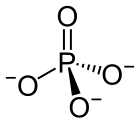
Phosphate
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Phosphate[1]
| |||
| Other names
Orthophosphate
Tetraoxophosphate(V) Tetraoxidophosphate(V) | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 3903772 | |||
| ChEBI | |||
| ChemSpider | |||
| 1997 | |||
| MeSH | Phosphates | ||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| PO3− 4 | |||
| Molar mass | 94.9714 g mol−1 | ||
Conjugate acid
|
Monohydrogen phosphate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
In
The phosphate or orthophosphate ion [PO
4]3−
is derived from phosphoric acid by the removal of three protons H+
. Removal of one proton gives the dihydrogen phosphate ion [H
2PO
4]−
while removal of two protons gives the hydrogen phosphate ion [HPO
4]2−
. These names are also used for salts of those anions, such as ammonium dihydrogen phosphate and trisodium phosphate.
-
[PO
4]3−
Phosphate or orthophosphate
In organic chemistry, phosphate or orthophosphate is an organophosphate, an ester of orthophosphoric acid of the form PO
4RR′R″ where one or more hydrogen atoms are replaced by organic groups. An example is trimethyl phosphate, (CH
3)
3PO
4. The term also refers to the trivalent functional group OP(O-)
3 in such esters. Phosphates may contain sulfur in place of one or more oxygen atoms (thiophosphates and organothiophosphates).
Orthophosphates are especially important among the various phosphates because of their key roles in biochemistry, biogeochemistry, and ecology, and their economic importance for agriculture and industry.[2] The addition and removal of phosphate groups (phosphorylation and dephosphorylation) are key steps in cell metabolism.
Orthophosphates can condense to form pyrophosphates.
Chemical properties
The phosphate ion has a
4)2−
, which in turn is the conjugate base of the dihydrogen phosphate ion H
2(PO
4)−
, which in turn is the conjugate base of orthophosphoric acid
3PO
4.
Many phosphates are
Equilibria in solution

In water solution, orthophosphoric acid and its three derived anions coexist according to the dissociation and recombination equilibria below[3]
| Equilibrium | Dissociation constant Ka[4] | pKa |
|---|---|---|
| H3PO4 ⇌ H2PO−4 + H+ | pKa1 = 2.14 | |
| H2PO−4 ⇌ HPO2−4 + H+ | pKa2 = 7.20 | |
| HPO2−4 ⇌ PO3−4 + H+ | pKa3 = 12.37 |
Values are at 25 °C and 0 ionic strength.
The pKa values are the
2PO
4]−
, is practically the only species present. Around pH 9.8 (mid-way between the second and third pKa values) the monohydrogen phosphate ion, [HPO
4]2−
, is the only species present. At pH 13 or higher, the acid is completely dissociated as the phosphate ion, (PO
4)3−
.
This means that salts of the mono- and di-phosphate ions can be selectively crystallised from aqueous solution by setting the pH value to either 4.7 or 9.8.
In effect, H
3PO
4, H
2(PO
4)−
and H(PO
4)2−
behave as separate
Phosphate can form many polymeric ions such as pyrophosphate, (P
2O
7)4−
, and triphosphate, (P
3O
10)5−
. The various metaphosphate ions (which are usually long linear polymers) have an empirical formula of (PO
3)−
and are found in many compounds.
Biochemistry of phosphates
In biological systems, phosphorus can be found as free phosphate anions in solution (inorganic phosphate) or bound to organic molecules as various organophosphates.
Inorganic phosphate is generally denoted Pi and at physiological (homeostatic) pH primarily consists of a mixture of [HPO
4]2−
and [H
2PO
4]−
ions. At a neutral pH, as in the cytosol (pH = 7.0), the concentrations of the orthophoshoric acid and its three anions have the ratios
Thus, only [H
2PO
4]−
and [HPO
4]2−
ions are present in significant amounts in the cytosol (62% [H
2PO
4]−
, 38% [HPO
4]2−
). In extracellular fluid (pH = 7.4), this proportion is inverted (61% [HPO
4]2−
, 39% [H
2PO
4]−
).
