Phentermine
This article may be confusing or unclear to readers. In particular, the lead states that weight loss stops after a few weeks, but "Medical use" states that it continues to occur "through the ninth month", so which is it?. (May 2021) |
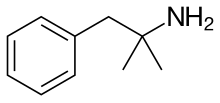 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Ionamin, Suprenza, others |
| Other names | α-methyl-amphetamine α,α-dimethylphenethylamine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682187 |
| Pregnancy category |
|
| Dependence liability | Physical: not typical Appetite suppressant[3] |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | High (almost complete)[5] |
| Protein binding | Approximately 96.3% |
| Metabolism | Liver[5] |
| Elimination half-life | 25 hours, urinary pH-dependent[5] |
| Excretion | Kidney (62–85% unchanged)[5] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Phentermine (
Common side effects include a fast heart beat, high blood pressure, trouble sleeping, dizziness, and restlessness.
Phentermine was approved for medical use in the United States in 1959.
Medical uses
Phentermine is used for a short period of time to promote weight loss, if exercise and calorie reduction are not sufficient, and in addition to exercise and calorie reduction.[5][12]
Phentermine is approved for up to 12 weeks of use and most weight loss occurs in the first weeks.[12] However, significant loss continues through the sixth month and has been shown to continue at a slower rate through the ninth month.[13]
Contraindications
Phentermine is
- have a history of drug abuse
- are allergic to sympathomimetic aminedrugs
- are taking a monoamine oxidase inhibitor (MAOI) or have taken one within the last 14 days
- have cardiovascular disease, hyperthyroidism, or glaucoma
- are breast-feeding.
Adverse effects
Tolerance usually occurs; however, risks of dependence and addiction are considered negligible.[13][14] People taking phentermine may be impaired when driving or operating machinery.[12] Consumption of alcohol with phentermine may produce adverse effects.[12]
There is currently no evidence regarding whether or not phentermine is safe for pregnant women.[5][12]
Other adverse effects include:[5][12]
- Cardiovascular effects like palpitations, tachycardia, high blood pressure, precordial pain; rare cases of stroke, angina, myocardial infarction, cardiac failure and cardiac arrest have been reported.
- Central nervous system effects like overstimulation, restlessness, nervousness, insomnia, tremor, dizziness and headache; there are rare reports of euphoria followed by fatigue and depression, and very rarely, psychotic episodes and hallucinations.
- Gastrointestinal effects include nausea, vomiting, dry mouth, cramps, unpleasant taste, diarrhea, and constipation.
- Other adverse effects include trouble urinating, rash, impotence, changes in libido, and facial swelling.
Interactions
Phentermine may decrease the effect of drugs like clonidine, methyldopa, and guanethidine. Drugs to treat hypothyroidism may increase the effect of phentermine.[12]
Mechanism of action
Phentermine has some similarity in its
History
In 1959, phentermine first received approval from the United States FDA as an appetite-suppressing drug.[17] Eventually a hydrochloride salt and a resin form became available.[17]
Phentermine was marketed with
Phentermine is still available by itself in most countries, including the US.[17] However, because it is similar to amphetamine, it is classified as a controlled substance in many countries. Internationally, phentermine is a schedule IV drug under the Convention on Psychotropic Substances.[21] In the United States, it is classified as a Schedule IV controlled substance under the Controlled Substances Act. In contrast, amphetamine preparations are classified as Schedule II controlled substances.[22]
A company called Vivus developed a combination drug, phentermine/topiramate that it originally called Qnexa and then called Qsymia, which was invented and used off-label by Thomas Najarian, who opened a weight-clinic in Los Osos, California in 2001; Najarian had previously worked at Interneuron Pharmaceuticals, which had developed one of the fen-phen drugs previously withdrawn from the market.[23] The FDA rejected the combination drug in 2010 due to concerns over its safety.[23] In 2012 the FDA approved it after Vivus re-applied with further safety data.[24] At the time, one obesity specialist estimated that around 70% of his colleagues were already prescribing the combination off-label.[23]
Chemistry
Phentermine is a
.Names
The term ‘phentermine' is contracted from
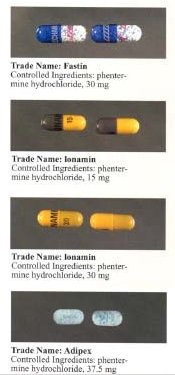
It is marketed under many brand names and formulations worldwide, including Acxion, Adipex, Adipex-P, Duromine, Elvenir, Fastin, Ionamin, Lomaira (phentermine hydrochloride), Panbesy, Qsymia (phentermine and topiramate), Razin, Redusa, Sentis, Suprenza, and Terfamex.[25]
References
- ISBN 9781284118971.
- ISBN 9781605472645.
- ^ a b c d e f g h i "Phentermine Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 13 April 2019.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b c d e f g h "METERMINE (Phentermine)" (PDF). TGA eBusiness Services. iNova Pharmaceuticals (Australia) Pty Limited. 22 July 2013. Retrieved 16 November 2013.
- ^ "Phentermine and topiramate Uses, Side Effects & Warnings". Drugs.com. Retrieved 13 April 2019.
- ^ "Phentermine Use During Pregnancy". Drugs.com. Retrieved 13 April 2019.
- ^ PMID 22502775.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Phentermine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ISBN 9781439854259.
- ^ a b c d e f g h "Phentermine label at FDA" (Last updated: January 2012). FDA. Retrieved 13 October 2016.
- ^ PMID 11493122.
- ^ PMID 26811245.
- PMID 18524885.
we confirmed agonistic activity at human TAAR1 of several other compounds, including the trace amines octopamine and tryptamine, the amphetamine derivatives l-amphetamine, d-methamphetamine, (+)-MDMA, and phentermine, and the catecholamine metabolites 3-MT and 4-MT (Bunzow et al., 2001; Lindemann and Hoener, 2005; Reese et al., 2007; Wainscott et al., 2007; Wolinsky et al., 2007; Xie and Miller, 2007; Xie et al., 2007).
- S2CID 15573624.
- ^ ISBN 9781841849829.
- ^ Kolata, Gina (23 September 1997). "How Fen-Phen, A Diet 'Miracle,' Rose and Fell". New York Times. NY, NY, USA.
- ^ "FDA Announces Withdrawal Fenfluramine and Dexfenfluramine (Fen-Phen)". Fda.gov. Retrieved 12 July 2013.
- PMID 12788841.
- ^ Convention on Psychotropic Substances Archived 14 March 2014 at the Wayback Machine
- S2CID 20072687.
- ^ a b c Pollack A (16 February 2012). "Diet Treatment, Already in Use, to Get F.D.A. Review". The New York Times.
- ^ "FDA approves weight-management drug Qsymia". FDA. 17 July 2012.
- ^ "International brands for phentermine". Drugs.com. Retrieved 13 October 2016.
External links
- "Phentermine". International Programme on Chemical Safety (IPCS). the World Health Organization (WHO).
