Phosphorus trifluoride
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Phosphorus trifluoride
Phosphorus(III) fluoride Trifluorophosphane Trifluoridophosphorus Perfluorophosphane | |||
| Other names
Trifluorophosphine
Phosphorous fluoride TL-75 | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.029.098 | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| PF3 | |||
| Molar mass | 87.968971 g/mol | ||
| Appearance | colorless gas | ||
| Density | 3.91 g/L, gas | ||
| Melting point | −151.5 °C (−240.7 °F; 121.6 K) | ||
| Boiling point | −101.8 °C (−151.2 °F; 171.3 K) | ||
| slow hydrolysis | |||
| Structure | |||
| Trigonal pyramidal | |||
| 1.03 D | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | Non-flammable | ||
| Related compounds | |||
Other anions
|
Phosphane
| ||
Other cations
|
Nitrogen trifluoride Arsenic trifluoride Antimony trifluoride Bismuth trifluoride | ||
Related ligands
|
Carbon monoxide | ||
Related compounds
|
Phosphorus pentafluoride | ||
| Supplementary data page | |||
| Phosphorus trifluoride (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Phosphorus trifluoride (formula
metal complexes. As a ligand, it parallels carbon monoxide in metal carbonyls,[1] and indeed its toxicity is due to its binding with the iron in blood hemoglobin
in a similar way to carbon monoxide.
Physical properties
Phosphorus trifluoride has an F−P−F bond angle of approximately 96.3°.
standard enthalpy of formation of −945 kJ/mol (−226 kcal/mol). The phosphorus atom has a nuclear magnetic resonance chemical shift of 97 ppm (downfield of H3PO4
).
Properties
Phosphorus trifluoride
Lewis bases such as ammonia addition products (adducts) are formed, and PF3 is oxidized by oxidizing agents such as bromine or potassium permanganate
.
As a ligand for transition metals, PF3 is a strong π-acceptor.[2] It forms a variety of
dibenzenechromium
:
- Cr(C6H6)2 + 6 PF3 → Cr(PF3)6 + 2 C6H6
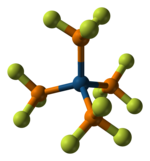 |

|
| Ball-and-stick model of [Pt(PF3)4] | Space-filling model of [Pt(PF3)4] |
Preparation
Phosphorus trifluoride is usually prepared from phosphorus trichloride via halogen exchange using various fluorides such as hydrogen fluoride, calcium fluoride, arsenic trifluoride, antimony trifluoride, or zinc fluoride:[6][7][8]
Biological activity
Phosphorus trifluoride is similar to carbon monoxide in that it is a gas which strongly binds to iron in hemoglobin, preventing the blood from absorbing oxygen.
Precautions
PF3 is highly toxic, comparable to phosgene.[9]
References
- PMID 15416738.
- ISBN 978-0-08-037941-8.
- ^ Nicholls, D. (1973). Complexes and First-Row Transition Elements. London: Macmillan Press.
- .
- .
- .
- ISBN 978-2-225-57123-7.
- ISBN 978-0-470-13259-3.
- ISBN 978-0-08-037941-8.
Further reading
- Toy, A. D. F. (1973). The Chemistry of Phosphorus. Oxford, UK: Pergamon Press.
- ISBN 978-0-08-037941-8.
- Lide, D. R., ed. (1990). Handbook of Chemistry and Physics (71st ed.). Ann Arbor, MI: CRC Press. ISBN 978-0-8493-0471-2.
- March, J. (1992). Advanced Organic Chemistry (4th ed.). New York: Wiley. p. 723. ISBN 978-0-471-60180-7.
- Stecher, P. G., ed. (1960). The Merck Index (7th ed.). Rahway, NJ, USA: Merck & Co.
- Holmes, R. R. (1960). "An Examination of the Basic Nature of the Trihalides of Phosphorus, Arsenic and Antimony". .



