H5CO2C6H5 + HCl
Phenol is reduced to benzene when it is distilled with zinc dust or when its vapour is passed over granules of zinc at 400 °C:[22]
- C6H5OH + Zn → C6H6 + ZnO
When phenol is treated with diazomethane in the presence of boron trifluoride (BF3), anisole is obtained as the main product and nitrogen gas as a byproduct.
- C6H5OH + CH2N2 → C6H5OCH3 + N2
Phenol and its derivatives react with iron(III) chloride to give intensely colored solutions containing phenoxide complexes.
Production
Because of phenol's commercial importance, many methods have been developed for its production, but the cumene process is the dominant technology.
Cumene process

Accounting for 95% of production (2003) is the
A route analogous to the cumene process begins with
Oxidation of benzene, toluene, cyclohexylbenzene
The direct oxidation of benzene (C6H6) to phenol is theoretically possible and of great interest, but it has not been commercialized:
- C6H6 + O → C6H5OH
Nitrous oxide is a potentially "green" oxidant that is a more potent oxidant than O2. Routes for the generation of nitrous oxide however remain uncompetitive.[26][23][25]
An electrosynthesis employing alternating current gives phenol from benzene.[27]
The oxidation of
- C6H5CH3 + 2 O2 → C6H5OH + CO2 + H2O
The reaction is proposed to proceed via formation of benzyoylsalicylate.[8]
Autoxidation of cyclohexylbenzene give the hydroperoxide. Decomposition of this hydroperoxide affords cyclohexanone and phenol.[8]
Older methods
Early methods relied on extraction of phenol from coal derivatives or the hydrolysis of benzene derivatives.
Hydrolysis of benzenesulfonic acid
The original commercial route was developed by
- C6H5SO3H + 2 NaOH → C6H5OH + Na2SO3 + H2O
Hydrolysis of chlorobenzene
Chlorobenzene can be hydrolyzed to phenol using base (Dow process) or steam (Raschig–Hooker process):[24][25][29]
- C6H5Cl + NaOH → C6H5OH + NaCl
- C6H5Cl + H2O → C6H5OH + HCl
These methods suffer from the cost of the chlorobenzene and the need to dispose of the chloride by product.
Coal pyrolysis
Phenol is also a recoverable byproduct of coal pyrolysis.[29] In the Lummus process, the oxidation of toluene to benzoic acid is conducted separately.
Miscellaneous methods
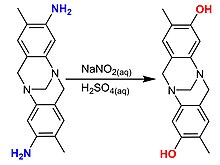
Salicylic acid decarboxylates to phenol.[31]
Uses
The major uses of phenol, consuming two thirds of its production, involve its conversion to precursors for plastics.
Phenol is also a versatile precursor to a large collection of drugs, most notably
Phenol is so inexpensive that it also attracts many small-scale uses. It is a component of industrial paint strippers used in the aviation industry for the removal of epoxy, polyurethane and other chemically resistant coatings.[33] Due to safety concerns, phenol is banned from use in cosmetic products in the European Union[34][35] and Canada.[36][37]
Medical
Phenol was widely used as an
Concentrated liquid phenol can be used topically as a local anesthetic for
History
Phenol was discovered in 1834 by
The antiseptic properties of phenol were used by Sir Joseph Lister in his pioneering technique of antiseptic surgery. Lister decided that the wounds had to be thoroughly cleaned. He then covered the wounds with a piece of rag or lint[44] covered in phenol. The skin irritation caused by continual exposure to phenol eventually led to the introduction of aseptic (germ-free) techniques in surgery. Lister's work was inspired by the works and experiments of his contemporary Louis Pasteur in sterilizing various biological media. He theorized that if germs could be killed or prevented, no infection would occur. Lister reasoned that a chemical could be used to destroy the micro-organisms that cause infection.[45]
Meanwhile, in Carlisle, England, officials were experimenting with sewage treatment using carbolic acid to reduce the smell of sewage cesspools. Having heard of these developments, and having previously experimented with other chemicals for antiseptic purposes without much success, Lister decided to try carbolic acid as a wound antiseptic. He had his first chance on August 12, 1865, when he received a patient: an eleven-year-old boy with a tibia bone fracture which pierced the skin of his lower leg. Ordinarily, amputation would be the only solution. However, Lister decided to try carbolic acid. After setting the bone and supporting the leg with splints, he soaked clean cotton towels in undiluted carbolic acid and applied them to the wound, covered with a layer of tin foil, leaving them for four days. When he checked the wound, Lister was pleasantly surprised to find no signs of infection, just redness near the edges of the wound from mild burning by the carbolic acid. Reapplying fresh bandages with diluted carbolic acid, the boy was able to walk home after about six weeks of treatment.[46]
By 16 March 1867, when the first results of Lister's work were published in the Lancet, he had treated a total of eleven patients using his new antiseptic method. Of those, only one had died, and that was through a complication that was nothing to do with Lister's wound-dressing technique. Now, for the first time, patients with compound fractures were likely to leave the hospital with all their limbs intact
- — Richard Hollingham, Blood and Guts: A History of Surgery, p. 62[46]
Before antiseptic operations were introduced at the hospital, there were sixteen deaths in thirty-five surgical cases. Almost one in every two patients died. After antiseptic surgery was introduced in the summer of 1865, there were only six deaths in forty cases. The mortality rate had dropped from almost 50 per cent to around 15 per cent. It was a remarkable achievement
- — Richard Hollingham, Blood and Guts: A History of Surgery, p. 63[47]
Phenol was the main ingredient of the "carbolic smoke ball," an ineffective device marketed in London in the 19th century as protection against influenza and other ailments, and the subject of the famous law case
Second World War
The toxic effect of phenol on the central nervous system causes sudden collapse and loss of consciousness in both humans and animals; a state of cramping precedes these symptoms because of the motor activity controlled by the central nervous system.
