Phosgene
| |

| |
 A sample case of toxic gases used in chemical warfare; the leftmost contains phosgene in a sealed capillary
| |
| Names | |
|---|---|
| Preferred IUPAC name
Carbonyl dichloride[2] | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.000.792 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 1076 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| COCl2 | |
| Molar mass | 98.91 g·mol−1 |
| Appearance | Colorless gas |
| Odor | Suffocating, like musty hay or grass[3] |
| Density | 4.248 g/L (15 °C, gas) 1.432 g/cm3 (0 °C, liquid) |
| Melting point | −118 °C (−180 °F; 155 K) |
| Boiling point | 8.3 °C (46.9 °F; 281.4 K) |
| Insoluble, reacts[4] | |
| Solubility | Soluble in benzene, toluene, acetic acid Decomposes in alcohol and acid |
| Vapor pressure | 1.6 atm (20°C)[3] |
| −48·10−6 cm3/mol | |
| Structure | |
Trigonal planar
| |
| 1.17 D | |
| Hazards | |
| GHS labelling: | |
   [5] [5]
| |
| Danger | |
| H280, H314, H330[5] | |
| P260, P280, P303+P361+P353+P315, P304+P340+P315, P305+P351+P338+P315, P403, P405[5] | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable
|
Threshold limit value (TLV)
|
0.1 mg /m3)
|
| Lethal dose or concentration (LD, LC): | |
LC50 (median concentration)
|
|
LCLo (lowest published)
|
|
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.1 ppm (0.4 mg/m3)[3] |
REL (Recommended)
|
TWA 0.1 ppm (0.4 mg/m3) C 0.2 ppm (0.8 mg/m3) [15-minute][3] |
IDLH (Immediate danger) |
2 ppm[3] 1 ppm = 4 mg/m3 |
| Safety data sheet (SDS) | [1] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phosgene is an
Phosgene is extremely poisonous and was used as a chemical weapon during World War I, where it was responsible for 85,000 deaths. It is a highly potent pulmonary irritant and quickly filled enemy trenches due to it being a heavy gas.
It is classified as a
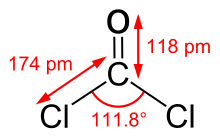
Structure and basic properties
Phosgene is a planar molecule as predicted by
Production
Industrially, phosgene is produced by passing purified
- CO + Cl2 → COCl2 (ΔHrxn = −107.6 kJ/mol)
This reaction is exothermic and is typically performed between 50 and 150 °C. Above 200 °C, phosgene reverts to carbon monoxide and chlorine, Keq(300 K) = 0.05. World production of this compound was estimated to be 2.74 million tonnes in 1989.[8]
Phosgene is fairly simple to produce, but is listed as a Schedule 3 substance under the Chemical Weapons Convention. As such, it is usually considered too dangerous to transport in bulk quantities. Instead, phosgene is usually produced and consumed within the same plant, as part of an "on demand" process. This involves maintaining equivalent rates of production and consumption, which keeps the amount of phosgene in the system at any one time fairly low, reducing the risks in the event of an accident. Some batch production does still take place, but efforts are made to reduce the amount of phosgene stored.[10]
Inadvertent generation
Atmospheric chemistry
Simple
Phosgene in the troposphere can last up to about 70 days and is removed primarily by hydrolysis with ambient humidity or cloudwater.[13] Less than 1% makes it to the stratosphere, where it is expected to have a lifetime of several years, since this layer is much drier and phosgene decomposes slowly through UV photolysis. Consequently, it does play a minor part in ozone depletion.
