Phenol formaldehyde resin

| |
| Identifiers | |
|---|---|
| ChemSpider |
|
ECHA InfoCard
|
100.105.516 |
CompTox Dashboard (EPA)
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phenol formaldehyde resins (PF) (phenolic resins or phenoplasts
There are two main production methods. One reacts phenol and formaldehyde directly to produce a thermosetting network polymer, while the other restricts the formaldehyde to produce a prepolymer known as novolac which can be moulded and then cured with the addition of more formaldehyde and heat.[2][3] There are many variations in both production and input materials that are used to produce a wide variety of resins for special purposes.
Formation and structure
Phenol-formaldehyde resins, as a group, are formed by a
Phenol reacts with formaldehyde at the
- HOC6H5 + CH2O → HOC6H4CH2OH
The hydroxymethyl group is capable of reacting with either another free ortho or para site, or with another hydroxymethyl group. The first reaction gives a methylene bridge, and the second forms an ether bridge:
- HOC6H4CH2OH + HOC6H5 → (HOC6H4)2CH2 + H2O
- 2 HOC6H4CH2OH → (HOC6H4CH2)2O + H2O
The diphenol (HOC6H4)2CH2 (sometimes called a "dimer") is called
Novolaks
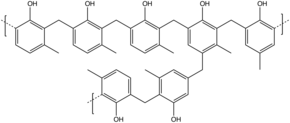
Novolaks (or novolacs) are phenol-formaldehyde resins with a formaldehyde to phenol molar ratio of less than one. In place of phenol itself, they are often produced from cresols (methylphenols). The polymerization is brought to completion using acid-catalysis such as sulfuric acid, oxalic acid, hydrochloric acid and rarely, sulfonic acids.[4] The phenolic units are mainly linked by methylene and/or ether groups. The molecular weights are in the low thousands, corresponding to about 10–20 phenol units. Obtained polymer is thermoplastic and require a curing agent or hardener to form a thermoset.
Hexamethylenetetramine is a hardener added to crosslink novolac. At a temperature greater than 90 °C, it forms methylene and dimethylene amino bridges. Resoles can also be used as a curing agent (hardener) for novolac resins. In either case, the curing agent is a source of formaldehyde which provides bridges between novolac chains, eventually completely crosslinking the system.[2]
Novolacs have multiple uses as tire
Resoles

Base-catalysed phenol-formaldehyde resins are made with a formaldehyde to phenol ratio of greater than one (usually around 1.5). These resins are called resoles. Phenol, formaldehyde, water and catalyst are mixed in the desired amount, depending on the resin to be formed, and are then heated. The first part of the reaction, at around 70 °C, forms a thick reddish-brown tacky material, which is rich in hydroxymethyl and benzylic ether groups.
The rate of the base-catalysed reaction initially increases with
Being
Resoles are major polymeric resin materials widely used for gluing and bonding building materials. Exterior plywood,
Crosslinking and the formaldehyde/phenol ratio
When the molar ratio of formaldehyde:phenol reaches one, in theory every phenol is linked together via methylene bridges, generating one single molecule, and the system is entirely crosslinked. This is why novolacs (F:P <1) do not harden without the addition of a crosslinking agents, and why resoles with the formula F:P >1 will.
Applications
Phenolic resins are found in myriad industrial products. Phenolic
Phenolic resins are also used for making exterior plywood commonly known as weather and boil proof (WBP) plywood because phenolic resins have no melting point but only a decomposing point in the temperature zone of 220 °C (428 °F) and above.
Phenolic resin is used as a
Higher end billiard balls are made from phenolic resins, as opposed to the polyesters used in less expensive sets.
Sometimes people select fibre reinforced phenolic resin parts because their
The Dutch painting forger Han van Meegeren mixed phenol formaldehyde with his oil paints before baking the finished canvas, in order to fake the drying out of the paint over the centuries.
Atmospheric re-entry spacecraft use phenol formaldehyde resin as a key component in ablative heat shields (e.g. AVCOAT on the Apollo modules). As the heat shield skin temperature can reach 1000-2000 °C, the resin pyrolizes due to aerodynamic heating. This reaction absorbs significant thermal energy, insulating the deeper layers of the heat shield. The outgassing of pyrolisis reaction products and the removal of charred material by friction (ablation) also contribute to vehicle insulation, by mechanically carrying away the heat absorbed in those materials.
Trade names

- wood flour.
- woodwindinstruments.
- Novotext is cotton fibre-reinforced phenolic, using randomly oriented fibres.
- Oasis Floral Foam is "an open-celled phenolic foam that readily absorbs water and is used as a base for flower arrangements."[6]
- printed circuit boards, although it is being replaced by fiberglass composites in many applications.
- Tufnol is a laminated plastic available as sheet and rods, which is made from layers of paper or cloth which have been soaked with phenolic resin and pressed under heat. Its high resistance to oils and solvents have made it suitable for many engineering applications.[7]
- Richlite is a paper-filled phenolic resin with many uses, from tabletops and cutting-boards to guitar fingerboards.
Biodegradation
Phenol-formaldehyde is degraded by the white rot fungus
See also
References
- ^ "Phenoplasts". polymerdatabase.com. Retrieved 13 June 2021.
- ^ a b A. Gardziella, L.A. Pilato, A. Knop, Phenolic Resins: Chemistry, Applications, Standardization, Safety and Ecology, 2nd edition, Springer, 2000
- .
- ISBN 9780819410191.
- ^ J. G. Ferguson, W. E. Grutzner, D. C. Koehler, R. S. Skinner, M. T. Skubiak, and D. H. Wetherell. "No. 1 ESS Apparatus and Equipment". The Bell System Technical Journal. 1964. p. 2417.
- ^ "Smithers Oasis Resource Center". Archived from the original on 7 April 2008. Retrieved 21 October 2010.
- ^ Godwin, R. (April 2015). "What is Tufnol?". ahistoryoftufnol.org.
- PMID 16856735.