Occurrences
Phenol is a normal metabolic product, excreted in quantities up to 40 mg/L in human urine.
Phenol is a measurable component in the aroma and taste of the distinctive
Biodegradation
Toxicity
Phenol and its vapors are corrosive to the eyes, the skin, and the respiratory tract.
Since phenol is absorbed through the skin relatively quickly, systemic poisoning can occur in addition to the local caustic burns.[48] Resorptive poisoning by a large quantity of phenol can occur even with only a small area of skin, rapidly leading to paralysis of the central nervous system and a severe drop in body temperature. The LD50 for oral toxicity is less than 500 mg/kg for dogs, rabbits, or mice; the minimum lethal human dose was cited as 140 mg/kg.[48] The Agency for Toxic Substances and Disease Registry (ATSDR), U.S. Department of Health and Human Services states the fatal dose for ingestion of phenol is from 1 to 32 g.[65]
Phenols
The word phenol is also used to refer to any compound that contains a six-membered
See also
References
- ISBN 978-0-85404-182-4.)
Only one name is retained, phenol, for C6H5-OH, both as a preferred name and for general nomenclature.
{{cite book}}: CS1 maint: DOI inactive as of April 2024 (link - ^ "Phenol_msds".
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0493". National Institute for Occupational Safety and Health (NIOSH).
- PMID 18324831.
- ^ a b c d e f "Phenol". PubChem, US National Library of Medicine. 10 June 2023. Retrieved 12 June 2023.
- ^ a b c Sigma-Aldrich Co., Phenol. Retrieved on 2022-02-15.
- ^ a b c "Phenol". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ ISBN 978-3527306732.
- ISBN 9780470857274.
- ISBN 978-0-471-72091-1
- ISBN 0-534-07968-7
- ^ Drago, R S. Physical Methods For Chemists, (Saunders College Publishing 1992), ISBN 0-03-075176-4
- ^ Laurence, C. and Gal, J-F. Lewis Basicity and Affinity Scales, Data and Measurement, (Wiley 2010) pp 50-51 ISBN 978-0-470-74957-9
- doi:10.1021/ed054p612. The plots shown in this paper used older parameters. Improved E&C parameters are listed in ECW model.
- .
- ISBN 978-0-19-850346-0.
- .
- ISBN 0471497371.
- doi:10.15227/orgsyn.046.0115. Archived from the originalon 2011-06-05. Retrieved 2010-03-31.
- ISBN 978-3527306732.
- ISBN 0471497371.
- ^ Roscoe, Henry (1891). A treatise on chemistry, Volume 3, Part 3. London: Macmillan & Co. p. 23.
- ^ a b "Phenol -- The essential chemical industry online". 2017-01-11. Retrieved 2018-01-02.
- ^ a b "Direct Routes to Phenol". Archived from the original on 2007-04-09. Retrieved 2007-04-09.
- ^ a b c Plotkin, Jeffrey S. (2016-03-21). "What's New in Phenol Production?". American Chemical Society. Archived from the original on 2019-10-27. Retrieved 2018-01-02.
- .
- PMID 22684819.
- ^ Wittcoff, H.A., Reuben, B.G. Industrial Organic Chemicals in Perspective. Part One: Raw Materials and Manufacture. Wiley-Interscience, New York. 1980.
- ^ a b Franck, H.-G., Stadelhofer, J.W. Industrial Aromatic Chemistry. Springer-Verlag, New York. 1988. pp. 148-155.
- ^ S2CID 99294625.
- .
- ISBN 978-3527306732.
- ^ "CH207 Aircraft paintstripper, phenolic, acid" (PDF). Callington. 14 October 2009. Archived from the original (PDF) on 23 September 2015. Retrieved 25 August 2015.