Combustion
Carbon tetrachloride (CCl4) can turn into phosgene when exposed to heat in air. This was a problem as carbon tetrachloride is an effective fire suppressant and was formerly in widespread use in fire extinguishers.[14] There are reports of fatalities caused by its use to fight fires in confined spaces.[15] Carbon tetrachloride's generation of phosgene and its own toxicity mean it is no longer used for this purpose.[14]
Biologically
Phosgene is also formed as a metabolite of
History
Phosgene was synthesized by the Cornish chemist John Davy (1790–1868) in 1812 by exposing a mixture of carbon monoxide and chlorine to sunlight. He named it "phosgene" from Greek φῶς (phos, light) and γεννάω (gennaō, to give birth) in reference of the use of light to promote the reaction.[17] It gradually became important in the chemical industry as the 19th century progressed, particularly in dye manufacturing.
Reactions and uses
The reaction of an organic substrate with phosgene is called phosgenation.
- n HO−CR2−X−CR2−OH + n COCl2 → [−O−CR2−X−CR2−O−C(=O)−]n + 2n HCl
An example is the reaction of phosgene with
Aside from the widely used reactions described above, phosgene is also used to produce acyl chlorides from carboxylic acids:
- R−C(=O)−OH + COCl2 → R−C(=O)−Cl + HCl + CO2
For this application, thionyl chloride is commonly used instead of phosgene.
Laboratory uses
The synthesis of
- R−NH2 + COCl2 → R−N=C=O + 2 HCl, where R = aryl
Such reactions are conducted on laboratory scale in the presence of a base such as pyridine that neutralizes the hydrogen chloride side-product.
Phosgene is used to produce chloroformates such as benzyl chloroformate:
- R−OH + COCl2 → R−O−C(=O)−Cl + HCl
In these syntheses, phosgene is used in excess to prevent formation of the corresponding carbonate ester.
With amino acids, phosgene (or its trimer) reacts to give amino acid N-carboxyanhydrides. More generally, phosgene acts to link two nucleophiles by a carbonyl group. For this purpose, alternatives to phosgene such as carbonyldiimidazole (CDI) are safer, albeit expensive.[19] CDI itself is prepared by reacting phosgene with imidazole.
Phosgene is stored in metal cylinders. In the US, the cylinder valve outlet is a tapered thread known as "CGA 160" that is used only for phosgene.
Alternatives to phosgene
In the research laboratory, due to safety concerns phosgene nowadays finds limited use in organic synthesis. A variety of substitutes have been developed, notably trichloromethyl chloroformate ("diphosgene"), a liquid at room temperature, and bis(trichloromethyl) carbonate ("triphosgene"), a crystalline substance.[20]
Other reactions
Phosgene reacts with water to release hydrogen chloride and carbon dioxide:
- COCl2 + H2O → CO2 + 2 HCl
Analogously, upon contact with ammonia, it converts to urea:
- COCl2 + 4 NH3 → CO(NH2)2 + 2 [NH4]Cl
Halide exchange with
Chemical warfare

It is listed on
Phosgene was first deployed as a chemical weapon by the French in 1915 in World War I.[23] It was also used in a mixture with an equal volume of chlorine, with the chlorine helping to spread the denser phosgene.[24][25] Phosgene was more potent than chlorine, though some symptoms took 24 hours or more to manifest.
Following the extensive use of phosgene during World War I, it was stockpiled by various countries.[26][27][28]
Phosgene was then only infrequently used by the Imperial Japanese Army against the Chinese during the Second Sino-Japanese War.[29] Gas weapons, such as phosgene, were produced by the IJA's Unit 731.
