Phenylacetylene

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Ethynylbenzene | |
| Other names
Phenylacetylene
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.007.861 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H6 | |
| Molar mass | 102.133 g/mol |
| Density | 0.93 g/cm3 |
| Melting point | −45 °C (−49 °F; 228 K) |
| Boiling point | 142 to 144 °C (288 to 291 °F; 415 to 417 K) |
| Acidity (pKa) | 28.7 (DMSO),[1] 23.2 (aq, extrapolated)[2] |
| -72.01·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phenylacetylene is an
phenyl group. It exists as a colorless, viscous liquid. In research, it is sometimes used as an analog for acetylene
; being a liquid, it is easier to handle than acetylene gas.
Preparation
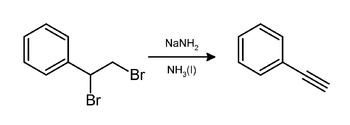
In the laboratory, phenylacetylene can be prepared by elimination of hydrogen bromide from styrene dibromide using sodium amide in ammonia:[3]
It can also be prepared by the elimination of hydrogen bromide from bromostyrene using molten
TBAF.[5]
Reactions
Phenylacetylene is a prototypical terminal acetylene, undergoing many reactions expected of that functional group. It undergoes semi
In the presence of gold or mercury reagents, phenylacetylene hydrates to give acetophenone:
- PhC2H + H2O → PhC(O)CH3
See also
References
- .
- .
- .
- .
- ISBN 978-0471936237.
- .
- PMID 15756340.
- .