Phosphoglycerate kinase
| Phosphoglycerate kinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Phosphoglycerate kinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
Phosphoglycerate kinase (
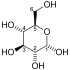
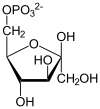
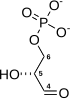
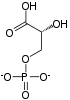
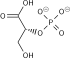
- 1,3-bisphosphoglycerate + ADP ⇌ glycerate 3-phosphate + ATP
Like all kinases it is a transferase. PGK is a major enzyme used in glycolysis, in the first ATP-generating step of the glycolytic pathway. In gluconeogenesis, the reaction catalyzed by PGK proceeds in the opposite direction, generating ADP and 1,3-BPG.
In humans, two
Biological function
PGK is present in all living organisms as one of the two ATP-generating enzymes in glycolysis. In the gluconeogenic pathway, PGK catalyzes the reverse reaction. Under biochemical
In the
PGK has been reported to exhibit thiol
The human isozyme PGK2, which is only expressed during spermatogenesis, was shown to be essential for sperm function in mice.[4]
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
Structure
Overview
PGK is found in all living organisms and its sequence has been
The enzyme has a tendency to exist in the open conformation with short periods of closure and catalysis, which allow for rapid
Role of magnesium
Mechanism

Without either substrate bound, PGK exists in an "open"
PGK proceeds through a charge-stabilized
In the
Regulation
The enzyme is activated by low concentrations of various multivalent anions, such as pyrophosphate, sulfate, phosphate, and citrate. High concentrations of MgATP and 3-PG activates PGK, while Mg2+ at high concentrations non-competitively inhibits the enzyme.[13]
PGK exhibits a wide specificity toward nucleotide substrates.[14] Its activity is inhibited by salicylates, which appear to mimic the enzyme's nucleotide substrate.[15]
Macromolecular crowding has been shown to increase PGK activity in both computer simulations and in vitro environments simulating a cell interior; as a result of crowding, the enzyme becomes more enzymatically active and more compact.[5]
Disease relevance
PGK1 overexpression has been associated with
Due to its wide specificity towards
Human isozymes
|
| ||||||||||||||||||||||||||||||
References
- ^ PMID 6765200.
- ^ PMID 22348148.
- ^ S2CID 4340557.
- PMID 19759366.
- ^ PMID 20921368.
- S2CID 41924983.
- ^ PMID 2124145.
- PMID 21349853.
- ^ PMID 23231058.
- PMID 20397725.
- S2CID 4321999.
- PMID 9521762.
- PMID 6030358.
- ^ PMID 21505655.
- PMID 343818.
- PMID 6689547.
- ^ S2CID 21111736.
- ^ NIH Genetics Home Reference
- S2CID 37605904.
- PMID 19688824.
- PMID 15450940.
External links
- Phosphoglycerate+kinase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Illustration at arizona.edu