Inorganic phosphate can also be present as pyrophosphate anions [P
2O
7]4−
, which give orthophosphate by hydrolysis:
- [P
2O
7]4−
+ H2O ⇌ 2 [HPO
4]2−
Organic phosphates are commonly found in the form of esters as
Bones and teeth
An important occurrence of phosphates in biological systems is as the structural material of bone and teeth. These structures are made of crystalline
Medical and biological research uses
Phosphates are medicinal salts of phosphorus. Some phosphates, which help cure many
Plant metabolism
Plants take up phosphorus through several pathways: the
Adverse health effects
This article needs additional citations for verification. (July 2022) |
Hyperphosphatemia, or a high blood level of phosphates, is associated with elevated mortality in the general population. The most common cause of hyperphosphatemia in people, dogs, and cats is kidney failure. In cases of hyperphosphatemia, limiting consumption of phosphate-rich foods, such as some meats and dairy items and foods with a high phosphate-to-protein ratio, such as soft drinks, fast food, processed foods, condiments, and other products containing phosphate-salt additives is advised.[6]
Phosphates induce vascular calcification, and a high concentration of phosphates in blood was found to be a predictor of cardiovascular events.[7]
Production
Geological occurrence


Phosphates are the naturally occurring form of the element phosphorus, found in many phosphate minerals. In mineralogy and geology, phosphate refers to a rock or ore containing phosphate ions. Inorganic phosphates are mined to obtain phosphorus for use in agriculture and industry.[2]
The largest global producer and exporter of phosphates is
Phosphorite mines are primarily found in:
- North America: United States, especially Florida, with lesser deposits in North Carolina, Idaho, and Tennessee
- Africa: Morocco, Algeria, Egypt, Niger, Senegal, Togo, Tunisia, Mauritania
- Middle East: Saudi Arabia, Jordan, Israel, Syria, Iran and Iraq, at the town of Akashat, near the Jordanian border.
- Central Asia: Kazakhstan
- Oceania: Banaba Island
In 2007, at the current rate of consumption, the supply of phosphorus was estimated to run out in 345 years.
Some phosphate rock deposits, such as Mulberry in Florida,[14] are notable for their inclusion of significant quantities of radioactive uranium isotopes. This is a concern because radioactivity can be released into surface waters[15] from application of the resulting phosphate fertilizer.
In December 2012,
Around 2018, Norway discovered phosphate deposits almost equal to those in the rest of Earth combined.[17][18]
In July 2022 China announced quotas on phosphate exportation.[19]
The largest importers in millions of metric tons of phosphate are Brazil 3.2, India 2.9 and the USA 1.6.[20]
Mining
The three principal phosphate producer countries (China, Morocco and the United States) account for about 70% of world production.
| Country | Production (millions kg) |
Share of global production (%) |
Reserves (millions kg) |
|---|---|---|---|
| Algeria | 1,300 | 0.54 | 2,200,000 |
| Australia | 2,700 | 1.17 | 1,100,000 |
| Brazil | 4,700 | 3.00 | 1,600,000 |
| China | 95,000 | 44.83 | 3,200,000 |
| Egypt | 5,000 | 2.47 | 2,800,000 |
| Finland | 995 | - | 1,000,000 |
| India | 1,480 | 0.49 | 46,000 |
| Iraq | 200 | 0.09 | 430,000 |
| Israel | 2,810 | 1.48 | 57,000 |
| Jordan | 9,220 | 3.36 | 800,000 |
| Kazakhstan | 1,500 | 0.72 | 260,000 |
| Mexico | 558 | 0.76 | 30,000 |
| Morocco and Western Sahara | 35,500 | 13.45 | 50,000,000 |
| Peru | 4,000 | 1.79 | 210,000 |
| Russia | 13,100 | 5.60 | 600,000 |
| Saudi Arabia | 6,500 | 1.48 | 1,400,000 |
| Senegal | 3,420 | 0.45 | 50,000 |
| South Africa | 2,100 | 0.99 | 1,400,000 |
| Syria | 2,000 | 0.34 | 1,800,000 |
| Togo | 800 | 0.45 | 30,000 |
| Tunisia | 4,110 | 1.79 | 100,000 |
| Uzbekistan | 900 | - | 100,000 |
| United States | 23,300 | 12.37 | 1,000,000 |
| Vietnam | 4,650 | 1.21 | 30,000 |
| Other countries | 1,140 | 1.17 | 840,000 |
| Total | 227,000 | 100 | 71,000,000 |
Ecology
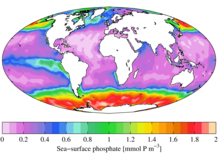

In ecological terms, because of its important role in biological systems, phosphate is a highly sought after resource. Once used, it is often a limiting nutrient in
Calcium hydroxyapatite and calcite precipitates can be found around
Phosphate deposits can contain significant amounts of naturally occurring heavy metals. Mining operations processing
See also
- Diammonium phosphate - (NH4)2HPO4
- Disodium phosphate – Na2HPO4
- Fertilizer
- Hypophosphite– H
2(PO
2)− - Metaphosphate – (POn
3) - Monosodium phosphate – NaH2PO4
- Organophosphoruscompounds
- Ouled Abdoun Basin
- Phosphate – OP(OR)3, such as triphenyl phosphate
- Phosphate conversion coating
- Phosphate soda, a soda fountain beverage
- Phosphinate – OP(OR)R2
- Phosphine – PR3
- Phosphine oxide – OPR3
- Phosphinite – P(OR)R2
- Phosphite– P(OR)3
- Phosphogypsum
- Phosphonate – OP(OR)2R
- Phosphonite – P(OR)2R
- Phosphorylation
- Polyphosphate – (HPO
3)
n - Pyrophosphate – (P
2O
7)4− - Sodium tripolyphosphate– Na5P3O10
References
- ^ "Phosphates – PubChem Public Chemical Database". The PubChem Project. USA: National Center of Biotechnology Information.
