Phosphoryl chloride
| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Phosphoryl trichloride[1] | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.030.030 | ||
| EC Number |
| ||
| 2272 | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1810 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| POCl3 | |||
| Molar mass | 153.32 g·mol−1 | ||
| Appearance | colourless liquid, fumes in moist air | ||
| Odor | pungent and musty | ||
| Density | 1.645 g/cm3, liquid | ||
| Melting point | 1.25 °C (34.25 °F; 274.40 K) | ||
| Boiling point | 105.8 °C (222.4 °F; 378.9 K) | ||
| Reacts | |||
| Solubility | highly soluble in benzene, chloroform, carbon disulfide, carbon tetrachloride | ||
| Vapor pressure | 40 mmHg (27 °C)[2] | ||
Refractive index (nD)
|
1.460 | ||
| Structure | |||
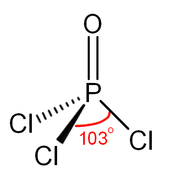
| Tetrahedral at the P atom | |||
| 2.54 D | |||
| Thermochemistry[3] | |||
Heat capacity (C)
|
138.8 J·mol−1·K−1 (liquid), 84.9 J·mol−1·K−1 (gas) | ||
Std molar
entropy (S⦵298) |
222.5 J·mol−1·K−1 (liquid), 325.5 J·mol−1·K−1 (gas) | ||
Std enthalpy of (ΔfH⦵298)formation |
−597.1 kJ·mol−1 (liquid), −558.5 kJ·mol−1 (gas) | ||
Gibbs free energy (ΔfG⦵)
|
−520.8 kJ·mol−1 (liquid), −512.9 kJ·mol−1(gas) | ||
Enthalpy of fusion (ΔfH⦵fus)
|
13.1 kJ·mol−1 | ||
Enthalpy of vaporization (ΔfHvap)
|
38.6 kJ·mol−1 | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
|||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H302, H314, H330, H372 | |||
| P260, P264, P270, P271, P280, P284, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P314, P320, P321, P330, P363, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
380 mg/kg (rat, oral) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none[2] | ||
REL (Recommended)
|
TWA 0.1 ppm (0.6 mg/m3) ST 0.5 ppm (3 mg/m3)[2] | ||
IDLH (Immediate danger) |
N.D.[2] | ||
| Safety data sheet (SDS) | ICSC 0190 | ||
| Related compounds | |||
Related compounds
|
| ||
| Supplementary data page | |||
| Phosphoryl chloride (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Phosphoryl chloride (commonly called phosphorus oxychloride) is a colourless liquid with the formula
Structure

Like phosphate, POCl3 is tetrahedral in shape.
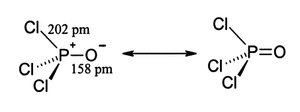
Phosphoryl chloride exists as neutral POCl3 molecules in the
Physical properties
It has a
Chemical properties
POCl3 reacts with water to give hydrogen chloride and phosphoric acid:
- O=PCl3 + 3 H2O → O=P(OH)3 + 3 HCl
Intermediates in the conversion have been isolated, including pyrophosphoryl chloride, O(−P(=O)Cl2)2.[9]
Upon treatment with excess
- O=PCl3 + 3 ROH → O=P(OR)3 + 3 HCl
Such reactions are often performed in the presence of an HCl acceptor such as pyridine or an amine.
POCl3 can also act as a
- POCl3 + TiCl4 → POCl3·TiCl4
The
POCl3 reacts with hydrogen bromide in the presence of Lewis-acidic catalysts to produce POBr3.
Preparation
Phosphoryl chloride can be prepared by many methods. Phosphoryl chloride was first reported in 1847 by the French chemist
By oxidation
The commercial method involves oxidation of phosphorus trichloride with oxygen:[11]
- 2 PCl3 + O2 → 2 POCl3
An alternative method involves the oxidation of phosphorus trichloride with potassium chlorate:[12]
- 3 PCl3 + KClO3 → 3 POCl3 + KCl
Oxygenations
The reaction of phosphorus pentachloride (PCl5) with phosphorus pentoxide (P4O10).
- 6 PCl5 + P4O10 → 10 POCl3
The reaction can be simplified by chlorinating a mixture of PCl3 and P4O10, generating the PCl5 in situ. The reaction of phosphorus pentachloride with boric acid or oxalic acid:[12]
Other methods
Reduction of tricalcium phosphate with carbon in the presence of chlorine gas:[13]
The reaction of phosphorus pentoxide with sodium chloride is also reported:[13]
- 2 P2O5 + 3 NaCl → 3 NaPO3 + POCl3
Uses
Phosphoryl chloride is used on an industrial scale for the manufacture of
In the semiconductor industry, POCl3 is used as a safe liquid phosphorus source in diffusion processes. The phosphorus acts as a dopant used to create
As a reagent
In the laboratory, POCl3 is a reagent in dehydrations. One example involves conversion of formamides to isonitriles (isocyanides);[14] primary amides to nitriles:[15]
- RC(O)NH2 + POCl3 → RCN + P(O)OHCl + 2 HCl
In a related reaction, certain aryl-substituted amides can be cyclized using the
Such reactions are believed to proceed via an imidoyl chloride. In certain cases, the imidoyl chloride is the final product. For example, pyridones and pyrimidones can be converted to chloro- derivatives such as 2-chloropyridines and 2-chloropyrimidines, which are intermediates in the pharmaceutical industry.[16]
In the
References
- ISBN 978-0-85404-182-4.
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0508". National Institute for Occupational Safety and Health (NIOSH).
- )
- OCLC 152398514.
- ^ .
- ^ Greenwood, N. N.; Earnshaw, A. (1997). Chemistry of the Elements (2nd ed.). Oxford: Butterworth-Heinemann.
- ISSN 0002-7863.
- ^ "Phosphoryl chloride".
- .
- ^ Wurtz, Adolphe (1847). "Sur l'acide sulfophosphorique et le chloroxyde de phosphore" [On monothiophosphoric acid and phosphoryl chloride]. Annales de Chimie et de Physique. 3rd series (in French). 20: 472–481.; see Chloroxyde de phosphore, pp. 477–481. (Note: Wurtz's empirical formulas are wrong because, like many chemists of his day, he used the wrong atomic mass for oxygen.)Roscoe, Henry Enfield; Schorlemmer, Carl; Cannell, John, eds. (1920). A Treatise on Chemistry. Vol. 1 (5th ed.). London, England: Macmillan and Co. p. 676.
- ^ ISBN 978-3527306732..
- ^ ISBN 0-07-049439-8.
- ^ ISBN 978-1-4398-1312-6.
- .
- ISBN 978-0-471-60180-7.
- ^ Elderfield, R. C. (ed.). Heterocyclic Compound. Vol. 6. New York, NY: John Wiley & Sons. p. 265.
- .
Further reading
- Handbook of Chemistry and Physics (71st ed.). Ann Arbor, MI: CRC Press. 1990.[ISBN missing]
- Stecher, Paul G. (1960). The Merck Index of Chemicals and Drugs (7th ed.). Rahway: OCLC 3653550.[ISBN missing]
- Wade, L. G. Jr (2005). Organic Chemistry (6th ed.). Upper Saddle River, NJ: Pearson/Prentice Hall. p. 477.[ISBN missing]
- Walker, B. J. (1972). Organophosphorus Chemistry. Harmondsworth: Penguin. pp. 101–116.[ISBN missing]
- "CDC – NIOSH Pocket Guide to Chemical Hazards".