- ^ "Prohibited substances in cosmetic product (Annex II, #1175, Phenol) - European Commission". ec.europa.eu. Retrieved 2018-07-06.
- ^ "CosIng - Cosmetics - GROWTH - European Commission". ec.europa.eu. Retrieved 2018-07-06.
- ^ Canada, Health (2004-06-18). "Cosmetic Ingredient Hotlist - Canada.ca". www.canada.ca. Retrieved 2018-07-06.
- ^ Canada, Health (2004-06-18). "Cosmetic Ingredient Hotlist: Prohibited and Restricted Ingredients - Canada.ca". www.canada.ca. Retrieved 2018-07-06.
- ^ "Phenol spray". drugs.com.
- ^ "How Does Our Lip Balm Work". Carmex. Archived from the original on 18 February 2015. Retrieved 18 February 2015.
- ^ F. F. Runge (1834) "Ueber einige Produkte der Steinkohlendestillation" (On some products of coal distillation), Annalen der Physik und Chemie, 31: 65-78. On page 69 of volume 31, Runge names phenol "Karbolsäure" (coal-oil-acid, carbolic acid). Runge characterizes phenol in: F. F. Runge (1834) "Ueber einige Produkte der Steinkohlendestillation," Annalen der Physik und Chemie, 31: 308-328.
- ^ Auguste Laurent (1841) "Mémoire sur le phényle et ses dérivés" (Memoir on benzene and its derivatives), Annales de Chimie et de Physique, series 3, 3: 195-228. On page 198, Laurent names phenol "hydrate de phényle" and "l'acide phénique".
- ^ Auguste Laurent (1836) "Sur la chlorophénise et les acides chlorophénisique et chlorophénèsique," Annales de Chemie et de Physique, vol. 63, pp. 27–45, see p. 44: Je donne le nom de phène au radical fondamental des acides précédens (φαινω, j'éclaire), puisque la benzine se trouve dans le gaz de l'éclairage. (I give the name of "phène" (φαινω, I illuminate) to the fundamental radical of the preceding acid, because benzene is found in illuminating gas.)
- ^ Gerhardt, Charles (1843) "Recherches sur la salicine," Annales de Chimie et de Physique, series 3, 7: 215-229. Gerhardt coins the name "phénol" on page 221.
- ^ Lister, Joseph (1867). "Antiseptic Principle Of The Practice Of Surgery".
- ISBN 9781407024530.
- ^ ISBN 9781407024530.
- ISBN 9781407024530.
- ^ a b c d e "Phenol". Ullmann's Encyclopedia of Industrial Chemistry. Vol. 25. Wiley-VCH. 2003. pp. 589–604.
- ^ The Experiments by Peter Tyson. NOVA
- ^ The Nazi Doctors Archived 2017-10-22 at the Wayback Machine, Chapter 14, Killing with Syringes: Phenol Injections. By Dr. Robert Jay Lifton
- ^ "Killing through phenol injection". Auschwitz: Final Station Extermination. Linz, Austria: Johannes Kepler University. Archived from the original on 2006-11-12.
- S2CID 21368454.
- ^ Musth in elephants. Deepa Ananth, Zoo's print journal, 15(5), pages 259-262 (article)
- ^ The Beaver: Its Life and Impact. Dietland Muller-Schwarze, 2003, page 43 (book at google books)
- ^ a b "Peat, Phenol and PPM, by Dr P. Brossard" (PDF). Retrieved 2008-05-27.
- ^ "Bruichladdich". Bruichladdich. BDCL. Archived from the original on 21 April 2016. Retrieved 8 August 2015.
- PMID 15653882.
- PMID 16261859.
- ^ a b Budavari, S, ed. (1996). The Merck Index: An Encyclopedia of Chemical, Drugs, and Biologicals. Whitehouse Station, NJ: Merck.
- PMID 16621299.
- PMID 2579602.
- ^ World Health Organization/International Labour Organization: International Chemical Safety Cards, http://www.inchem.org/documents/icsc/icsc/eics0070.htm
- ^ U.S. Department of Health and Human Services. "How can phenol affect my health?" (PDF). Toxicological Profile for Phenol: 24.
- PMID 10903419.
- ^ "Medical Management Guidelines for Phenol (C6H6O)". Agency for Toxic Substances and Disease Registry. U.S. Department of Health and Human Services. October 21, 2014. Retrieved 8 August 2015.
- PMID 1109265.
- PMID 1416322.
- PMID 625760.
External links
- International Chemical Safety Card 0070
- Phenol Material Safety Data Sheet
- National Pollutant Inventory: Phenol Fact Sheet
- NIOSH Pocket Guide to Chemical Hazards
- CDC - Phenol - NIOSH Workplace Safety and Health Topic
- IARC Monograph: "Phenol"
- Arcane Radio Trivia outlines competing uses for Phenol circa 1915