- ^ a b "Phosphate Primer". Florida Industrial and Phosphate Research Institute. Florida Polytechnic University. Archived from the original on 29 August 2017. Retrieved 30 March 2018.
- ISBN 0-8053-7171-0.
- ^ a b c "Phosphate Supplement (Oral Route, Parenteral Route) Description and Brand Names - Mayo Clinic". www.mayoclinic.org. Retrieved 2020-11-20.
- ^ Renal Dietitian Team, Reducing phosphate in your diet, Oxford University Hospitals NHS Foundation Trust, 2022 review
- PMID 22334826.
- .
- ^ Leo Lewis (2008-06-23). "Scientists warn of lack of vital phosphorus as biofuels raise demand". The Times.
- ^ U.S. Geological Survey Phosphate Rock
- U.S. Geological Survey "Phosphorus Soil Samples"(PDF).
- ^ Floor Anthoni. "Abundance of Elements". Seafriends.org.nz. Retrieved 2013-01-10.
- ^ American Geophysical Union, Fall Meeting 2007, abstract #V33A-1161. Mass and Composition of the Continental Crust
- ^ Central Florida Phosphate Industry: Environmental Impact Statement. Vol. 2. United States. Environmental Protection Agency. 1979.
- ^ C. Michael Hogan (2010). "Water pollution". In Mark McGinley and C. Cleveland (Washington, DC.: National Council for Science and the Environment) (ed.). Encyclopedia of Earth. Archived from the original on 2010-09-16.
- ^ "Updated Hinda Resource Announcement: Now world's largest phosphate deposit (04/12/2012)". Cominco Resources. Archived from the original on 2016-10-05. Retrieved 2013-05-03.
- ^ Bushuev, Mikhail (26 January 2021). "EU pins hope on Norway's raw materials". Retrieved 2 July 2023.
- ^ https://www.euractiv.com/section/energy-environment/news/great-news-eu-hails-discovery-of-massive-phosphate-rock-deposit-in-norway/
- ^ "China issues phosphate quotas to rein in fertiliser exports - analysts". Reuters. 15 July 2022.
- ^ "Top countries for Phosphate Fertilizer Imports".
- ^ "PHOSPHATE ROCK, usgs" (PDF).
- ^ Hochanadel, Dave (December 10, 2010). "Limited amount of total phosphorus actually feeds algae, study finds". Lake Scientist. Retrieved June 10, 2012.
[B]ioavailable phosphorus – phosphorus that can be utilized by plants and bacteria – is only a fraction of the total, according to Michael Brett, a UW engineering professor ...
- ^ S2CID 140680495.
- S2CID 129497587.
External links
- US Minerals Databrowser provides data graphics covering consumption, production, imports, exports and price for phosphate and 86 other minerals
- Phosphate: analyte monograph – The Association for Clinical Biochemistry and Laboratory Medicine



![[H 2PO 4]− Dihydrogen phosphate](http://upload.wikimedia.org/wikipedia/commons/thumb/d/db/2-dihydrogenphosphate-3D-balls.png/209px-2-dihydrogenphosphate-3D-balls.png)
![[HPO 4]2− Hydrogen phosphate](http://upload.wikimedia.org/wikipedia/commons/thumb/8/87/1-hydrogenphosphate-3D-balls.png/169px-1-hydrogenphosphate-3D-balls.png)
![[PO 4]3− Phosphate or orthophosphate](http://upload.wikimedia.org/wikipedia/commons/thumb/b/b0/0-phosphate-3D-balls.png/165px-0-phosphate-3D-balls.png)
![{\displaystyle K_{a1}={\frac {[{\ce {H+}}][{\ce {H2PO4-}}]}{[{\ce {H3PO4}}]}}\approx 7.5\times 10^{-3}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/eed347325ee75e9e7cec75402ae9a0d68c5aca9a)
![{\displaystyle K_{a2}={\frac {[{\ce {H+}}][{\ce {HPO4^2-}}]}{[{\ce {H2PO4-}}]}}\approx 6.2\times 10^{-8}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/3795cf914ff31fa4feb59c417cc8ae8da71ef519)
![{\displaystyle K_{a3}={\frac {[{\ce {H+}}][{\ce {PO4^3-}}]}{[{\ce {HPO4^2-}}]}}\approx 2.14\times 10^{-13}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/90d1ae9985ce6207490cf9e7a6ab1f6d001a8a24)
![{\displaystyle {\begin{aligned}{\frac {[{\ce {H2PO4-}}]}{[{\ce {H3PO4}}]}}&\approx 7.5\times 10^{4}\\[4pt]{\frac {[{\ce {HPO4^2-}}]}{[{\ce {H2PO4-}}]}}&\approx 0.62\\[4pt]{\frac {[{\ce {PO4^3-}}]}{[{\ce {HPO4^2-}}]}}&\approx 2.14\times 10^{-6}\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a7bfb55f5ed7d0ba3805ba68970255099eca8291)