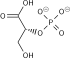
Phosphofructokinase 1
| 6-phosphofructokinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Phosphofructokinase | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||
| Symbol | PFK | ||||||||||
SCOP2 | 5pfk / SCOPe / SUPFAM | ||||||||||
| |||||||||||
Phosphofructokinase-1 (PFK-1) is one of the most important regulatory
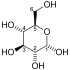
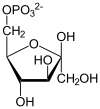
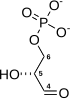
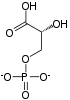
| β-D-fructose 6-phosphate | Phosphofructokinase 1 | β-D-fructose 1,6-bisphosphate | |

|

| ||
| ATP | ADP | ||
| |||
| Pi | H2O | ||
| Fructose bisphosphatase | |||
Structure
Mammalian PFK1 is a 340kd
PFK1 is an allosteric enzyme and has a structure similar to that of hemoglobin in so far as it is a dimer of a dimer.[5] One half of each dimer contains the ATP binding site whereas the other half the substrate (fructose-6-phosphate or (F6P)) binding site as well as a separate allosteric binding site.[6]
Each subunit of the tetramer is 319 amino acids and consists of two domains: one that binds the substrate ATP, and the other that binds fructose-6-phosphate. Each domain is a b barrel, and has cylindrical b sheet surrounded by alpha helices.
On the opposite side of the each subunit from each active site is the allosteric site, at the interface between subunits in the dimer. ATP and AMP compete for this site. The N-terminal domain has a catalytic role binding the ATP, and the C-terminal has a regulatory role [7]
Mechanism
PFK1 is an allosteric enzyme whose activity can be described using the
PFK1 belongs to the family of
Allosteric activators such as AMP and ADP bind to the allosteric site as to facilitate the formation of the R state by inducing structural changes in the enzyme. Similarly, inhibitors such as ATP and PEP bind to the same allosteric site and facilitate the formation of the T state, thereby inhibiting enzyme activity.
The hydroxyl oxygen of carbon 1 does a nucleophilic attack on the beta phosphate of ATP. These electrons are pushed to the anhydride oxygen between the beta and gamma phosphates of ATP.[10][11]

Regulation
PFK1 is the most important control site in the mammalian glycolytic pathway. This step is subject to extensive regulation since it is not only highly
PFK1 is
PFK1 is also inhibited by low pH levels which augment the inhibitory effect of ATP. The pH falls when muscle is functioning anaerobically and producing excessive quantities of lactic acid (although lactic acid is not itself the cause of the decrease in pH[12]). This inhibitory effect serves to protect the muscle from damage that would result from the accumulation of too much acid.[3]
Finally, PFK1 is allosterically inhibited by PEP, citrate, and ATP. Phosphoenolpyruvic acid is a product further downstream the glycolytic pathway. Although citrate does build up when the Krebs Cycle enzymes approach their maximum velocity, it is questionable whether citrate accumulates to a sufficient concentration to inhibit PFK-1 under normal physiological conditions[citation needed]. ATP concentration build up indicates an excess of energy and does have an allosteric modulation site on PFK1 where it decreases the affinity of PFK1 for its substrate.
PFK1 is
PFK activity is reduced through repression of synthesis by
The precise regulation of PFK1 prevents
Serotonin (5-HT) increases PFK by binding to the 5-HT(2A) receptor, causing the tyrosine residue of PFK to be phosphorylated via phospholipase C. This in turn redistributes PFK within the skeletal muscle cells. Because PFK regulates glycolytic flux, serotonin plays a regulatory role in glycolysis [13]
Genes
There are three phosphofructokinase genes in humans:
Clinical significance
A genetic mutation in the
Tarui disease is a glycogen storage disease with symptoms including muscle weakness (myopathy) and exercise induced cramping and spasms, myoglobinuria (presence of myoglobin in urine, indicating muscle destruction) and compensated hemolysis. ATP is a natural allosteric inhibitor of PFK, in order to prevent unnecessary production of ATP through glycolysis. However, a mutation in Asp(543)Ala can result in ATP having a stronger inhibitory effect (due to increased binding to PFK's inhibitory allosteric binding site).[15][16]
Phosphofructokinase mutation and cancer: In order for cancer cells to meet their energy requirements due to their rapid cell growth and division, they survive more effectively when they have a hyperactive phosphofructokinase 1 enzyme.[17][18] When cancer cells grow and divide quickly, they initially do not have as much blood supply, and can thus have hypoxia (oxygen deprivation), and this triggers O-GlcNAcylation at serine 529 of PFK. This modification inhibits PFK1 activity and supports cancer proliferation, in contrast with the view that high PFK1 activity is necessary for cancer. This may be due to redirecting glucose flux towards the pentose phosphate pathway to generate NADPH to detoxify reactive oxygen species.[19]
Herpes simplex type 1 and phosphofructokinase: Some viruses, including HIV, HCMV and Mayaro affect cellular metabolic pathways such as glycolysis by a MOI-dependent increase in the activity of PFK. The mechanism that Herpes increases PFK activity is by phosphorylating the enzyme at the serine residues. The HSV-1 induced glycolysis increases ATP content, which is critical for the virus's replication.[20]
See also
- PFK2(converts fructose 6-phosphate to fructose 2,6-bisphosphate through on site, or the opposite, on another site)
- PFP (reversibly interconverts fructose 6-phosphate and fructose 1,6-bisphosphate using inorganic pyrophosphate rather than ATP)
- Fructose bisphosphatase (hydrolyses fructose 1,6-bisphosphate to fructose 6-phosphate)
References
- ISSN 0014-4894.
- PMID 21124851.
- ^ ISBN 978-0-7167-8724-2.
- PMID 2970843.
- PMID 6115424.
- "The Glycolytic Enzymes". Protein Data Bank. Archived from the original on 2010-12-12.
- PMID 2975709.
- PMID 21241708.
- PMID 18763746.
- S2CID 4357039.
- PMID 23409118.
- PMID 22759530.
- PMID 16105823.
- S2CID 14570273.
- PMID 11949936.
- PMID 22995305.
- PMID 22995305.
- PMID 23454376.
- PMID 22964025.
- PMID 22923583.
- PMID 22542512.
External links
- Phosphofructokinase-1 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Proteopedia.org Phosphofructokinase
- Overview of all the structural information available in the PDB for UniProt: P08237 ( ATP-dependent 6-phosphofructokinase, muscle type) at the PDBe-KB.
- Overview of all the structural information available in the PDB for UniProt: Q01813 ( ATP-dependent 6-phosphofructokinase, platelet type) at the PDBe-KB.