Toxicology and safety
Phosgene is an insidious poison as the odor may not be noticed and symptoms may be slow to appear.[30]
The odor detection threshold for phosgene is 0.4 ppm, four times the threshold limit value (time weighted average). Its high toxicity arises from the action of the phosgene on the −OH, −NH2 and −SH groups of the proteins in pulmonary alveoli (the site of gas exchange), respectively forming ester, amide and thioester functional groups in accord with the reactions discussed above. This results in disruption of the blood–air barrier, eventually causing pulmonary edema. The extent of damage in the alveoli does not primarily depend on phosgene concentration in the inhaled air, with the dose (amount of inhaled phosgene) being the critical factor.[31] Dose can be approximately calculated as "concentration" × "duration of exposure".[31][32] Therefore, persons in workplaces where there exists risk of accidental phosgene release usually wear indicator badges close to the nose and mouth.[33] Such badges indicate the approximate inhaled dose, which allows for immediate treatment if the monitored dose rises above safe limits.[33]
In case of low or moderate quantities of inhaled phosgene, the exposed person is to be monitored and subjected to precautionary therapy, then released after several hours. For higher doses of inhaled phosgene (above 150 ppm × min) a pulmonary edema often develops which can be detected by X-ray imaging and regressive blood oxygen concentration. Inhalation of such high doses can eventually result in fatality within hours up to 2–3 days of the exposure.
The risk connected to a phosgene inhalation is based not so much on its toxicity (which is much lower in comparison to modern chemical weapons like sarin or tabun) but rather on its typical effects: the affected person may not develop any symptoms for hours until an edema appears, at which point it could be too late for medical treatment to assist.[34] Nearly all fatalities as a result of accidental releases from the industrial handling of phosgene occurred in this fashion. On the other hand, pulmonary edemas treated in a timely manner usually heal in the mid- and longterm, without major consequences once some days or weeks after exposure have passed.[35][36] Nonetheless, the detrimental health effects on pulmonary function from untreated, chronic low-level exposure to phosgene should not be ignored; although not exposed to concentrations high enough to immediately cause an edema, many synthetic chemists (e.g. Leonidas Zervas) working with the compound were reported to experience chronic respiratory health issues and eventual respiratory failure from continuous low-level exposure.
If accidental release of phosgene occurs in an industrial or laboratory setting, it can be mitigated with ammonia gas; in the case of liquid spills (e.g. of diphosgene or phosgene solutions) an absorbent and sodium carbonate can be applied.[37]
Accidents
- The first major phosgene-related incident happened in May 1928 when eleven tons of phosgene escaped from a war surplus store in central Hamburg.[38] Three hundred people were poisoned, of whom ten died.[38]
- In the second half of 20th century several fatal incidents implicating phosgene occurred in Europe, Asia and the US. Most of them have been investigated by authorities and the outcome made accessible to the public. For example, phosgene was initially blamed for the Bhopal disaster, but investigations proved methyl isocyanateto be responsible for the numerous poisonings and fatalities.
- Recent major incidents happened in January 2010 and May 2016. An accidental release of phosgene gas at a DuPont facility in West Virginia killed one employee in 2010.[39] The US Chemical Safety Board released a video detailing the accident.[40] Six years later, a phosgene leak occurred in a BASF plant in South Korea, where a contractor inhaled a lethal dose of phosgene.[41]
- 2023 Ohio train derailment: A freight train carrying vinyl chloride derailed and burned in East Palestine, Ohio, releasing phosgene and hydrogen chloride into the air and contaminating the Ohio River.[42]
See also
References
- ^ Merck Index, 11th Edition, 7310.
- ISBN 978-0-85404-182-4.
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0504". National Institute for Occupational Safety and Health (NIOSH).
- ^ "PHOSGENE (cylinder)". Inchem (Chemical Safety Information from Intergovernmental Organizations). International Programme on Chemical Safety and the European Commission.
- ^ a b c Record of Phosgene in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 16 March 2021.
- ^ a b "Phosgene". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ CBRNE - Lung-Damaging Agents, Phosgene May 27, 2009
- ^ ISBN 978-3527306732.
- .
- S2CID 110707551.
- ^ S2CID 4209599.
- S2CID 44164908.
- .
- ^ ISBN 978-0-203-48499-9.
- . Retrieved 2022-02-03.
- PMID 597296.
- JSTOR 107310. Phosgene was named on p. 151: " ... it will be necessary to designate it by some simple name. I venture to propose that of phosgene, or phosgene gas; from φως, light, γινομαι, to produce, which signifies formed by light; ... "
- ^ R. L. Shriner, W. H. Horne, and R. F. B. Cox (1943). "p-Nitrophenyl Isocyanate". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 2, p. 453. - doi:10.1039/B002127J.
- ^ Annex on Implementation and Verification ("Verification Annex") Archived 2006-05-15 at the Wayback Machine.
- ^ https://itportal.decc.gov.uk/cwc_files/S2AAD_guidance.pdf Archived 2016-03-04 at the Wayback Machine.
- ISBN 0-674-06382-1.
- ^ Staff (2004). "Choking Agent: CG". CBWInfo. Archived from the original on 2006-02-18. Retrieved 2007-07-30.
- ISBN 978-1-74045-970-9.
- ^ Base's phantom war reveals its secrets, Lithgow Mercury, 7/08/2008
- ^ Chemical warfare left its legacy Archived 2008-12-05 at the Wayback Machine, Lithgow Mercury, 9/09/2008
- ^ Chemical bombs sit metres from Lithgow families for 60 years, The Daily Telegraph, September 22, 2008
- ^ Yuki Tanaka, "Poison Gas, the Story Japan Would Like to Forget", Bulletin of the Atomic Scientists, October 1988, pp. 16–17
- S2CID 41169682.
- ^ a b Werner F. Diller, Early Diagnosis of Phosgene Overexposure.Toxicology and Industrial Health, Vol.1, Nr.2, April 1985, p. 73 -80
- ^ W. F. Diller, R. Zante : Zentralbl. Arbeitsmed. Arbeitsschutz Prophyl. Ergon. 32, (1982) 60 -368
- ^ a b W. F.Diller, E.Drope, E. Reichold: Ber. Int. Kolloq. Verhütung von Arbeitsunfällen und Berufskrankheiten Chem. Ind.6 th (1979) Chem. Abstr. 92 (1980) 168366x
- ^ W. F. Diller: Radiologische Untersuchungen zur verbesserten Frühdiagnose von industriellen Inhalationsvergiftungen mit verzögertem Wirkungseintritt, Verlag für Medizin Dr. E. Fischer, Heidelberg. Zentralbatt für Arbeitsmedizin, Arbeitsschutz und Ergonomie, Nr. 3, Mai 2013, p. 160 - 163
- ^ W.F. Diller, F. Schnellbächer, F. Wüstefeld : Zentralbl. Arbeitsmed. Arbeitsschutz Prophyl. 29 (1979) p.5-16
- ^ Results From the US Industry-Wide Phosgene Surveillance "The Diller Registry" : Journal of Occ. and Env. Med., March 2011-Vol.53-iss. 3 p.239- 244
- ^ "Phosgene: Health and Safety Guide". International Programme on Chemical Safety. 1998.
- ^ ISBN 0444824456.
- ^ "DuPont Corporation Toxic Chemical Releases | CSB".
- ^ Fatal Exposure: Tragedy at DuPont, retrieved 2021-07-02
- ^ Archived at Ghostarchive and the Wayback Machine: "Fatal Exposure: Tragedy at DuPont". YouTube.
- ^ "Ohio catastrophe is 'wake-up call' to dangers of deadly train derailments". The Guardian. February 11, 2023. Archived from the original on February 13, 2023. Retrieved February 13, 2023.
External links
- Davy's account of his discovery of phosgene
- International Chemical Safety Card 0007
- CDC - Phosgene - NIOSH Workplace Safety and Health Topic
- NIOSH Pocket Guide to Chemical Hazards
- U.S. CDC Emergency Preparedness & Response
- U.S. EPA Acute Exposure Guideline Levels
- Regime For Schedule 3 Chemicals And Facilities Related To Such Chemicals, OPCW website
- CBWInfo website
- Use of Phosgene in WWII and in modern-day warfare
- US Chemical Safety Board Video on accidental release at DuPont facility in West Virginia

